J Vet Sci.
2008 Sep;9(3):309-315. 10.4142/jvs.2008.9.3.309.
Cytokine response in Balb/c mice infected with Francisella tularensis LVS and the Pohang isolate
- Affiliations
-
- 1Divison of Zoonoses, Center for Immunology and Pathology, Korea National Institute of Health, Seoul 122-701, Korea. kyuhwang@nih.go.kr
- 2Divison of Epidemiology and Health Index, Center for Genome Sciences, Korea National Institute of Health, Seoul 122-701, Korea.
- KMID: 1718585
- DOI: http://doi.org/10.4142/jvs.2008.9.3.309
Abstract
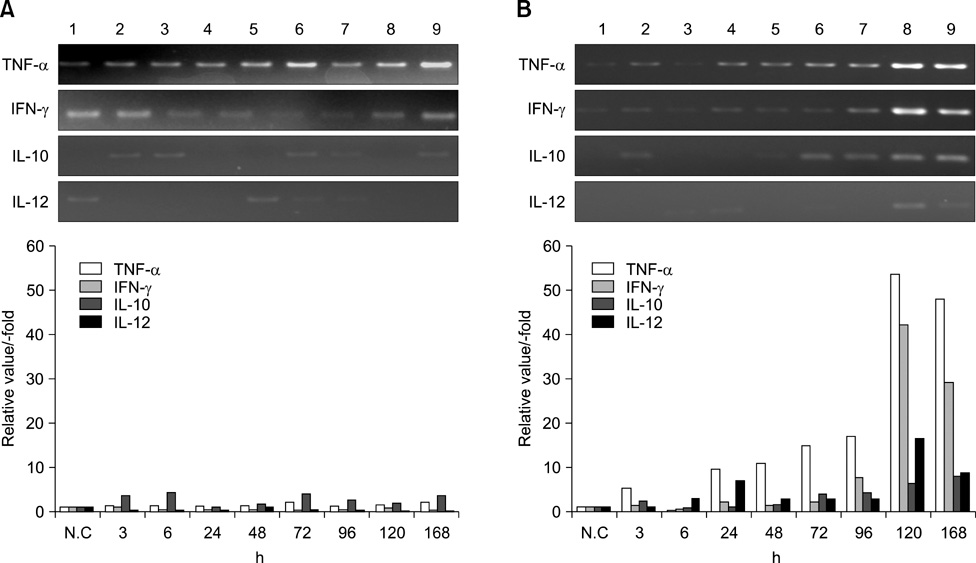
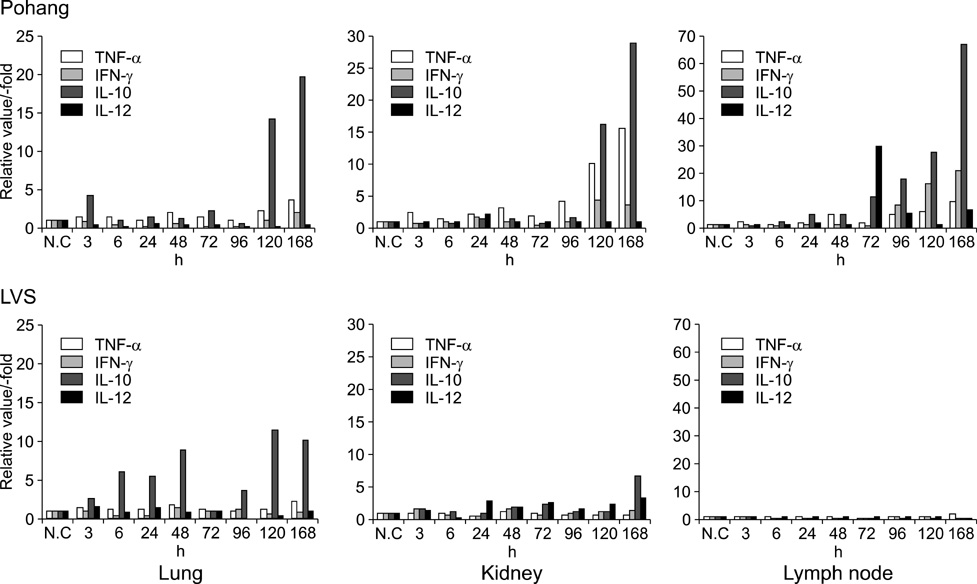
- We investigated the immune response induced by the Francisella (F.) tularensis live vaccine strain (LVS) and the Pohang isolate. After the Balb/c mice were infected intradermally (i.d) with 2 x 10(4) cfu of F. tularensis LVS and Pohang, respectively, their blood and organs were collected at different times; 0, 3, 6, 24, 72, 96, 120 and 168 h after infection. Using these samples, RT-PCR and ELISA analysis were carried out for the comparative study of the cytokines, including TNF-alpha, INF-gamma, IL-2, IL-4, IL-10 and IL-12. In the Pohang-infected mice at 120 h, the liver showed a 53 times higher level of TNF-alpha and a 42 times higher level of IFN-gamma than the respective levels at the early time points after infection. The levels of TNF-alpha and IFN-gamma induced by LVS were 5 times lower than those induced by the Pohang isolate. Also, the organs from the Pohang-infected mice showed higher levels of TNF-alpha, IFN-gamma, IL-10 and IL-12 than the levels in the LVS-infected mice. The blood from the Pohang-infected mice at 120 h revealed about a 40 times increased level of IFN-gamma, and IL-10 was also increased by 4 times at 96 h compared to an early