Cancer Res Treat.
2008 Sep;40(3):101-105.
Phase II Study of Gemcitabine plus Cisplatin in Patients with Anthracycline- and Taxane- Pretreated Metastatic Breast Cancer
- Affiliations
-
- 1Department of Internal Medicine, Dong-A University College of Medicine, Busan, Korea. kimhj@dau.ac.kr
- 2Department of Pathology, Dong-A University College of Medicine, Busan, Korea.
- 3Department of Diagnostic Radiology, Dong-A University College of Medicine, Busan, Korea.
- 4Department of Radiation Oncology, Dong-A University College of Medicine, Busan, Korea.
- 5Department of Surgery, Dong-A University College of Medicine, Busan, Korea.
Abstract
-
PURPOSE: Metastatic breast cancer patients are usually exposed to taxane and anthracycline as neoadjuvant, adjuvant and palliative chemotherapeutic agents. This study was designed to determine the efficacy and safety of the use of a gemcitabine and cisplatin (GP) combination treatment in patients with metastatic breast cancer that were pretreated with anthracycline and taxane.
MATERIALS AND METHODS
We evaluated the use of a GP regimen (1,000 mg/m2 gemcitabine administered on days 1 and 8 plus 60 mg/m2 cisplatin administered on day 1 every 3 weeks) in 38 breast cancer patients who had received prior chemotherapy with anthracycline and taxane as an adjuvant or neoadjuvant therapy, or as a palliative therapy.
RESULTS
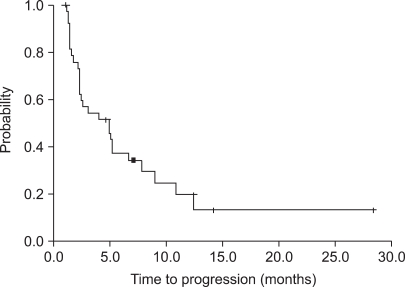
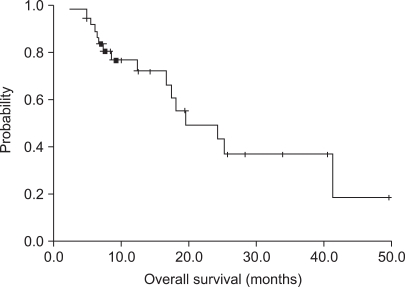
The median patient age was 49 years (age range, 35~69 years). The overall response rate was 28.9% in 11 patients (95% confidence interval [CI], 14~44%). The median time to progression was 5.2 months (95% CI, 3.6~6.8 months). Median survival was 19.5 months (95% CI, 11.2~27.8 months). Major grade 3/4 hematological toxicity was due to leukopenia (36 of 157 cycles, 23.1%). Non-hematological toxicity was rarely severe; grade1/2 nausea and vomiting were observed in 37.8% of the patients. There were no treatment related deaths.
CONCLUSIONS
Our results suggest that the use of gemcitabine plus cisplatin appears to be effective and has an acceptable toxicity profile in patients with advanced breast cancer that have been pretreated with anthracycline and taxane.
Keyword
MeSH Terms
Figure
Reference
-
1. Shin HR, Jung KW, Won YJ, Park JG. 2002 annual report of the Korea Central Cancer Registry: based on registered data from 139 hospitals. Cancer Res Treat. 2004; 36:103–114.
Article2. Wirk B, Perez E. Role of gemcitabine in breast cancer management: an update. Semin Oncol. 2006; 33:S6–S14. PMID: 16472712.
Article3. Hortobagyi GN. Treatment of breast cancer. N Engl J Med. 1998; 339:974–984. PMID: 9753714.
Article4. Seidman AD. Monotherapy options in the management of metastatic breast cancer. Semin Oncol. 2003; 30(2 Suppl 3):6–10. PMID: 12722018.
Article5. Heinemann V. Role of gemcitabine in the treatment of advanced and metastatic breast cancer. Oncology. 2003; 64:191–206. PMID: 12697958.
Article6. van Moorsel CJ, Veerman G, Bergman AM, Guechev A, Vermorken JB, Postmus PE, et al. Combination chemotherapy studies with gemcitabine. Semin Oncol. 1997; 24:S7-17–S7-23. PMID: 9194475.7. Achanta G, Pelicano H, Feng L, Plunkett W, Huang P. Interaction of p53 and DNA-PK in response to nucleoside analogues: potential role as a sensor complex for DNA damage. Cancer Res. 2001; 61:8723–8729. PMID: 11751391.8. Nagourney RA, Link JS, Blitzer JB, Forsthoff C, Evans SS. Gemcitabine plus cisplatin repeating doublet therapy in previously treated, relapsed breast cancer patients. J Clin Oncol. 2000; 18:2245–2249. PMID: 10829044.
Article9. Miller AB, Hoogstraten B, Staquet M, Winkler A. Reporting results of cancer treatment. Cancer. 1981; 47:207–214. PMID: 7459811.
