J Korean Med Sci.
2013 Nov;28(11):1563-1572. 10.3346/jkms.2013.28.11.1563.
Korean Guideline for Iron Chelation Therapy in Transfusion-Induced Iron Overload
- Affiliations
-
- 1Division of Hematology-Oncology, Department of Medicine, Samsung Medical Center, Sungkyunkwan University School of Medicine, Seoul, Korea.
- 2Department of Hematology, Asan Medical Center, University of Ulsan College of Medicine, Seoul, Korea.
- 3Division of Hematology-Oncology, Seoul National University College of Medicine, Seoul, Korea.
- 4Division of Hematology-Oncology, Chungnam National University School of Medicine, Daejeon, Korea.
- 5Division of Hematology-Oncology, Chonnam National University Medical School, Gwangju, Korea.
- 6Division of Hematology-Oncology, Pusan National University Hospital, Busan, Korea.
- 7Division of Hematology, Department of Internal Medicine, Seoul St. Mary's Hospital, The Catholic University of Korea, Seoul, Korea. jwlee@catholic.ac.kr
- KMID: 1777651
- DOI: http://doi.org/10.3346/jkms.2013.28.11.1563
Abstract
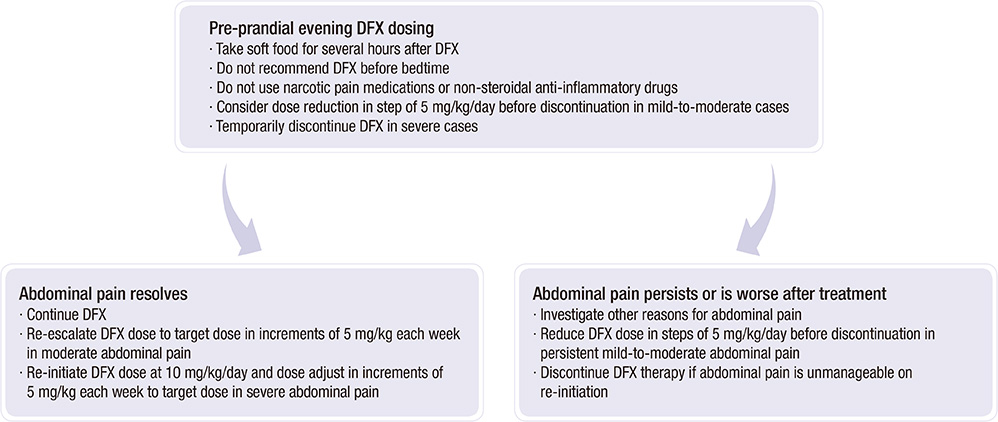
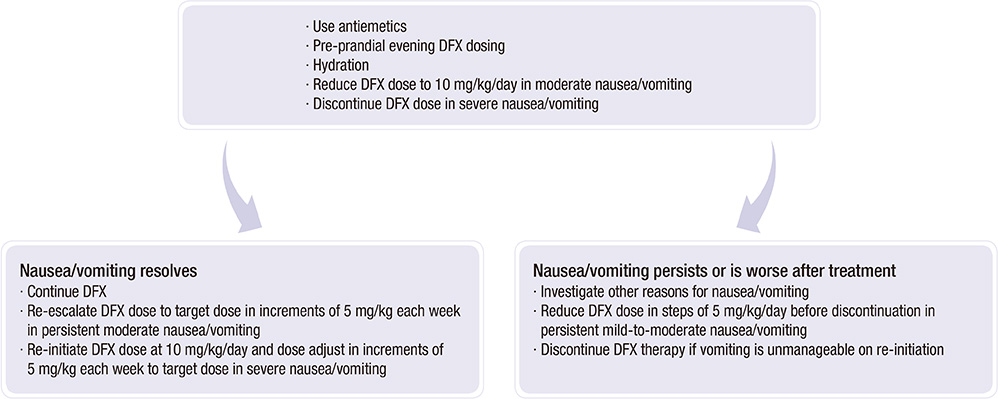
- Many Korean patients with transfusion-induced iron overload experience serious clinical sequelae, including organ damage, and require lifelong chelation therapy. However, due to a lack of compliance and/or unavailability of an appropriate chelator, most patients have not been treated effectively. Deferasirox (DFX), a once-daily oral iron chelator for both adult and pediatric patients with transfusion-induced iron overload, is now available in Korea. The effectiveness of deferasirox in reducing or maintaining body iron has been demonstrated in many studies of patients with a variety of transfusion-induced anemias such as myelodysplastic syndromes, aplastic anemia, and other chronic anemias. The recommended initial daily dose of DFX is 20 mg/kg body weight, taken on an empty stomach at least 30 min before food and serum ferritin levels should be maintained below 1000 ng/mL. To optimize the management of transfusion-induced iron overload, the Korean Society of Hematology Aplastic Anemia Working Party (KSHAAWP) reviewed the general consensus on iron overload and the Korean data on the clinical benefits of iron chelation therapy, and developed a Korean guideline for the treatment of iron overload.