infection time point, while IL-4 was not induced during the whole infection period. These results suggest that F. tularensis may induce a Th1-mediated immune response to in vivo infection and the Pohang isolate has a higher capacity than the LVS to induce an acute immune response in Blab/c mice.
Keyword
MeSH Terms
-
Animals
*Bacterial Vaccines
Cytokines/*biosynthesis
Francisella tularensis/immunology/isolation & purification/*pathogenicity
Humans
Interferon-gamma/genetics/metabolism
Interleukins/genetics/metabolism
Korea
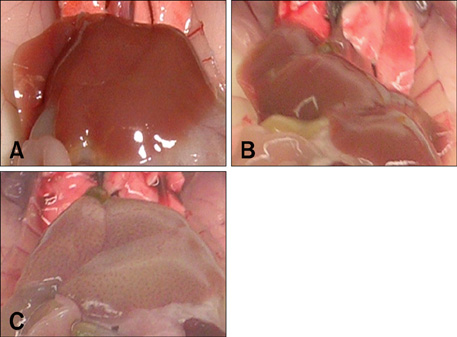
Liver/microbiology/pathology
Mice
Mice, Inbred BALB C
Polymerase Chain Reaction
Tularemia/*diagnosis/*immunology
Tumor Necrosis Factor-alpha/genetics/metabolism
Figure
Reference
-
1. Bachiller Luque P, Pérez Castrillon JL, Martín Luquero M, Mena Martin FJ, de la Lama López-Areal J, Pérez Pascual P, Mazón MA, Herreros Guilarte V. Preliminary report of an epidemic tularemia outbreak in Valladolid. Rev Clin Esp. 1998. 198:789–793.2. Berdal BP, Mehl R, Meidell NK, Lorentzen-Styr AM, Scheel O. Field investigations of tularemia in Norway. FEMS Immunol Med Microbiol. 1996. 13:191–195.
Article3. Chu MC, Weyant RS. Murray PR, Baron EJ, Jorgensen JH, Pfaller MA, Yolken RH, editors. Francisella and brucella. Manual of Clinical Microbiology. 2003. 8th ed. Washington DC: American Society for Microbiology;789–808.4. Fortier AH, Polsinelli T, Green SJ, Nacy CA. Activation of macrophages for destruction of Francisella tularensis: identification of cytokines, effector cells, and effector molecules. Infect Immun. 1992. 60:817–825.
Article5. Fortier AH, Slayter MV, Ziemba R, Meltzer MS, Nacy CA. Live vaccine strain of Francisella tularensis: infection and immunity in mice. Infect Immun. 1991. 59:2922–2928.
