Chonnam Med J.
2014 Apr;50(1):6-14. 10.4068/cmj.2014.50.1.6.
Gastroprotective Effects of Glutinous Rice Extract against Ethanol-, Indomethacin-, and Stress-induced Ulcers in Rats
- Affiliations
-
- 1Chonnam University Research Institute of Medical Sciences, Chonnam National University Medical School, Gwangju, Korea.
- 2Department of Biochemistry, Chonnam National University Medical School, Gwangju, Korea. bwahn@jnu.ac.kr
- 3Department of Internal Medicine, Chonnam National University Medical School, Gwangju, Korea.
- KMID: 1889756
- DOI: http://doi.org/10.4068/cmj.2014.50.1.6
Abstract
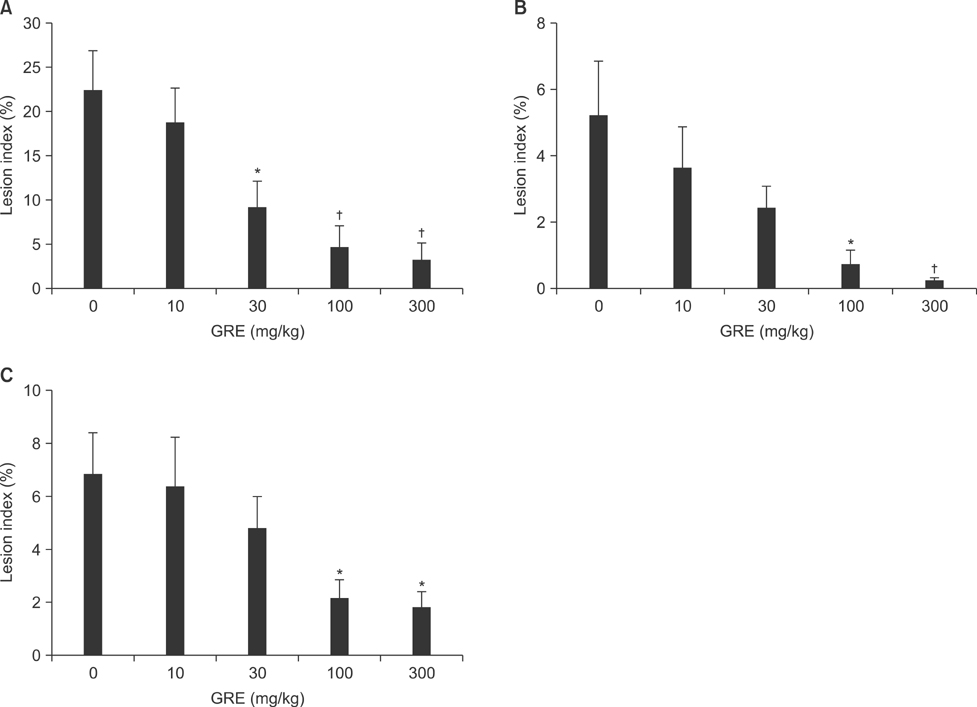
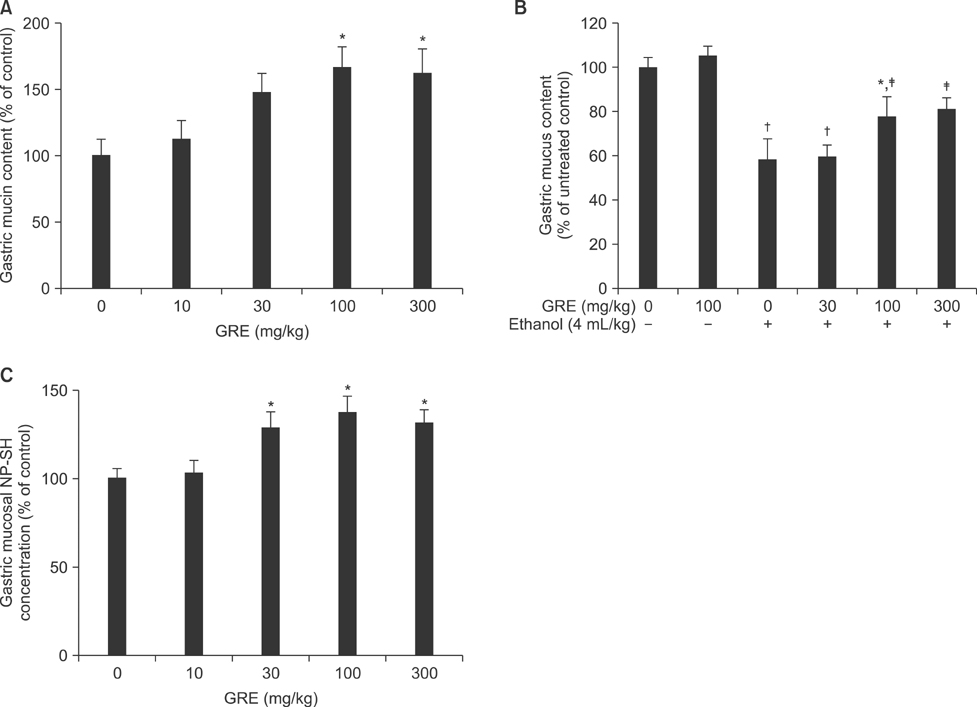
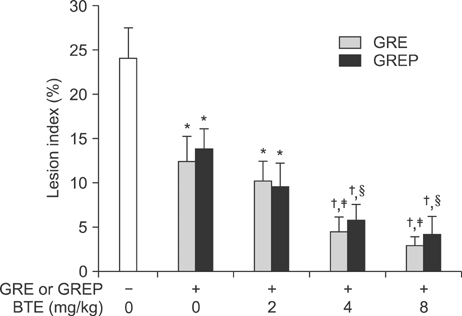
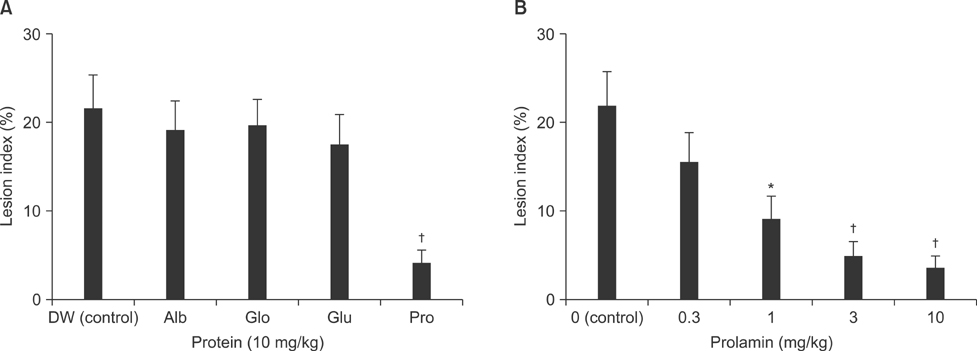
- This study was designed to evaluate the efficacy of an orally administered aqueous extract of glutinous rice (GRE) to protect against acute gastric mucosal lesions induced by ethanol, indomethacin, and water immersion restraint stress in rats and to characterize the active substances responsible for the protection. GRE was shown to dose-dependently prevent the gastric lesions induced by the above ulcerogenic treatments at doses of 30 to 300 mg/kg. GRE treatment increased the gastric mucin content and partially blocked the ethanol-induced depletion of the gastric mucus