Keyword
MeSH Terms
-
Anemia, Aplastic/therapy
Benzoates/therapeutic use
Blood Transfusion/*adverse effects
Chelation Therapy/*methods
Humans
Iron Chelating Agents/*therapeutic use
Iron Overload/*therapy
Myelodysplastic Syndromes/therapy
Pyridones/therapeutic use
Republic of Korea
Triazoles/therapeutic use
Benzoates
Iron Chelating Agents
Pyridones
Triazoles
Figure
Reference
-
1. Vuille-Lessard E, Boudreault D, Girard F, Ruel M, Chagnon M, Hardy JF. Red blood cell transfusion practice in elective orthopedic surgery: a multicenter cohort study. Transfusion. 2010; 50:2117–2124.2. Napolitano LM, Kurek S, Luchette FA, Corwin HL, Barie PS, Tisherman SA, Hebert PC, Anderson GL, Bard MR, Bromberg W, et al. American College of Critical Care Medicine of the Society of Critical Care Medicine. Eastern Association for the Surgery of Trauma Practice Management Workgroup. Clinical practice guideline: red blood cell transfusion in adult trauma and critical care. Crit Care Med. 2009; 37:3124–3157.3. Practice guidelines for blood transfusion. Osong: Korea Centers for Disease Control and Prevention;2011. p. 18–19.4. Lee JW, Yoon SS, Shen ZX, Ganser A, Hsu HC, Habr D, Domokos G, Roubert B, Porter JB. EPIC study investigators. Iron chelation therapy with deferasirox in patients with aplastic anemia: a subgroup analysis of 116 patients from the EPIC trial. Blood. 2010; 116:2448–2454.5. Cappellini MD, Porter J, El-Beshlawy A, Li CK, Seymour JF, Elalfy M, Gattermann N, Giraudier S, Lee JW, Chan LL, et al. EPIC Study Investigators. Tailoring iron chelation by iron intake and serum ferritin: the prospective EPIC study of deferasirox in 1744 patients with transfusion-dependent anemias. Haematologica. 2010; 95:557–566.6. Porter JB. Practical management of iron overload. Br J Haematol. 2001; 115:239–252.7. Malcovati L. Impact of transfusion dependency and secondary iron overload on the survival of patients with myelodysplastic syndromes. Leuk Res. 2007; 31:S2–S6.8. Seshadri R, Colebatch JH, Gordon P, Ekert H. Long-term administration of desferrioxamine in thalassaemia major. Arch Dis Child. 1974; 49:621–626.9. Hoffbrand AV, Gorman A, Laulicht M, Garidi M, Economidou J, Georgipoulou P, Hussain MA, Flynn DM. Improvement in iron status and liver function in patients with transfusional iron overload with long-term subcutaneous desferrioxamine. Lancet. 1979; 1:947–949.10. De Virgiliis S, Sanna G, Cornacchia G, Argiolu F, Murgia V, Porcu M, Cao A. Serum ferritin, liver iron stores, and liver histology in children with thalassaemia. Arch Dis Child. 1980; 55:43–45.11. Malcovati L, Porta MG, Pascutto C, Invernizzi R, Boni M, Travaglino E, Passamonti F, Arcaini L, Maffioli M, Bernasconi P, et al. Prognostic factors and life expectancy in myelodysplastic syndromes classified according to WHO criteria: a basis for clinical decision making. J Clin Oncol. 2005; 23:7594–7603.12. Porter JB, Davis BA. Monitoring chelation therapy to achieve optimal outcome in the treatment of thalassaemia. Best Pract Res Clin Haematol. 2002; 15:329–368.13. Olivieri NF, Brittenham GM. Iron-chelating therapy and the treatment of thalassemia. Blood. 1997; 89:739–761.14. Brittenham GM, Farrell DE, Harris JW, Feldman ES, Danish EH, Muir WA, Tripp JH, Bellon EM. Magnetic-susceptibility measurement of human iron stores. N Engl J Med. 1982; 307:1671–1675.15. Papakonstantinou O, Alexopoulou E, Economopoulos N, Benekos O, Kattamis A, Kostaridou S, Ladis V, Efstathopoulos E, Gouliamos A, Kelekis NL. Assessment of iron distribution between liver, spleen, pancreas, bone marrow, and myocardium by means of R2 relaxometry with MRI in patients with beta-thalassemia major. J Magn Reson Imaging. 