Article6. Gavrilin MA, Bouakl IJ, Knatz NL, Duncan MD, Hall MW, Gunn JS, Wewers MD. Internalization and phagosome escape required for Francisella to induce human monocyte IL-1beta processing and release. Proc Natl Acad Sci USA. 2006. 103:141–146.
Article7. Golovliov I, Sandström G, Ericsson M, Sjöstedt A, Tarnvik A. Cytokine expression in the liver during the early phase of murine tularemia. Infect Immun. 1995. 63:534–538.
Article8. Kärttunen R, Surcel HM, Andersson G, Ekre HP, Herva E. Francisella tularensis-induced in vitro gamma interferon, tumor necrosis factor alpha, and interleukin 2 responses appear within 2 weeks of tularemia vaccination in human beings. J Clin Microbiol. 1991. 29:753–756.
Article9. Katz J, Zhang P, Martin M, Vogel SN, Michalek SM. Toll-like receptor 2 is required for inflammatory responses to Francisella tularensis LVS. Infect Immun. 2006. 74:2809–2816.
Article10. Kim MY, Ha GY, Ahn WS, Lim HS, Kim DH, Chong YS. A case of tularemia caused by Francisella Tularensis. Korean J Clin Pathol. 1998. 18:90–95.11. McCoy GW, Chapin CW. Further observations on a plague-like disease of rodents with a preliminary note on the causative agent, Bacterium tularense. J Infect Dis. 1912. 10:61–72.
Article12. Ohara Y, Sato T, Fujita H, Ueno T, Homma M. Clinical manifestations of tularemia in Japan--analysis of 1,355 cases observed between 1924 and 1987. Infection. 1991. 19:14–17.
Article13. Ohara Y, Sato T, Homma M. Arthropod-borne tularemia in Japan: clinical analysis of 1,374 cases observed between 1924 and 1996. J Med Entomol. 1998. 35:471–473.
Article14. Olsufjev NG, Meshcheryakova IS. Infraspecific taxonomy of tularemia agent Francisella tularensis McCoy et Chapin. J Hyg Epidemiol Microbiol Immunol. 1982. 26:291–299.15. Reintjes R, Dedushaj I, Gjini A, Jorgensen TR, Cotter B, Lieftucht A, D'Ancona F, Dennis DT, Kosoy MA, Mulliqi-Osmani G, Grunow R, Kalaveshi A, Gashi L, Humolli I. Tularemia outbreak investigation in Kosovo: case control and environmental studies. Emerg Infect Dis. 2002. 8:69–73.
Article16. Rogers HW, Sheehan KC, Brunt LM, Dower SK, Unanue ER, Schreiber RD. Interleukin 1 participates in the development of anti-Listeria responses in normal and SCID mice. Proc Natl Acad Sci USA. 1992. 89:1011–1015.
Article17. Stenmark S, Sunnemark D, Bucht A, Sjöstedt A. Rapid local expression of interleukin-12, tumor necrosis factor alpha, and gamma interferon after cutaneous Francisella tularensis infection in tularemia-immune mice. Infect Immun. 1999. 67:1789–1797.
Article18. Tärnvik A, Ericsson M, Golovliov I, Sandström G, Sjöstedt A. Orchestration of the protective immune response to intracellular bacteria: Francisella tularensis as a model organism. FEMS Immunol Med Microbiol. 1996. 13:221–225.
Article19. Wickstrum JR, Hong KJ, Bokhari S, Reed N, McWilliams N, Horvat RT, Parmely MJ. Coactivating signals for the hepatic lymphocyte gamma interferon response to Francisella tularensis. Infect Immun. 2007. 75:1335–1342.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Current status of vaccine development for tularemia preparedness
- Development of dual reporter imaging system for Francisella tularensis to monitor the spatio-temporal pathogenesis and vaccine efficacy
- A Case of Tularemia Caused by Francisella Tularensis
- The first pediatric case of tularemia in Korea: manifested with pneumonia and possible infective endocarditis
- A Case of Ulceroglandular Tularemia