J Korean Med Sci.
2014 Jul;29(7):926-933. 10.3346/jkms.2014.29.7.926.
Clinical Significance of Previously Cryptic Copy Number Alterations and Loss of Heterozygosity in Pediatric Acute Myeloid Leukemia and Myelodysplastic Syndrome Determined Using Combined Array Comparative Genomic Hybridization plus Single-Nucleotide Polymorphism Microarray Analyses
- Affiliations
-
- 1Division of Pediatric Hematology/Oncology, Asan Medical Center Children's Hospital, Department of Pediatrics, University of Ulsan College of Medicine, Seoul, Korea. jjseo@amc.seoul.kr
- 2Genome Research Center, Asan Medical Center, Seoul, Korea.
- 3Department of Laboratory Medicine, Asan Medical Center, University of Ulsan College of Medicine, Seoul, Korea.
- KMID: 1789951
- DOI: http://doi.org/10.3346/jkms.2014.29.7.926
Abstract
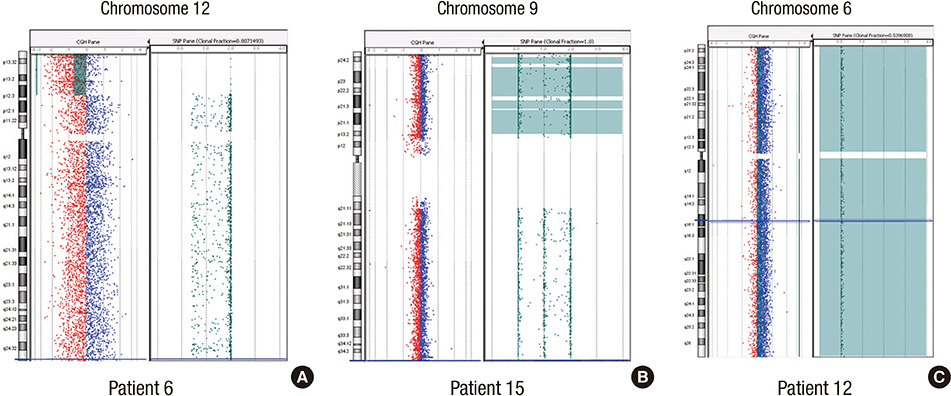
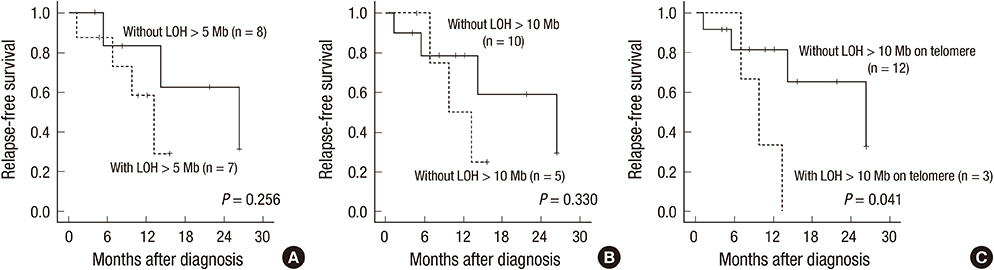
- The combined array comparative genomic hybridization plus single-nucleotide polymorphism microarray (CGH+SNP microarray) platform can simultaneously detect copy number alterations (CNA) and copy-neutral loss of heterozygosity (LOH). Eighteen children with acute myeloid leukemia (AML) (n=15) or myelodysplastic syndrome (MDS) (n=3) were studied using CGH+SNP microarray to evaluate the clinical significance of submicroscopic chromosomal aberrations. CGH+SNP microarray revealed CNAs at 14 regions in 9 patients, while metaphase cytogenetic (MC) analysis detected CNAs in 11 regions in 8 patients. Using CGH+SNP microarray, LOHs>10 Mb involving terminal regions or the whole chromosome were detected in 3 of 18 patients (17%). CGH+SNP microarray revealed cryptic LOHs with or without CNAs in 3 of 5 patients with normal karyotypes. CGH+SNP microarray detected additional cryptic CNAs (n=2) and LOHs (n=5) in 6 of 13 patients with abnormal MC. In total, 9 patients demonstrated additional aberrations, including CNAs (n=3) and/or LOHs (n=8). Three of 15 patients with AML and terminal LOH>10 Mb demonstrated a significantly inferior relapse-free survival rate (P=0.041). This study demonstrates that CGH+SNP microarray can simultaneously detect previously cryptic CNAs and LOH, which may demonstrate prognostic implications.