2009; 29:853–859.16. Cappellini MD. Exjade(R) (deferasirox, ICL670) in the treatment of chronic iron overload associated with blood transfusion. Ther Clin Risk Manag. 2007; 3:291–299.17. Pippard MJ, Callender ST, Weatherall DJ. Intensive iron-chelation therapy with desferrioxamine in iron-loading anaemias. Clin Sci Mol Med. 1978; 54:99–106.18. Hershko CM, Link GM, Konijn AM, Cabantchik ZI. Iron chelation therapy. Curr Hematol Rep. 2005; 4:110–116.19. Hider RC, Zhou T. The design of orally active iron chelators. Ann N Y Acad Sci. 2005; 1054:141–154.20. Pennell DJ, Berdoukas V, Karagiorga M, Ladis V, Piga A, Aessopos A, Gotsis ED, Tanner MA, Smith GC, Westwood MA, et al. Randomized controlled trial of deferiprone or deferoxamine in beta-thalassemia major patients with asymptomatic myocardial siderosis. Blood. 2006; 107:3738–3744.21. Taher A, Sheikh-Taha M, Sharara A, Inati A, Koussa S, Ellis G, Dhillon AP, Hoffbrand AV. Safety and effectiveness of 100 mg/kg/day deferiprone in patients with thalassemia major: a two-year study. Acta Haematol. 2005; 114:146–149.22. Cappellini MD, Pattoneri P. Oral iron chelators. Annu Rev Med. 2009; 60:25–38.23. Won SC, Han DK, Seo JJ, Chung NG, Park SK, Park KB, Kook H, Lyu CJ. Efficacy and safety of deferiprone (Ferriprox), an oral iron-chelating agent, in pediatric patients. Korean J Hematol. 2010; 45:58–61.24. Ceci A, Baiardi P, Felisi M, Cappellini MD, Carnelli V, De Sanctis V, Galanello R, Maggio A, Masera G, Piga A, et al. The safety and effectiveness of deferiprone in a large-scale, 3-year study in Italian patients. Br J Haematol. 2002; 118:330–336.25. Nick H, Acklin P, Lattmann R, Buehlmayer P, Hauffe S, Schupp J, Alberti D. Development of tridentate iron chelators: from desferrithiocin to ICL670. Curr Med Chem. 2003; 10:1065–1076.26. Rachmilewitz EA, Weizer-Stern O, Adamsky K, Amariglio N, Rechavi G, Breda L, Rivella S, Cabantchik ZI. Role of iron in inducing oxidative stress in thalassemia: can it be prevented by inhibition of absorption and by antioxidants? Ann N Y Acad Sci. 2005; 1054:118–123.27. Waldmeier F, Bruin GJ, Glaenzel U, Hazell K, Sechaud R, Warrington S, Porter JB. Pharmacokinetics, metabolism, and disposition of deferasirox in beta-thalassemic patients with transfusion-dependent iron overload who are at pharmacokinetic steady state. Drug Metab Dispos. 2010; 38:808–816.28. De Domenico I, Ward DM, Kaplan J. Specific iron chelators determine the route of ferritin degradation. Blood. 2009; 114:4546–4551.29. Neufeld EJ. Update on iron chelators in thalassemia. Hematology Am Soc Hematol Educ Program. 2010; 2010:451–455.30. Brittenham GM. Iron-chelating therapy for transfusional iron overload. N Engl J Med. 2011; 364:146–156.31. Lee JW. Iron chelation therapy in the myelodysplastic syndromes and aplastic anemia: a review of experience in South Korea. Int J Hematol. 2008; 88:16–23.32. Marsh JC, Ball SE, Cavenagh J, Darbyshire P, Dokal I, Gordon-Smith EC, Keidan J, Laurie A, Martin A, Mercieca J, et al. British Committee for Standards in Haematology. Guidelines for the diagnosis and management of aplastic anaemia. Br J Haematol. 