layer. Also, it increased the nonprotein sulfhydryl concentration in the gastric mucosa. The gastroprotective action of GRE was markedly enhanced by co-treatment with 4-8 mg/kg tea extracts. The activity of GRE was completely lost by heat treatment at 80degrees C for 3 min or treatment with 0.01% pepsin at 37degrees C for 1 h. Protein extraction studies indicated that prolamins are involved in the gastroprotective activity of GRE. Our results suggest that glutinous rice proteins are useful for the prevention and treatment of gastritis and peptic ulcer.
Keyword
MeSH Terms
Figure
Reference
-
1. Matsuhashi T, Otaka M, Odashima M, Jin M, Komatsu K, Wada I, et al. Protective effect of a novel rice extract against ethanol-induced gastric mucosal injury in rat. Dig Dis Sci. 2007; 52:434–441.
Article2. Furihata C, Ishida S, Ohta H, Tokuyama T, Katsuyama T, Ogita Z. Cytotoxicity of NaCl, a stomach tumor promoter, and prevention by rice extract in stomach mucosa of F344 rats. Cancer Detect Prev. 1996; 20:193–198.3. Murakami M, Ota H, Sugiyama A, Ishizone S, Maruta F, Akita N, et al. Suppressive effect of rice extract on Helicobacter pylori infection in a Mongolian gerbil model. J Gastroenterol. 2005; 40:459–466.
Article4. Wallace JL, Granger DN. The cellular and molecular basis of gastric mucosal defense. FASEB J. 1996; 10:731–740.
