Antiallodynic Effect of Thalidomide and Morphine on Rat Spinal Nerve Ligation-induced Neuropathic Pain
- Affiliations
-
- 1Department of Anesthesiology and Pain Medicine, Chonnam National University, Medical School, Gwangju, Korea. leehg@chonnam.ac.kr
- KMID: 1767873
- DOI: http://doi.org/10.3344/kjp.2010.23.3.172
Abstract
- BACKGROUND
Tumor necrosis factor-alpha and other proinflammatory cytokines are becoming well recognized as key mediators in the pathogenesis of many types of neuropathic pain. Thalidomide has profound immunomodulatory actions in addition to their originally intended pharmacological actions. There has been debate on the analgesic efficacy of opioids in neuropathic pain. The aim of this study was to investigate the effect of thalidomide and morphine on a spinal nerve ligation model in rats.
METHODS
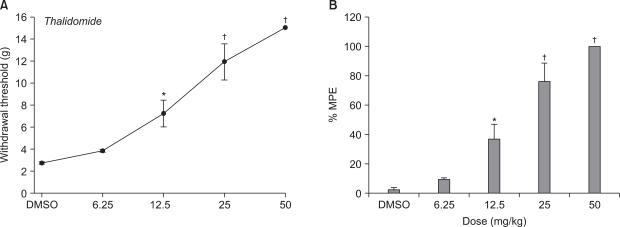
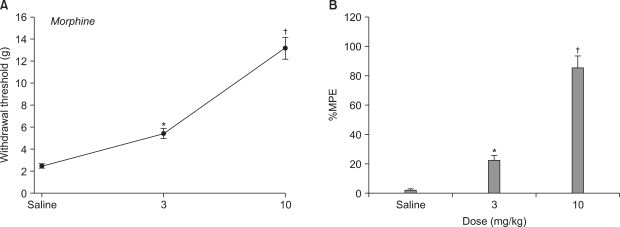
Male Sprague-Dawley rats weighing 100-120 g were used. Lumbar (L) 5 and 6 spinal nerve ligations were performed to induce neuropathic pain. For assessment of mechanical allodynia, mechanical stimulus using von Frey filament was applied to the paw to measure withdrawal threshold. The effects of intraperitoneal thalidomide (6.25, 12.5, 25 and 50 mg/kg, respectively) and morphine (3 and 10 mg/kg, respectively) were examined on a withdrawal threshold evoked by spinal nerve ligation.
RESULTS
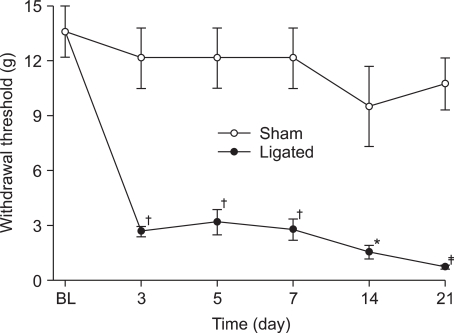
After L5 and 6 spinal nerve ligation, paw withdrawal thresholds on the ipsilateral side were significantly decreased compared with pre-operative baseline and with those in the sham-operated group. Intraperitoneal thalidomide and morphine significantly increased the paw withdrawal threshold compared to controls and produced dose-responsiveness.
CONCLUSIONS
Systemic thalidomide and morphine have antiallodynic effect on neuropathic pain induced by spinal nerve ligation in rat. These results suggest that morphine and thalidomide may be alternative therapeutic approaches for neuropathic pain.
