Korean J Ophthalmol.
2013 Dec;27(6):446-453. 10.3341/kjo.2013.27.6.446.
In Vitro Effects of Preservative-free and Preserved Prostaglandin Analogs on Primary Cultured Human Conjunctival Fibroblast Cells
- Affiliations
-
- 1Cheil Eye Reserch Institute, Cheil Eye Hospital, Daegu, Korea. eyepark9@naver.com
- 2Developmental Biology Laboratory, Department of Biology, College of Natural Sciences, Kyungpook National University, Daegu, Korea.
- KMID: 1792081
- DOI: http://doi.org/10.3341/kjo.2013.27.6.446
Abstract
- PURPOSE
Long-term use of topical medication is needed for glaucoma treatment. One of the most commonly prescribed classes of hypotensive agents are prostaglandin analogs (PGs) used as both first-line monotherapy; as well as in combination therapy with other hypotensive agents. Several side effects of eye drops can be caused by preservatives. The purpose of this study was to evaluate the effects of PGs with varying concentrations of benzalkonium chloride (BAC), alternative preservatives, or no preservatives on human conjunctival fibroblast cells.
METHODS
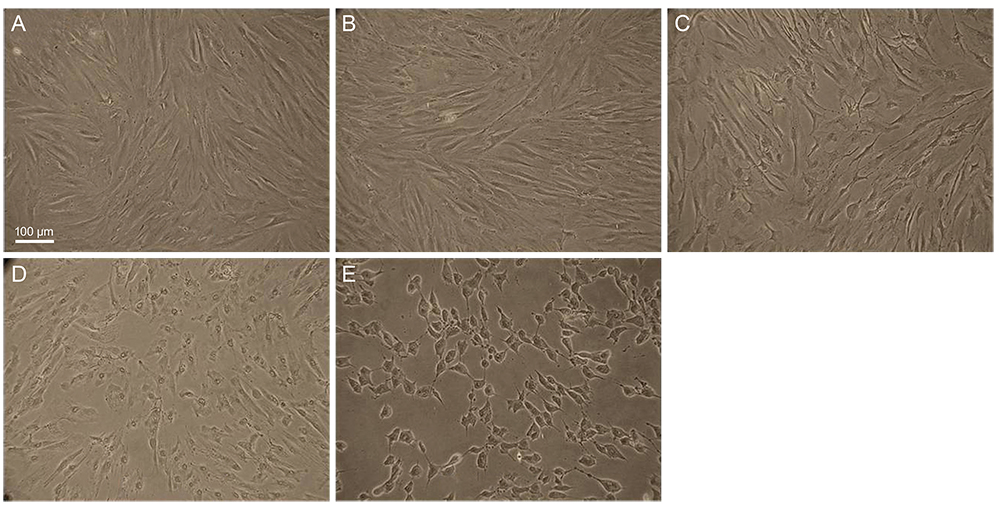
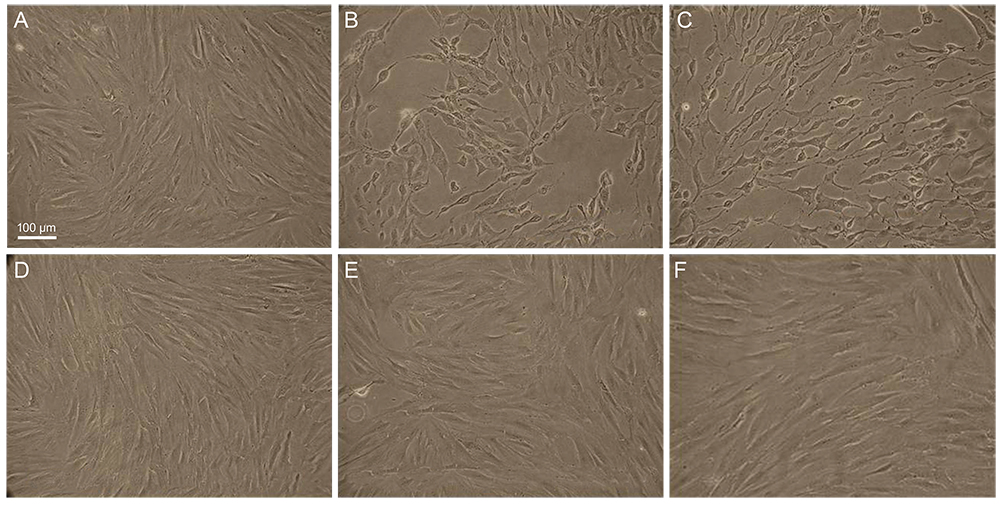
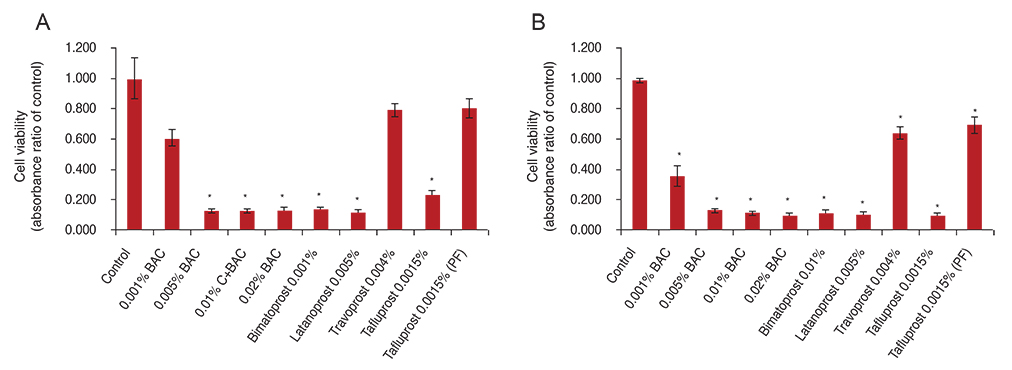
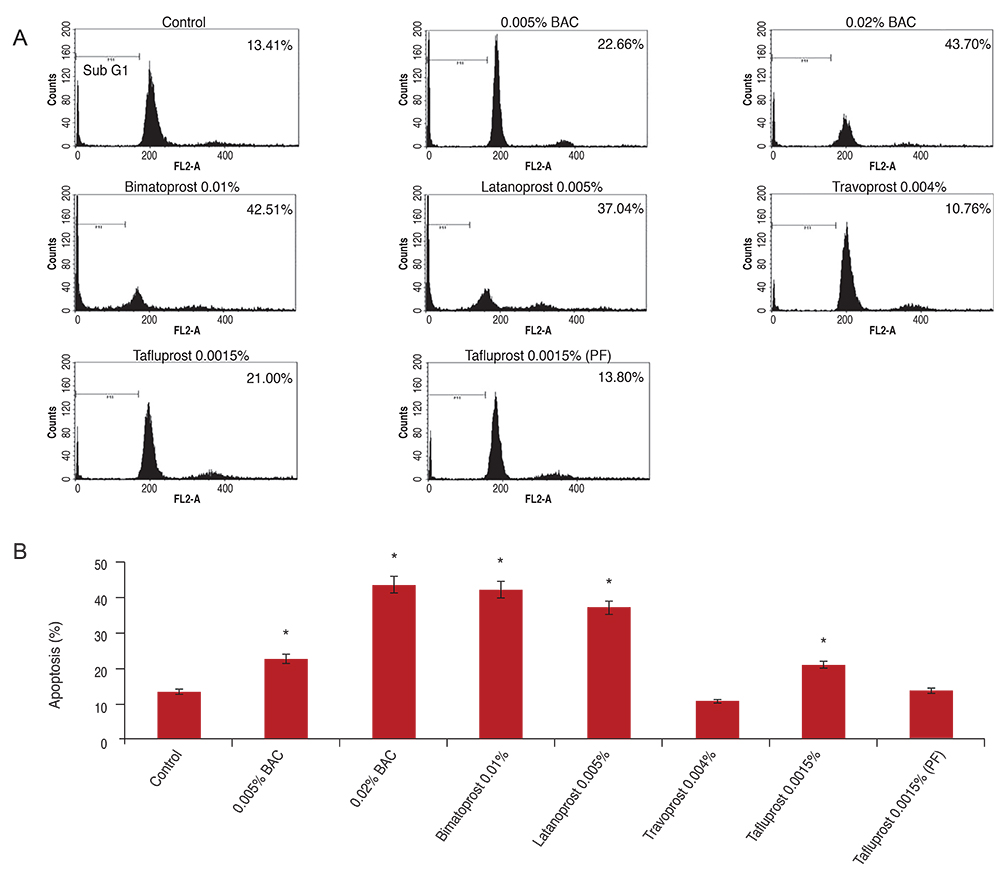
Primary human conjunctival fibroblast cells were used in these experiments. Cells were exposed to the following drugs: BAC at different concentrations, bimatoprost 0.01% (with BAC 0.02%), latanoprost 0.005% (with BAC 0.02%), tafluprost 0.0015% with/without 0.001% BAC and travoprost 0.004% (with 0.001% Polyquad) for 15 and 30 minutes. Cell cytotoxicity was evaluated by phase-contrast microscopy to monitor morphological changes of cells, Counting Kit-8 (CCK-8) assay to cell viability, and fluorescent activated cell sorting (FACS) analysis to measure apoptosis.
