J Korean Med Sci.
2010 May;25(5):684-690. 10.3346/jkms.2010.25.5.684.
Individualized Tumor Response Testing for Prediction of Response to Paclitaxel and Cisplatin Chemotherapy in Patients with Advanced Gastric Cancer
- Affiliations
-
- 1Department of Internal Medicine, Seoul National University Bundang Hospital, Seongnam, Korea.
- 2Department of Internal Medicine, Korea University Anam Hospital, Seoul, Korea.
- 3Department of Internal Medicine, Yeungnam University Medical Center, Daegu, Korea.
- 4Department of Internal Medicine, Seoul National University Boramae Hospital, Seoul, Korea.
- 5Department of Internal Medicine, Seoul Paik Hospital, Seoul, Korea.
- 6Department of Internal Medicine, Korea Cancer Center Hospital, Seoul, Korea.
- 7Department of Internal Medicine, Seoul National University Hospital, Seoul, Korea. bangyj@snu.ac.kr
- 8Isu Abxis Co., Ltd, Seoul, Korea.
- KMID: 1713950
- DOI: http://doi.org/10.3346/jkms.2010.25.5.684
Abstract
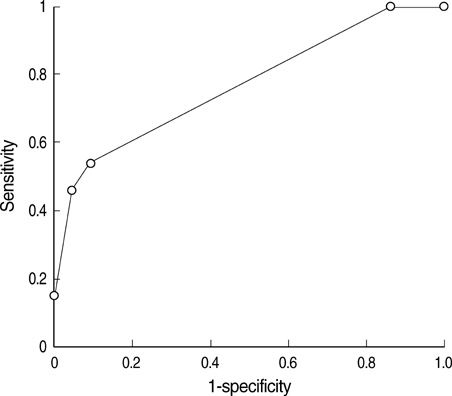
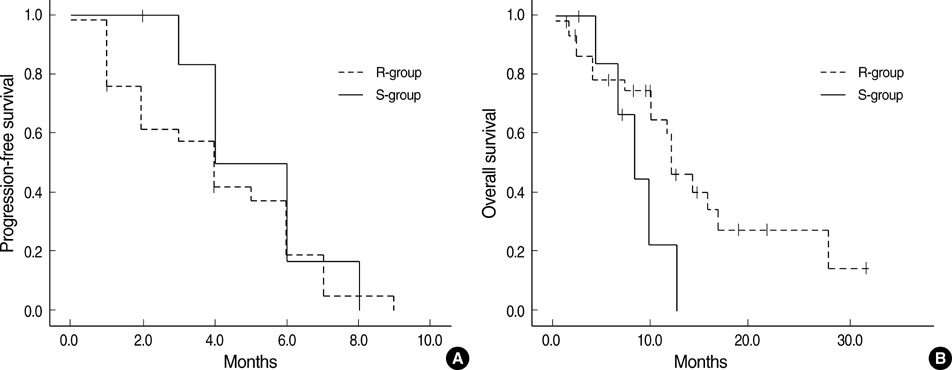
- The purpose of our study was to determine the most accurate analytic method to define in vitro chemosensitivity, using clinical response as reference standard in prospective clinical trial, and to assess accuracy of adenosine triphosphate-based chemotherapy response assay (ATP-CRA). Forty-eight patients with chemo-naive, histologically confirmed, locally advanced or metastatic gastric cancer were enrolled for the study and were treated with combination chemotherapy of paclitaxel 175 mg/m2 and cisplatin 75 mg/m2 for maximum of six cycles after obtaining specimen for ATP-CRA. We performed the receiver operator characteristic curve analysis using patient responses by WHO criteria and ATP-CRA results to define the method with the highest accuracy. Median progression free survival was 4.2 months (95% confidence interval [CI]: 3.4-5.0) and median overall survival was 11.8 months (95% CI: 9.7-13.8) for all enrolled patients. Chemosensitivity index method yielded highest accuracy of 77.8% by ROC curve analysis, and the specificity, sensitivity, positive and negative predictive values were 95.7%, 46.2%, 85.7%, and 75.9%. In vitro chemosensitive group showed higher response rate (85.7% vs. 24.1%) (P=0.005) compared to chemoresistant group. ATP-CRA could predict clinical response to paclitaxel and cisplatin chemotherapy with high accuracy in advanced gastric cancer patients. Our study supports the use of ATP-CRA in further validation studies.