2009; 147:43–70.33. Schmid M. Iron chelation therapy in MDS: what have we learnt recently? Blood Rev. 2009; 23:S21–S25.34. Leitch HA. Controversies surrounding iron chelation therapy for MDS. Blood Rev. 2011; 25:17–31.35. Gattermann N. Overview of guidelines on iron chelation therapy in patients with myelodysplastic syndromes and transfusional iron overload. Int J Hematol. 2008; 88:24–29.36. Rose C, Brechignac S, Vassilief D, Pascal L, Stamatoullas A, Guerci A, Larbaa D, Dreyfus F, Beyne-Rauzy O, Chaury MP, Roy L, Cheze S, Morel P, Fenaux P. GFM (Groupe Francophone des Myélodysplasies). Does iron chelation therapy improve survival in regularly transfused lower risk MDS patients? a multicenter study by the GFM (Groupe Francophone des Myélodysplasies). Leuk Res. 2010; 34:864–870.37. Leitch HA. Improving clinical outcome in patients with myelodysplastic syndrome and iron overload using iron chelation therapy. Leuk Res. 2007; 31:S7–S9.38. Davis BA, Porter JB. Long-term outcome of continuous 24-hour deferoxamine infusion via indwelling intravenous catheters in high-risk beta-thalassemia. Blood. 2000; 95:1229–1236.39. Takatoku M, Uchiyama T, Okamoto S, Kanakura Y, Sawada K, Tomonaga M, Nakao S, Nakahata T, Harada M, Murate T, et al. Japanese National Research Group on Idiopathic Bone Marrow Failure Syndromes. Retrospective nationwide survey of Japanese patients with transfusion-dependent MDS and aplastic anemia highlights the negative impact of iron overload on morbidity/mortality. Eur J Haematol. 2007; 78:487–494.40. Gattermann N, Finelli C, Porta MD, Fenaux P, Ganser A, Guerci-Bresler A, Schmid M, Taylor K, Vassilieff D, Habr D, et al. EPIC study investigators. Deferasirox in iron-overloaded patients with transfusion-dependent myelodysplastic syndromes: results from the large 1-year EPIC Study. Leuk Res. 2010; 34:1143–1150.41. Pullarkat V. Objectives of iron chelation therapy in myelodysplastic syndromes: more than meets the eye? Blood. 2009; 114:5251–5255.42. Bullen JJ, Rogers HJ, Spalding PB, Ward CG. Natural resistance, iron and infection: a challenge for clinical medicine. J Med Microbiol. 2006; 55:251–258.43. Naka K, Muraguchi T, Hoshii T, Hirao A. Regulation of reactive oxygen species and genomic stability in hematopoietic stem cells. Antioxid Redox Signal. 2008; 10:1883–1894.44. Pullarkat V, Blanchard S, Tegtmeier B, Dagis A, Patane K, Ito J, Forman SJ. Iron overload adversely affects outcome of allogeneic hematopoietic cell transplantation. Bone Marrow Transplant. 2008; 42:799–805.45. Altès A, Remacha AF, Sureda A, Martino R, Briones J, Canals C, Brunet S, Sierra J, Gimferrer E. Iron overload might increase transplant-related mortality in haematopoietic stem cell transplantation. Bone Marrow Transplant. 2002; 29:987–989.46. Strasser SI, Kowdley KV, Sale GE, McDonald GB. Iron overload in bone marrow transplant recipients. Bone Marrow Transplant. 1998; 22:167–173.47. Kamble RT, Selby GB, Mims M, Kharfan-Dabaja MA, Ozer H, George JN. Iron overload manifesting as apparent exacerbation of hepatic graft-versus-host disease after allogeneic hematopoietic stem cell transplantation. Biol Blood Marrow Transplant. 2006; 12:506–510.48. Armand P, Kim HT, Cutler CS, Ho VT, Koreth J, Alyea EP, Soiffer RJ, Antin JH. Prognostic impact of elevated pretransplantation serum ferritin in patients undergoing myeloablative stem cell transplantation. Blood. 