RESULTS
BAC caused cell shrinkage and detachment from the plate in a dose-dependent manner. Morphological changes were observed in cells treated with bimatoprost 0.01% and latanoprost 0.005%. However, mild cell shrinkage was noted in cells treated with tafluprost 0.0015%, while a non-toxic effect was noted with travoprost 0.004% and preservative-free tafluprost 0.0015%. CCK-8 assay and FACS analysis showed all groups had a significantly decreased cell viability and higher apoptosis rate compared with the control group. However, travoprost 0.004% and preservative-free tafluprost 0.0015% showed lower cytotoxicity and apoptosis rate than other drugs.
CONCLUSIONS
This in vitro study revealed that BAC-induced cytotoxicity is dose-dependent, although it is important to emphasize that the clinical significance of toxicity differences observed among the different PGs formulations has not yet been firmly established. Alternatively preserved or preservative-free glaucoma medications seem to be a reasonable and viable alternative to those preserved with BAC.
MeSH Terms
Figure
Reference
-
1. Noecker RJ, Herrygers LA, Anwaruddin R. Corneal and conjunctival changes caused by commonly used glaucoma medications. Cornea. 2004; 23:490–496.2. Baudouin C, Riancho L, Warnet JM, Brignole F. In vitro studies of antiglaucomatous prostaglandin analogues: travoprost with and without benzalkonium chloride and preserved latanoprost. Invest Ophthalmol Vis Sci. 2007; 48:4123–4128.3. Ammar DA, Noecker RJ, Kahook MY. Effects of benzalkonium chloride-preserved, polyquad-preserved, and sof-Zia-preserved topical glaucoma medications on human ocular epithelial cells. Adv Ther. 2010; 27:837–845.4. Liang H, Baudouin C, Pauly A, Brignole-Baudouin F. Conjunctival and corneal reactions in rabbits following short- and repeated exposure to preservative-free tafluprost, commercially available latanoprost and 0.02% benzalkonium chloride. Br J Ophthalmol. 2008; 92:1275–1282.5. Brasnu E, Brignole-Baudouin F, Riancho L, et al. In vitro effects of preservative-free tafluprost and preserved latanoprost, travoprost, and bimatoprost in a conjunctival epithelial cell line. Curr Eye Res. 2008; 33:303–312.6. Guenoun JM, Baudouin C, Rat P, et al. In vitro study of inflammatory potential and toxicity profile of latanoprost, travoprost, and bimatoprost in conjunctiva-derived epithelial cells. Invest Ophthalmol Vis Sci. 2005; 46:2444–2450.7. Baudouin C. Allergic reaction to topical eyedrops. Curr Opin Allergy Clin Immunol. 2005; 5:459–463.8. Broadway DC, Grierson I, O'Brien C, Hitchings RA. Adverse effects of topical antiglaucoma medication. II. The outcome of filtration surgery. Arch Ophthalmol. 1994; 112:1446–1454.9. Lee JK, Ryu YH. The effect of antiglaucoma medication on cultured human conjunctival epithelial cells. J Korean Ophthalmol Soc. 2006; 47:1811–1818.10. Noecker R. Effects of common ophthalmic preservatives on ocular health. Adv Ther. 2001; 18:205–215.11. De Saint Jean M, Brignole F, Bringuier AF, et al. Effects of benzalkonium chloride on growth and survival of Chang conjunctival cells. Invest Ophthalmol Vis Sci. 1999; 40:619–630.12. Sharif NA, Kelly CR, Crider JY, et al. Ocular hypotensive FP prostaglandin (PG) analogs: PG receptor subtype binding affinities and selectivities, and agonist potencies at FP and other PG receptors in cultured cells. J Ocul Pharmacol Ther. 2003; 19:501–515.13. Ota T, Murata H, Sugimoto E, et al. Prostaglandin analogues and mouse intraocular pressure: effects of tafluprost, latanoprost, travoprost, and unoprostone, considering 24-hour variation. Invest Ophthalmol Vis Sci. 2005; 46:2006–2011.14. Kahook MY, Noecker RJ. Comparison of corneal and conjunctival changes after dosing of travoprost preserved with sofZia, latanoprost with 0.02% benzalkonium chloride, and preservative-free artificial tears. Cornea. 