Keyword
MeSH Terms
-
Adenosine Triphosphate/*analysis
Adult
Aged
Antineoplastic Agents, Phytogenic/administration & dosage
Antineoplastic Combined Chemotherapy Protocols/*administration & dosage
Cisplatin/administration & dosage
Drug Screening Assays, Antitumor/*methods
Female
Humans
Korea
Male
Middle Aged
Outcome Assessment (Health Care)/methods
Paclitaxel/administration & dosage
Reproducibility of Results
Sensitivity and Specificity
Stomach Neoplasms/*diagnosis/*drug therapy/metabolism
Treatment Outcome
Antineoplastic Agents, Phytogenic
Cisplatin
Paclitaxel
Adenosine Triphosphate
Figure
Reference
-
1. Schrag D, Garewal HS, Burstein HJ, Samson DJ, Von Hoff DD, Somerfield MR. American society of clinical oncology technology assessment: chemotherapy sensitivity and resistance assays. J Clin Oncol. 2004. 22:3631–3638.
Article2. Von Hoff DD, Clark GM, Stogdill BJ, Sarosdy MF, O'Brien MT, Casper JT, Mattox DE, Page CP, Cruz AB, Sandbach JF. Prospective clinical trial of a human tumor cloning system. Cancer Res. 1983. 43:1926–1931.3. Carmichael J, DeGraff WG, Gazdar AF, Minna JD, Mitchell JB. Evaluation of a tetrazolium-based semiautomated colorimetric assay: assessment of chemosensitivity testing. Cancer Res. 1987. 47:936–942.4. Gazdar AF, Steinberg SM, Russell EK, Linnoila RI, Oie HK, Ghosh BC, Cotelingam JD, Johnson BE, Minna JD, Ihde DC. Correlation of in vitro drug-sensitivity testing results with response to chemotherapy and survival in extensive-stage small cell lung cancer: a prospective clinical trial. J Natl Cancer Inst. 1990. 82:117–124.
Article5. Samson DJ, Seidenfeld J, Ziegler K, Aronson N. Chemotherapy sensitivity and resistance assays: a systematic review. J Clin Oncol. 2004. 22:3618–3630.
Article6. Andreotti PE, Cree IA, Kurbacher CM, Hartmann DM, Linder D, Harel G, Gleiberman I, Caruso PA, Ricks SH, Untch M. Chemosensitivity testing of human tumors using a microplate adenosine triphosphate luminescence assay: clinical correlation for cisplatin resistance of ovarian carcinoma. Cancer Res. 1995. 55:5276–5282.7. Cree IA, Neale MH, Myatt NE, de Takats PG, Hall P, Grant J, Kurbacher CM, Reinhold U, Neuber K, MacKie RM, Chana J, Weaver PC, Khoury GG, Sartori C, Andreotti PE. Heterogeneity of chemosensitivity of metastatic cutaneous melanoma. Anticancer Drugs. 1999. 10:437–444.
Article8. Konecny G, Crohns C, Pegram M, Felber M, Lude S, Kurbacher C, Cree IA, Hepp H, Untch M. Correlation of drug response with the ATP tumorchemosensitivity assay in primary figo stage III ovarian cancer. Gynecol Oncol. 2000. 77:258–263.
Article9. Kurbacher CM, Cree IA, Brenne U, Bruckner HW, Kurbacher JA, Mallmann P, Andreotti PE, Krebs D. Heterogeneity of in vitro chemosensitivity in perioperative breast cancer cells to mitoxantrone versus doxorubicin evaluated by a microplate ATP bioluminescence assay. Breast Cancer Res Treat. 1996. 41:161–170.10. Ugurel S, Schadendorf D, Pfohler C, Neuber K, Thoelke A, Ulrich J, Hauschild A, Spieth K, Kaatz M, Rittgen W, Delorme S, Tilgen W, Reinhold U. In vitro drug sensitivity predicts response and survival after individualized sensitivity-directed chemotherapy in metastatic melanoma: a multicenter phase ii trial of the dermatologic cooperative oncology group. Clin Cancer Res. 2006. 12:5454–5463.