Article5. Brunton LL, Chabner BA, Knollmann BC. Pharmacotherapy of gastric acidity, peptic ulcers, and gastroesophageal reflux disease. In : Wallace JL, Sharkey KA, editors. Goodman & Gilman's The Pharmacological Basis of Therapeutics. 11th ed. New York: McGraw-Hill Education;2006. p. 967–981.6. Yamasaki K, Kanbe T, Chijiwa T, Ishiyama H, Morita S. Gastric mucosal protection by OPC-12759, a novel antiulcer compound, in the rat. Eur J Pharmacol. 1987; 142:23–29.
Article7. Chávez-Piña AE, Tapia-Alvarez GR, Navarrete A. Inhibition of endogenous hydrogen sulfide synthesis by PAG protects against ethanol-induced gastric damage in the rat. Eur J Pharmacol. 2010; 630:131–136.
Article8. Süleyman H, Akçay F, Altinkaynak K. The effect of nimesulide on the indomethacin- and ethanol-induced gastric ulcer in rats. Pharmacol Res. 2002; 45:155–158.
Article9. Park CH, Son HU, Son M, Lee SH. Protective effect of Acer mono Max. sap on water immersion restraint stress-induced gastric ulceration. Exp Ther Med. 2011; 2:843–848.
Article10. Yamasaki K, Ishiyama H, Imaizumi T, Kanbe T, Yabuuchi Y. Effect of OPC-12759, a novel antiulcer agent, on chronic and acute experimental gastric ulcer, and gastric secretion in rats. Jpn J Pharmacol. 1989; 49:441–448.
Article11. Bae DK, Park D, Lee SH, Yang G, Yang YH, Kim TK, et al. Different antiulcer activities of pantoprazole in stress, alcohol and pylorus ligation-induced ulcer models. Lab Anim Res. 2011; 27:47–52.
Article12. Ichikawa T, Ito Y, Saegusa Y, Iwai T, Goso Y, Ikezawa T, et al. Effects of combination treatment with famotidine and methylmethionine sulfonium chloride on the mucus barrier of rat gastric mucosa. J Gastroenterol Hepatol. 2009; 24:488–492.
Article13. Dubois M, Gilles KA, Hamilton JK, Rebers PA, Smith F. Colorimetric method for determination of sugar and related substances. Anal Chem. 1956; 28:350–356.14. Corne SJ, Morrissey SM, Woods RJ. Proceedings: a method for the quantitative estimation of gastric barrier mucus. J Physiol. 1974; 242:116P–117P.15. Szabo S, Trier JS, Frankel PW. Sulfhydryl compounds may mediate gastric cytoprotection. Science. 1981; 214:200–202.
Article16. Sedlak J, Lindsay RH. Estimation of total, protein-bound, and nonprotein sulfhydryl groups in tissue with Ellman's reagent. Anal Biochem. 1968; 25:192–205.
Article17. Ju ZY, Hettiarachchy NS, Rath N. Extraction, denaturation and hydrophobic Properties of Rice Flour Protein. J Food Sci. 2001; 66:229–232.
Article18. Slomiany A, Morita M, Sano S, Piotrowski J, Skrodzka D, Slomiany BL. Effect of ethanol on gastric mucus glycoprotein synthesis, translocation, transport, glycosylation, and secretion. Alcohol Clin Exp Res. 1997; 21:417–423.
Article19. Lee JH, Lee SJ, Choi YH, Chung KT, Jeong YK, Choi BT. Effects of mycelial culture of Phellinus linteus on ethanol-induced gastric ulcer in rats. Phytother Res. 2006; 20:396–402.
Article20. Adhikary B, Yadav SK, Roy K, Bandyopadhyay SK, Chattopadhyay S. Black tea and theaflavins assist healing of indomethacin-induced gastric ulceration in mice by antioxidative action. Evid Based Complement Alternat Med. 2011; 2011:pii: 546560. doi: 10.1155/2011/546560.21. Paul A, Goswami S, Santani D. Gastroprotective effects of β3-adrenoceptor agonists on water immersion plus restraint stress-induced gastric ulcer in rats. Indian J Pharmacol. 2004; 36:151–154.22. Goso Y, Ogata Y, Ishihara K, Hotta K. Effects of traditional herbal medicine on gastric mucin against ethanol-induced gastric injury in rats. Comp Biochem Physiol C Pharmacol Toxicol Endocrinol. 1996; 113:17–21.