Keyword
MeSH Terms
-
Adolescent
Child
Child, Preschool
Chromosome Aberrations
*Comparative Genomic Hybridization
DNA/*analysis/metabolism
DNA Copy Number Variations
Female
Hematopoietic Stem Cell Transplantation
Humans
Infant
Kaplan-Meier Estimate
Leukemia, Myeloid, Acute/*diagnosis/*genetics/therapy
Loss of Heterozygosity
Male
Myelodysplastic Syndromes/*diagnosis/*genetics/therapy
*Oligonucleotide Array Sequence Analysis
Polymorphism, Single Nucleotide
Real-Time Polymerase Chain Reaction
Transplantation, Homologous
DNA
Figure
Reference
-
1. Grimwade D, Mrózek K. Diagnostic and prognostic value of cytogenetics in acute myeloid leukemia. Hematol Oncol Clin North Am. 2011; 25:1135–1161.2. Haase D. Cytogenetic features in myelodysplastic syndromes. Ann Hematol. 2008; 87:515–526.3. Theilgaard-Mönch K, Boultwood J, Ferrari S, Giannopoulos K, Hernandez-Rivas JM, Kohlmann A, Morgan M, Porse B, Tagliafico E, Zwaan CM, et al. Gene expression profiling in MDS and AML: potential and future avenues. Leukemia. 2011; 25:909–920.4. Mawad R, Estey EH. Acute myeloid leukemia with normal cytogenetics. Curr Oncol Rep. 2012; 14:359–368.5. Kang HJ, Lee JW, Kho SH, Kim MJ, Seo YJ, Kim H, Shin HY, Ahn HS. High transcript level of FLT3 associated with high risk of relapse in pediatric acute myeloid leukemia. J Korean Med Sci. 2010; 25:841–845.6. Cho YU, Chi HS, Park CJ, Jang S, Seo EJ. Rapid detection of prognostically significant fusion transcripts in acute leukemia using simplified multiplex reverse transcription polymerase chain reaction. J Korean Med Sci. 2012; 27:1155–1161.7. Hong SD, Kim YK, Kim HN, Lee SR, Ahn JS, Yang DH, Lee JJ, Lee IK, Shin MG, Kim HJ. Treatment outcome of all-trans retinoic acid/anthracycline combination chemotherapy and the prognostic impact of FLT3/ITD mutation in acute promyelocytic leukemia patients. Korean J Hematol. 2011; 46:24–30.8. O'Keefe C, McDevitt MA, Maciejewski JP. Copy neutral loss of heterozygosity: a novel chromosomal lesion in myeloid malignancies. Blood. 2010; 115:2731–2739.9. Makishima H, Maciejewski JP. Pathogenesis and consequences of uniparental disomy in cancer. Clin Cancer Res. 2011; 17:3913–3923.10. Bullinger L, Fröhling S. Array-based cytogenetic approaches in acute myeloid leukemia: clinical impact and biological insights. Semin Oncol. 2012; 39:37–46.11. Maciejewski JP, Tiu RV, O'Keefe C. Application of array-based whole genome scanning technologies as a cytogenetic tool in haematological malignancies. Br J Haematol. 2009; 146:479–488.12. Suela J, Alvarez S, Cigudosa JC. DNA profiling by arrayCGH in acute myeloid leukemia and myelodysplastic syndromes. Cytogenet Genome Res. 2007; 118:304–309.13. Swerdlow SH, Campo E, Harris NL, Jaffe ES, Pileri SA, Stein H, Thiele J, Vardiman JW. WHO classification of tumours of haematopoietic and lymphoid tissues. 4th ed. Lyon: IARC;2008.14. Schaffer L, Tommerup N. An International System for Human Cytogenetic Nomenclature. 2nd ed. Basel: Karger;2009.15. Tiu RV, Gondek LP, O'Keefe CL, Huh J, Sekeres MA, Elson P, McDevitt MA, Wang XF, Levis MJ, Karp JE, et al. New lesions detected by single nucleotide polymorphism array-based chromosomal analysis have important clinical impact in acute myeloid leukemia. J Clin Oncol. 2009; 27:5219–5226.16. Thiel A, Beier M, Ingenhag D, Servan K, Hein M, Moeller V, Betz B, Hildebrandt B, Evers C, Germing U, et al. Comprehensive array CGH of normal karyotype myelodysplastic syndromes reveals hidden recurrent and individual genomic copy number alterations with prognostic relevance. Leukemia. 2011; 25:387–399.17. Bullinger L, Krönke J, Schön C, Radtke I, Urlbauer K, Botzenhardt U, Gaidzik V, Carió A, Senger C, Schlenk RF, et al. Identification of acquired copy number alterations and uniparental disomies in cytogenetically normal acute myeloid leukemia using high-resolution single-nucleotide polymorphism analysis. Leukemia. 