Keyword
MeSH Terms
Figure
Cited by 3 articles
-
Synergistic anti-allodynic effect between intraperitoneal thalidomide and morphine on rat spinal nerve ligation-induced neuropathic pain
Hyung Gon Lee, Woong Mo Kim, Myung Ha Yoon, A Reum Park, Jeong-Il Choi
Korean J Anesthesiol. 2013;65(4):331-336. doi: 10.4097/kjae.2013.65.4.331.Tumor Necrosis Factor-alpha and Apoptosis Following Spinal Nerve Ligation Injury in Rats
Sung Hoon Kim, Jae Sik Nam, Dae Kee Choi, Won Wook Koh, Jeong Hun Suh, Jun Gol Song, Jin Woo Shin, Jeong Gil Leem
Korean J Pain. 2011;24(4):185-190. doi: 10.3344/kjp.2011.24.4.185.Effect of Ethyl Pyruvate on Paclitaxel-Induced Neuropathic Pain in Rats
Seong Soo Choi, Won Uk Koh, Jae Sik Nam, Jin Woo Shin, Jeong Gill Leem, Jeong Hun Suh
Korean J Pain. 2013;26(2):135-141. doi: 10.3344/kjp.2013.26.2.135.
Reference
-
2. Barkin RL, Barkin SJ, Barkin DS. Perception, assessment, treatment, and management of pain in the elderly. Clin Geriatr Med. 2005; 21:465–490. PMID: 15911202.
Article3. Wallace JM. Update on pharmacotherapy guidelines for treatment of neuropathic pain. Curr Pain Headache Rep. 2007; 11:208–214. PMID: 17504648.
Article4. Watkins LR, Milligan ED, Maier SF. Glial activation: a driving force for pathological pain. Trends Neurosci. 2001; 24:450–455. PMID: 11476884.
Article5. White FA, Jung H, Miller RJ. Chemokines and the pathophysiology of neuropathic pain. Proc Natl Acad Sci USA. 2007; 104:20151–20158. PMID: 18083844.
Article6. Bennett GJ. A neuroimmune interaction in painful peripheral neuropathy. Clin J Pain. 2000; 16(3 Suppl):S139–S143. PMID: 11014458.
Article7. Laffitte E, Revuz J. Thalidomide: an old drug with new clinical applications. Expert Opin Drug Saf. 2004; 3:47–56. PMID: 14680461.
Article8. Teo SK, Stirling DI, Zeldis JB. Thalidomide as a novel therapeutic agent: new uses for an old product. Drug Discov Today. 2005; 10:107–114. PMID: 15718159.
Article9. Laffitte E. The revival of thalidomide: an old drug with new indications. Rev Prat. 2006; 56:1977–1983. PMID: 17274497.10. Ribeiro RA, Vale ML, Ferreira SH, Cunha FQ. Analgesic effect of thalidomide on inflammatory pain. Eur J Pharmacol. 2000; 391:97–103. PMID: 10720640.
Article11. Rocha AC, Fernandes ES, Quintão NL, Campos MM, Calixto JB. Relevance of tumour necrosis factor-alpha for the inflammatory and nociceptive responses evoked by carrageenan in the mouse paw. Br J Pharmacol. 2006; 148:688–695. PMID: 16702985.
Article12. George A, Marziniak M, Schäfers M, Toyka KV, Sommer C. Thalidomide treatment in chronic constrictive neuropathy decreases endoneurial tumor necrosis factor-alpha, increases interleukin-10 and has long-term effects on spinal cord dorsal horn met-enkephalin. Pain. 2000; 88:267–275. PMID: 11068114.
Article13. Xu JT, Xin WJ, Zang Y, Wu CY, Liu XG. The role of tumor necrosis factor-alpha in the neuropathic pain induced by Lumbar 5 ventral root transection in rat. Pain. 2006; 123:306–321. PMID: 16675114.
Article14. Rajkumar SV, Fonseca R, Witzig TE. Complete resolution of reflex sympathetic dystrophy with thalidomide treatment. Arch Intern Med. 2001; 161:2502–2503. PMID: 11700165.
Article15. Ching DW, McClintock A, Beswick F. Successful treatment with low-dose thalidomide in a patient with both Behçet's disease and complex regional pain syndrome type I: case report. J Clin Rheumatol. 2003; 9:96–98. PMID: 17041438.
Article16. Arnér S, Meyerson BA. Lack of analgesic effect of opioids on neuropathic and idiopathic forms of pain. Pain. 1988; 33:11–23. PMID: 2454440.
Article17. Dellemijn P. Are opioids effective in relieving neuropathic pain? Pain. 1999; 80:453–462. PMID: 10342407.