Article10. Blackstein M, Vogel CL, Ambinder R, Cowan J, Iglesias J, Melemed A. Gemcitabine as first-line therapy in patients with metastatic breast cancer: a phase II trial. Oncology. 2002; 62:2–8. PMID: 11810037.
Article11. Brodowicz T, Kostler WJ, Moslinger R, Tomek S, Vaclavik I, Herscovici V, et al. Single-agent gemcitabine as second- and third-line treatment in metastatic breast cancer. Breast. 2000; 9:338–342. PMID: 14965758.
Article12. Spielmann M, Llombart-Cussac A, Kalla S, Espie M, Namer M, Ferrero JM, et al. Single-agent gemcitabine is active in previously treated metastatic breast cancer. Oncology. 2001; 60:303–307. PMID: 11408796.
Article13. Possinger K, Kaufmann M, Coleman R, Stuart NS, Helsing M, Ohnmacht U, et al. Phase II study of gemcitabine as first-line chemotherapy in patients with advanced or metastatic breast cancer. Anticancer Drugs. 1999; 10:155–162. PMID: 10211545.
Article14. Martin M. Platinum compounds in the treatment of advanced breast cancer. Clin Breast Cancer. 2001; 2:190–208. PMID: 11899413.15. Heinemann V, Stemmler HJ, Wohlrab A, Bosse D, Losem C, Kahlert S, et al. High efficacy of gemcitabine and cisplatin in patients with predominantly anthracycline- and taxane-pretreated metastatic breast cancer. Cancer Chemother Pharmacol. 2006; 57:640–646. PMID: 16163537.
Article16. Smith IE. Overview of gemcitabine activity in advanced breast cancer. Semin Oncol. 2006; 33(3 Suppl 9):S19–S23. PMID: 16797378.
Article17. Valenza R, Leonardi V, Gebbia V, Agostara B. Gemcitabine and vinorelbine in pretreated advanced breast cancer: a pilot study. Ann Oncol. 2000; 11:495–496. PMID: 10847474.
Article18. Stathopoulos GP, Rigatos SK, Pergantas N, Tsavdarides D, Athanasiadis I, Malamos NA, et al. Phase II trial of biweekly administration of vinorelbine and gemcitabine in pretreated advanced breast cancer. J Clin Oncol. 2002; 20:37–41. PMID: 11773151.
Article19. Nicolaides C, Dimopoulos MA, Samantas E, Bafaloukos D, Kalofonos C, Fountzilas G, et al. Gemcitabine and vinorelbine as second-line treatment in patients with metastatic breast cancer progressing after first-line taxane-based chemotherapy: a phase II study conducted by the Hellenic Cooperative Oncology Group. Ann Oncol. 2000; 11:873–875. PMID: 10997817.
Article20. Fountzilas G, Christodoulou C, Tsavdaridis D, Kalogera-Fountzila A, Aravantinos G, Razis E, et al. Paclitaxel and gemcitabine, as first-line chemotherapy, combined with trastuzumab in patients with advanced breast cancer: a phase II study conducted by the Hellenic Cooperative Oncology Group (HeCOG). Cancer Invest. 2004; 22:655–662. PMID: 15581045.
Article21. Fountzilas G, Nicolaides C, Bafaloukos D, Kalogera-Fountzila A, Kalofonos H, Samelis G, et al. Docetaxel and gemcitabine in anthracycline-resistant advanced breast cancer: a Hellenic Cooperative Oncology Group Phase II study. Cancer Invest. 2000; 18:503–509. PMID: 10923097.
Article22. Laufman LR, Spiridonidis CH, Pritchard J, Roach R, Zangmeister J, Larrimer N, et al. Monthly docetaxel and weekly gemcitabine in metastatic breast cancer: a phase II trial. Ann Oncol. 2001; 12:1259–1264. PMID: 11697837.
Article23. Alexopoulos A, Tryfonopoulos D, Karamouzis MV, Gerasimidis G, Karydas I, Kandilis K, et al. Evidence for in vivo synergism between docetaxel and gemcitabine in patients with metastatic breast cancer. Ann Oncol. 2004; 15:95–99. PMID: 14679126.
Article24. Murad AM. Paclitaxel and gemcitabine as salvage treatment in metastatic breast cancer. Oncology (Williston Park). 2003; 17(12 Suppl 14):26–32. PMID: 14768402.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Treatment with Cisplatin and Etoposide Chemotherapy in Patient with Metastatic Breast Cancer
- Gemcitabine and Vinorelbine Combination Chemotherapy in Anthracycline- and Taxane-pretreated Advanced Breast Cancer
- A Phase II Study of Gemcitabine Monotherapy in Breast Cancer Patients Refractory to Anthracycline and Taxane
- Outcomes of Palliative Weekly Low-Dose Gemcitabine-Cisplatin Chemotherapy in Anthracycline- and Taxane- Pretreated Metastatic Breast Cancer Patients
- Maintenance chemotherapy after 6 cycles of platinum-doublet regimen in anthracycline-and taxane-pretreated metastatic breast cancer