Article23. Szelenyi I, Brune K. Possible role of oxygen free radicals in ethanol-induced gastric mucosal damage in rats. Dig Dis Sci. 1988; 33:865–871.
Article24. Naito Y, Yoshikawa T. Oxidative stress involvement and gene expression in indomethacin-induced gastropathy. Redox Rep. 2006; 11:243–253.
Article25. Nishida K, Ohta Y, Kobayashi T, Ishiguro I. Involvement of the xanthine-xanthine oxidase system and neutrophils in the development of acute gastric mucosal lesions in rats with water immersion restraint stress. Digestion. 1997; 58:340–351.
Article26. Park SW, Oh TY, Kim YS, Sim H, Park SJ, Jang EJ, et al. Artemisia asiatica extracts protect against ethanol-induced injury in gastric mucosa of rats. J Gastroenterol Hepatol. 2008; 23:976–984.
Article27. Yoshikawa T, Naito Y, Kishi A, Tomii T, Kaneko T, Iinuma S, et al. Role of active oxygen, lipid peroxidation, and antioxidants in the pathogenesis of gastric mucosal injury induced by indomethacin in rats. Gut. 1993; 34:732–737.
Article28. Nishida K, Ohta Y, Ishiguro I. Relation of inducible nitric oxide synthase activity to lipid peroxidation and nonprotein sulfhydryl oxidation in the development of stress-induced gastric mucosal lesions in rats. Nitric Oxide. 1998; 2:215–223.
Article29. Kaplan KA, Odabasoglu F, Halici Z, Halici M, Cadirci E, Atalay F, et al. Alpha-lipoic acid protects against indomethacin-induced gastric oxidative toxicity by modulating antioxidant system. J Food Sci. 2012; 77:H224–H230.
Article30. Szabo S, Nagy L, Plebani M. Glutathione, protein sulfhydryls and cysteine proteases in gastric mucosal injury and protection. Clin Chim Acta. 1992; 206:95–105.
Article31. Mutoh H, Hiraishi H, Ota S, Yoshida H, Ivey KJ, Terano A, et al. Protective role of intracellular glutathione against ethanol-induced damage in cultured rat gastric mucosal cells. Gastroenterology. 1990; 98:1452–1459.
Article32. Nagy L, Nagata M, Szabo S. Protein and non-protein sulfhydryls and disulfides in gastric mucosa and liver after gastrotoxic chemicals and sucralfate: possible new targets of pharmacologic agents. World J Gastroenterol. 2007; 13:2053–2060.
Article33. Walker MW, Kinter MT, Roberts RJ, Spitz DR. Nitric oxide-induced cytotoxicity: involvement of cellular resistance to oxidative stress and the role of glutathione in protection. Pediatr Res. 1995; 37:41–49.
Article34. Bilici M, Ozturk C, Dursun H, Albayrak F, Saglam MB, Uyanik A, et al. Protective effect of mirtazapine on indomethacin-induced ulcer in rats and its relationship with oxidant and antioxidant parameters. Dig Dis Sci. 2009; 54:1868–1875.
Article35. Avila JR, de la Lastra CA, Martin MJ, Motilva V, Luque I, Delgado D, et al. Role of endogenous sulphydryls and neutrophil infiltration in the pathogenesis of gastric mucosal injury induced by piroxicam in rats. Inflamm Res. 1996; 45:83–88.
Article36. Body SC, Sasame HA, Body MR. High concentrations of glutathione in glandular stomach: possible implications for carcinogenesis. Science. 1979; 205:1010–1012.
Article37. Boyd SC, Sasame HA, Boyd MR. Gastric glutathione depletion and acute ulcerogenesis by diethylmaleate given subcutaneously to rats. Life Sci. 1981; 28:2987–2992.
Article38. Jayaraj AP, Tovey FI, Clark CG, Rees KR, White JS, Lewin MR. The ulcerogenic and protective action of rice and rice fractions in experimental peptic ulceration. Clin Sci (Lond). 1987; 72:463–466.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Gastroprotective effects of irsogladine maleate on ethanol/hydrochloric acid induced gastric ulcers in mice
- The Impact of Kinds of Dietary Grain and Dietary Lipid Level on the Glucose Metabolism and Antithrombogenic Capacity of Full Grown Obesity Induced Rats
- Gastroprotective Effect of the Three Glucuronopyranoside Flavonoids in Rats
- Augmenting Effect of DA-9601 on Ghrelin in an Acute Gastric Injury Model
- Evaluation of black glutinous rice (Oryza sativa L) extract as a novel nuclear stain for human sperm head assessment by microscopic examination