Article18. Gimbel JS, Richards P, Portenoy RK. Controlled-release oxycodone for pain in diabetic neuropathy: a randomized controlled trial. Neurology. 2003; 60:927–934. PMID: 12654955.
Article19. Raja SN, Haythornthwaite JA, Pappagallo M, Clark MR, Travison TG, Sabeen S, et al. Opioids versus antidepressants in postherpetic neuralgia: a randomized, placebo-controlled trial. Neurology. 2002; 59:1015–1021. PMID: 12370455.
Article20. Zimmermann M. Ethical guidelines for investigations on experimental pain in conscious animals. Pain. 1983; 16:109–110. PMID: 6877845.
Article21. Kim SH, Chung JM. An experimental model for peripheral neuropathy produced by segmental spinal nerve ligation in the rat. Pain. 1992; 50:355–363. PMID: 1333581.
Article22. Chaplan SR, Bach FW, Pogrel JW, Chung JM, Yaksh TL. Quantitative assessment of tactile allodynia in the rat paw. J Neurosci Methods. 1994; 53:55–63. PMID: 7990513.
Article23. DeLeo JA, Yezierski RP. The role of neuroinflammation and neuroimmune activation in persistent pain. Pain. 2001; 90:1–6. PMID: 11166964.
Article24. Myers RR, Campana WM, Shubayev VI. The role of neuroinflammation in neuropathic pain: mechanisms and therapeutic targets. Drug Discov Today. 2006; 11:8–20. PMID: 16478686.
Article25. Sampaio EP, Sarno EN, Galilly R, Cohn ZA, Kaplan G. Thalidomide selectively inhibits tumor necrosis factor alpha production by stimulated human monocytes. J Exp Med. 1991; 173:699–703. PMID: 1997652.
Article26. Moreira AL, Sampaio EP, Zmuidzinas A, Frindt P, Smith KA, Kaplan G. Thalidomide exerts its inhibitory action on tumor necrosis factor alpha by enhancing mRNA degradation. J Exp Med. 1993; 177:1675–1680. PMID: 8496685.
Article27. Turk BE, Jiang H, Liu JO. Binding of thalidomide to alpha1-acid glycoprotein may be involved in its inhibition of tumor necrosis factor alpha production. Proc Natl Acad Sci USA. 1996; 93:7552–7556. PMID: 8755512.
Article28. Vale ML, Cunha FQ, Brito GA, Benevides VM, Ferreira SH, Poole S, et al. Anti-nociceptive effect of thalidomide on zymosan-induced experimental articular incapacitation. Eur J Pharmacol. 2006; 536:309–317. PMID: 16597438.
Article29. Bian D, Nichols ML, Ossipov MH, Lai J, Porreca F. Characterization of the antiallodynic efficacy of morphine in a model of neuropathic pain in rats. Neuroreport. 1995; 6:1981–1984. PMID: 8580422.
Article30. Lee YW, Chaplan SR, Yaksh TL. Systemic and supraspinal, but not spinal, opiates suppress allodynia in a rat neuropathic pain model. Neurosci Lett. 1995; 199:111–114. PMID: 8584236.
Article31. Ossipov MH, Lopez Y, Nichols ML, Bian D, Porreca F. Inhibition by spinal morphine of the tail-flick response is attenuated in rats with nerve ligation injury. Neurosci Lett. 1995; 199:83–86. PMID: 8584249.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Synergistic anti-allodynic effect between intraperitoneal thalidomide and morphine on rat spinal nerve ligation-induced neuropathic pain
- The Mechanical Antiallodynic Effect of intrathecal Morphine and R-Phenylisopropyl-Adenosine in Rats with Spinal Nerve Ligation
- The Mechanism of Antiallodynic Effect of Intrathecal Morphine in Neuropathic Pain Induced by Spinal Nerve Ligation: The Effect of Methysergide and Theophylline
- The Mechanism of Action of Intrathecal Morphine in Neuropathic Pain Induced by Nerve Ligation: The Effect of Naloxone
- The Mechanism of Antiallodynic Effect of Intrathecal Morphine in Neuropathic Pain Induced by Spinal Nerve Ligation