2007; 109:4586–4588.49. Platzbecker U, Bornhäuser M, Germing U, Stumpf J, Scott BL, Kröger N, Schwerdtfeger R, Böhm A, Kobbe G, Theuser C, et al. Red blood cell transfusion dependence and outcome after allogeneic peripheral blood stem cell transplantation in patients with de novo myelodysplastic syndrome (MDS). Biol Blood Marrow Transplant. 2008; 14:1217–1225.50. Lee JW, Kang HJ, Kim EK, Kim H, Shin HY, Ahn HS. Effect of iron overload and iron-chelating therapy on allogeneic hematopoietic SCT in children. Bone Marrow Transplant. 2009; 44:793–797.51. Alessandrino EP, Della Porta MG, Bacigalupo A, Malcovati L, Angelucci E, Van Lint MT, Falda M, Onida F, Bernardi M, Guidi S, et al. Prognostic impact of pre-transplantation transfusion history and secondary iron overload in patients with myelodysplastic syndrome undergoing allogeneic stem cell transplantation: a GITMO Study. Haematologica. 2010; 95:476–484.52. Lee SE, Yahng SA, Cho BS, Eom KS, Kim YJ, Lee S, Min CK, Kim HJ, Cho SG, Kim DW, et al. Improvement in hematopoiesis after iron chelation therapy with deferasirox in patients with aplastic anemia. Acta Haematol. 2013; 129:72–77.53. Porter J, Galanello R, Saglio G, Neufeld EJ, Vichinsky E, Cappellini MD, Olivieri N, Piga A, Cunningham MJ, Soulières D, et al. Relative response of patients with myelodysplastic syndromes and other transfusion-dependent anaemias to deferasirox (ICL670): a 1-yr prospective study. Eur J Haematol. 2008; 80:168–176.54. Vichinsky E, Pakbaz Z, Onyekwere O, Porter J, Swerdlow P, Coates T, Lane P, Files B, Mueller BU, Coïc L, et al. Patient-reported outcomes of deferasirox (Exjade, ICL670) versus deferoxamine in sickle cell disease patients with transfusional hemosiderosis: substudy of a randomized open-label phase II trial. Acta Haematol. 2008; 119:133–141.55. Cappellini MD, Cohen A, Piga A, Bejaoui M, Perrotta S, Agaoglu L, Aydinok Y, Kattamis A, Kilinc Y, Porter J, et al. A phase 3 study of deferasirox (ICL670), a once-daily oral iron chelator, in patients with beta-thalassemia. Blood. 2006; 107:3455–3462.56. Galanello R, Piga A, Forni GL, Bertrand Y, Foschini ML, Bordone E, Leoni G, Lavagetto A, Zappu A, Longo F, et al. Phase II clinical evaluation of deferasirox, a once-daily oral chelating agent, in pediatric patients with beta-thalassemia major. Haematologica. 2006; 91:1343–1351.57. Nisbet-Brown E, Olivieri NF, Giardina PJ, Grady RW, Neufeld EJ, Séchaud R, Krebs-Brown AJ, Anderson JR, Alberti D, Sizer KC, et al. Effectiveness and safety of ICL670 in iron-loaded patients with thalassaemia: a randomised, double-blind, placebo-controlled, dose-escalation trial. Lancet. 2003; 361:1597–1602.58. Vichinsky E. Clinical application of deferasirox: practical patient management. Am J Hematol. 2008; 83:398–402.59. Taher A, Cappellini MD, Vichinsky E, Galanello R, Piga A, Lawniczek T, Clark J, Habr D, Porter JB. Efficacy and safety of deferasirox doses of >30 mg/kg per d in patients with transfusion-dependent anaemia and iron overload. Br J Haematol. 2009; 147:752–759.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Diagnosis and treatment of transfusion-related iron overload
- Role of redox iron towards an increase in mortality among patients: a systemic review and meta-analysis
- Efficacy and safety of combined oral iron chelation therapy with deferasirox and deferiprone in a patient with beta-thalassemia major and persistent iron overload
- Treatment and Management of Late Complications in Hereditary Hemolytic Anemia
- Molecular perspective of iron uptake, related diseases, and treatments