2008; 27:339–343.15. Horsley MB, Kahook MY. Effects of prostaglandin analog therapy on the ocular surface of glaucoma patients. Clin Ophthalmol. 2009; 3:291–295.16. Ammar DA, Noecker RJ, Kahook MY. Effects of benzalkonium chloride- and polyquad-preserved combination glaucoma medications on cultured human ocular surface cells. Adv Ther. 2011; 28:501–510.17. Brignole-Baudouin F, Riancho L, Liang H, et al. In vitro comparative toxicology of polyquad-preserved and benzalkonium chloride-preserved travoprost/timolol fixed combination and latanoprost/timolol fixed combination. J Ocul Pharmacol Ther. 2011; 27:273–280.18. De Saint Jean M, Debbasch C, Rahmani M, et al. Fas- and interferon gamma-induced apoptosis in Chang conjunctival cells: further investigations. Invest Ophthalmol Vis Sci. 2000; 41:2531–2543.19. Han SB, Shin YJ, Hyon JY, Wee WR. Cytotoxicity of vorico nazole on cultured human corneal endothelial cells. Antimicrob Agents Chemother. 2011; 55:4519–4523.20. Ishiyama M, Tominaga H, Shiga M, et al. A combined assay of cell viability and in vitro cytotoxicity with a highly water-soluble tetrazolium salt, neutral red and crystal violet. Biol Pharm Bull. 1996; 19:1518–1520.21. Lu TH, Hsieh SY, Yen CC, et al. Involvement of oxidative stress-mediated ERK1/2 and p38 activation regulated mitochondria-dependent apoptotic signals in methylmercury-induced neuronal cell injury. Toxicol Lett. 2011; 204:71–80.22. Tripathi BJ, Tripathi RC, Kolli SP. Cytotoxicity of ophthalmic preservatives on human corneal epithelium. Lens Eye Toxic Res. 1992; 9:361–375.23. Baudouin C, Labbe A, Liang H, et al. Preservatives in eyedrops: the good, the bad and the ugly. Prog Retin Eye Res. 2010; 29:312–334.24. Asada H, Takaoka-Shichijo Y, Nakamura M, Kimura A. Optimization of benzalkonium chloride concentration in 0.0015% tafluprost ophthalmic solution from the points of ocular surface safety and preservative efficacy. Yakugaku Zasshi. 2010; 130:867–871.25. Jaenen N, Baudouin C, Pouliquen P, et al. Ocular symptoms and signs with preserved and preservative-free glaucoma medications. Eur J Ophthalmol. 2007; 17:341–349.26. Nakagawa S, Usui T, Yokoo S, et al. Toxicity evaluation of antiglaucoma drugs using stratified human cultivated corneal epithelial sheets. Invest Ophthalmol Vis Sci. 2012; 53:5154–5160.27. Liang H, Baudouin C, Labbe A, et al. Conjunctiva-associated lymphoid tissue (CALT) reactions to antiglaucoma prostaglandins with or without BAK-preservative in rabbit acute toxicity study. PLoS One. 2012; 7:e33913.28. Liang H, Brignole-Baudouin F, Riancho L, Baudouin C. Reduced in vivo ocular surface toxicity with polyquad-preserved travoprost versus benzalkonium-preserved travoprost or latanoprost ophthalmic solutions. Ophthalmic Res. 2012; 48:89–101.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Efficacy of Preservative-free Latanoprost in Normal-tension Glaucoma with Mild to Moderate Dry Eye
- Effects of Corneal Toxicity and Conjunctival Injection of Preservative-free 0.1% Fluorometholone after Pediatric Strabismus Surgery
- Efficacy and Safety of Preservative-free Latanoprost Eyedrops Compared with Preserved Prostaglandin Analogues in Patients with Open-angle Glaucoma
- Effect of Preservative-free Artificial Eye Drop on Human Corneal Epithelial Cell in vitro
- Co-cultivation of Adenovirus with Cultivated Conjunctival Epithelial Cell: Morphological Changes in Infected Cell