2010; 24:438–449.18. Akagi T, Ogawa S, Dugas M, Kawamata N, Yamamoto G, Nannya Y, Sanada M, Miller CW, Yung A, Schnittger S, et al. Frequent genomic abnormalities in acute myeloid leukemia/myelodysplastic syndrome with normal karyotype. Haematologica. 2009; 94:213–223.19. Rücker FG, Bullinger L, Schwaenen C, Lipka DB, Wessendorf S, Fröhling S, Bentz M, Miller S, Scholl C, Schlenk RF, et al. Disclosure of candidate genes in acute myeloid leukemia with complex karyotypes using microarray-based molecular characterization. J Clin Oncol. 2006; 24:3887–3894.20. Akagi T, Shih LY, Ogawa S, Gerss J, Moore SR, Schreck R, Kawamata N, Liang DC, Sanada M, Nannya Y, et al. Single nucleotide polymorphism genomic arrays analysis of t(8;21) acute myeloid leukemia cells. Haematologica. 2009; 94:1301–1306.21. Akagi T, Shih LY, Kato M, Kawamata N, Yamamoto G, Sanada M, Okamoto R, Miller CW, Liang DC, Ogawa S, et al. Hidden abnormalities and novel classification of t(15;17) acute promyelocytic leukemia (APL) based on genomic alterations. Blood. 2009; 113:1741–1748.22. Mullighan CG, Downing JR. Global genomic characterization of acute lymphoblastic leukemia. Semin Hematol. 2009; 46:3–15.23. Kralovics R, Passamonti F, Buser AS, Teo SS, Tiedt R, Passweg JR, Tichelli A, Cazzola M, Skoda RC. A gain-of-function mutation of JAK2 in myeloproliferative disorders. N Engl J Med. 2005; 352:1779–1790.24. Delhommeau F, Dupont S, Della Valle V, James C, Trannoy S, Massé A, Kosmider O, Le Couedic JP, Robert F, Alberdi A, et al. Mutation in TET2 in myeloid cancers. N Engl J Med. 2009; 360:2289–2301.25. Ernst T, Chase AJ, Score J, Hidalgo-Curtis CE, Bryant C, Jones AV, Waghorn K, Zoi K, Ross FM, Reiter A, et al. Inactivating mutations of the histone methyltransferase gene EZH2 in myeloid disorders. Nat Genet. 2010; 42:722–726.26. Fitzgibbon J, Smith LL, Raghavan M, Smith ML, Debernardi S, Skoulakis S, Lillington D, Lister TA, Young BD. Association between acquired uniparental disomy and homozygous gene mutation in acute myeloid leukemias. Cancer Res. 2005; 65:9152–9154.27. Mohamed AN, Varterasian ML, Dobin SM, McConnell TS, Wolman SR, Rankin C, Willman CL, Head DR, Slovak ML. Trisomy 6 as a primary karyotypic aberration in hematologic disorders. Cancer Genet Cytogenet. 1998; 106:152–155.28. Kelly MJ, Meloni-Ehrig AM, Manley PE, Altura RA. Poor outcome in a pediatric patient with acute myeloid leukemia associated with a variant t(8;21) and trisomy 6. Cancer Genet Cytogenet. 2009; 189:48–52.29. Quentmeier H, Tonelli R, Geffers R, Pession A, Uphoff CC, Drexler HG. Expression of BEX1 in acute myeloid leukemia with MLL rearrangements. Leukemia. 2005; 19:1488–1489.30. Kritharis A, Brody J, Koduru P, Teichberg S, Allen SL. Acute basophilic leukemia associated with loss of gene ETV6 and protean complications. J Clin Oncol. 2011; 29:e623–e626.31. Sherborne AL, Hosking FJ, Prasad RB, Kumar R, Koehler R, Vijayakrishnan J, Papaemmanuil E, Bartram CR, Stanulla M, Schrappe M, et al. Variation in CDKN2A at 9p21.3 influences childhood acute lymphoblastic leukemia risk. Nat Genet. 2010; 42:492–494.32. Kim M, Choi JE, She CJ, Hwang SM, Shin HY, Ahn HS, Yoon SS, Kim BK, Park MH, Lee DS. PAX5 deletion is common and concurrently occurs with CDKN2A deletion in B-lineage acute lymphoblastic leukemia. Blood Cells Mol Dis. 2011; 47:62–66.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Array-based Comparative Genomic Hybridization and Its Application to Cancer Genomes and Human Genetics
- Bioinformatics Interpretation of Exome Sequencing: Blood Cancer
- Diagnostic approach for genetic causes of intellectual disability
- Next generation sequencing and array-based comparative genomic hybridization for molecular diagnosis of pediatric endocrine disorders
- Understanding of epigenetics and dna methylation