Article11. Kang SM, Park MS, Chang J, Kim SK, Kim H, Shin D, Chung KY, Kim DJ, Sohn JH, Choi SH, Kim J, Yoon EJ, Kim JH. A feasibility study of adenosine triphosphate-based chemotherapy response assay (ATP-CRA) as a chemosensitivity test for lung cancer. Cancer Res Treat. 2005. 37:223–227.
Article12. Moon YW, Choi SH, Kim YT, Sohn JH, Chang J, Kim SK, Park MS, Chung KY, Lee HJ, Kim JH. Adenosine triphosphate-based chemotherapy response assay (ATP-CRA)-guided platinum-based 2-drug chemotherapy for unresectable nonsmall-cell lung cancer. Cancer. 2007. 109:1829–1835.
Article13. Han SS, Choi SH, Lee YK, Kim JW, Park NH, Song YS, Lee HP, Kang SB. Predictive value of individualized tumor response testing by atp-based chemotherapy response assay in ovarian cancer. Cancer Invest. 2008. 26:426–430.
Article14. Kim HA, Yom CK, Moon BI, Choe KJ, Sung SH, Han WS, Choi HY, Kim HK, Park HK, Choi SH, Yoon EJ, Oh SY. The use of an in vitro adenosine triphosphate-based chemotherapy response assay to predict chemotherapeutic response in breast cancer. Breast. 2008. 17:19–26.
Article15. Bepler G, O'Briant K. In vitro chemosensitivity testing of human non-small cell lung cancer cell lines. Anticancer Res. 1998. 18:3181–3185.16. Boddy AV, Griffin MJ, Sludden J, Thomas HD, Fishwick K, Wright JG, Plumner ER, Highley M, Calvert AH. Pharmacological study of paclitaxel duration of infusion combined with GFR-based carboplatin in the treatment of ovarian cancer. Cancer Chemother Pharmacol. 2001. 48:15–21.17. Miller AB, Hoogstraten B, Staquet M, Winkler A. Reporting results of cancer treatment. Cancer. 1981. 47:207–214.
Article18. Cree IA, Kurbacher CM, Untch M, Sutherland LA, Hunter EM, Subedi AM, James EA, Dewar JA, Preece PE, Andreotti PE, Bruckner HW. Correlation of the clinical response to chemotherapy in breast cancer with ex vivo chemosensitivity. Anticancer Drugs. 1996. 7:630–635.
Article19. Kurbacher CM, Cree IA, Bruckner HW, Brenne U, Kurbacher JA, Muller K, Ackermann T, Gilster TJ, Wilhelm LM, Engel H, Mallmann PK, Andreotti PE. Use of an ex vivo ATP luminescence assay to direct chemotherapy for recurrent ovarian cancer. Anticancer Drugs. 1998. 9:51–57.
Article20. Sevin BU, Peng PZ, Perras JP, Ganjei P, Penalver M, Averette HE. Application of an ATP-bioluminescence assay in human tumor chemosensitivity testing. Gynecol Oncol. 1988. 31:191–204.
Article21. Bosanquet AG, Bell PB. Enhanced ex vivo drug sensitivity testing of chronic lymphocytic leukaemia using refined disc assay methodology. Leukemia research. 1996. 20:143–153.
Article22. O'Meara AT, Sevin BU. Predictive value of the ATP chemosensitivity assay in epithelial ovarian cancer. Gynecol Oncol. 2001. 83:334–342.23. Loong TW. Understanding sensitivity and specificity with the right side of the brain. BMJ. 2003. 327:716–719.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- A Phase II Study of Paclitaxel and Cisplatin as Salvage Therapy for Patients with Advanced or Metastatic Gastric Cancer
- A Phase II Trial of Paclitaxel, 5-fluorouracil (5-FU) and Cisplatin in Patients with Metastatic or Recurrent Gastric Cancer
- Efficacy of Combination Chemotherapy with Paclitaxel and Cisplatin in Patients with Advanced Non-Small Cell Lung Cancer
- A case of advanced ovarian cancer which was treated with topotecan after taxol-cisplatin treatment failed
- Combination chemotherapy with docetaxel and cisplatin as first-line treatment in advanced gastric cancer: is it a new effective chemotherapy?