Cancer Res Treat.
2007 Mar;39(1):6-9.
A Phase II Study of Paclitaxel and Cisplatin as Salvage Therapy for Patients with Advanced or Metastatic Gastric Cancer
- Affiliations
-
- 1Department of Internal Medicine, Dong-A University College of Medicine, Busan, Korea. kimhj@dau.ac.kr
Abstract
-
PURPOSE: To evaluate the therapeutic activity and safety of paclitaxel and cisplatin combination chemotherapy in patients with advanced or metastatic gastric cancers that are unresponsive to primary chemotherapy.
MATERIALS AND METHODS
Advanced or metastatic gastric cancer patients unresponsive to first line chemotherapy were entered into this trial. The treatment regimen consisted of paclitaxel, 175 mg/m(2) by 3-hour infusion on day 1, and cisplatin, 60 mg/m(2) by 1 hour infusion on day 1, with the treatment repeated every 3 weeks.
RESULTS
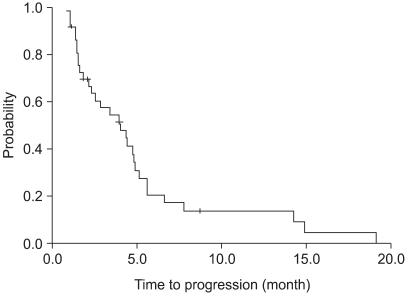
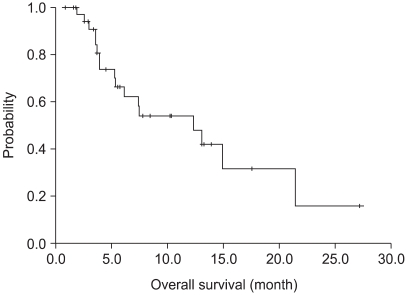
37 patients were entered in this study, with 32 fully evaluable for response. 4 (13%), 13 (40%) and 15 (47%) patients achieved a partial response, stable disease and progressed, respectively. The median time to progression was 4.0 months (95% CI: 2.0~6.0 months), and the median overall survival was 12.6 months (95% CI: 5.5~19.7 months), with a 1-year survival rate of 54%. Of a total of 135 cycles of chemotherapy, grades 3 and 4 hematological toxicities were neutropenia (14%) and anemia (3%). Grade > or =2 neuropathy was observed in 6 patients (17%).
CONCLUSION
The combination of paclitaxel and cisplatin is an effective and tolerable salvage treatment modality for advanced gastric cancer.
Keyword
MeSH Terms
Figure
Reference
-
1. Bae JM, Won YJ, Jung KW, Park JG. Annual report of the Korea Central Cancer Registry Program 2000: based on registered data from 131 hospitals. Cancer Res Treat. 2002; 34:77–83.
Article2. International Agency for Research on Cancer (IARC). 2000.3. Ho D. Ho D, editor. Epidemiologic studies in gastric cancer. Gastric cancer. 1988. New York, NY: Churchill Livingstone;p. 1–25.4. Wilke HJ, Van Cutsem E. Current treatments and future perspectives in colorectal and gastric cancer. Ann Oncol. 2003; 14(Suppl 2):ii49–ii55. PMID: 12810459.
Article5. Waters JS, Norman A, Cunningham D, Scarffe JH, Webb A, Harper P, et al. Long-term survival after epirubicin, cisplatin and fluorouracil for gastric cancer: results of a randomized trial. Br J Cancer. 1999; 80:269–272. PMID: 10390007.
Article6. Schiff PB, Fant J, Horwitz SB. Promotion of microtubule assembly in vitro by taxol. Nature. 1979; 277:665–667. PMID: 423966.
Article7. Jordan MA, Wendell K, Gardiner S, Derry WB, Copp H, Wilson L. Mitotic block induced in HeLa cells by low concentrations of paclitaxel (Taxol) results in abnormal mitotic exit and apoptotic cell death. Cancer Res. 1996; 56:816–825. PMID: 8631019.8. Schiller JH, Harrington D, Belani CP, Langer C, Sandler A, Krook J, et al. Comparison of four chemotherapy regimens for advanced non-small-cell lung cancer. N Engl J Med. 2002; 346:92–98. PMID: 11784875.
Article9. Ajani JA, Fairweather J, Dumas P, Patt YZ, Pazdur R, Mansfield PF. Phase II study of Taxol in patients with advanced gastric carcinoma. Cancer J Sci Am. 1998; 4:269–274. PMID: 9689986.10. Bokemeyer C, Lampe CS, Clemens MR, Hartmann JT, Quietzsch D, Forkmann L, et al. A phase II trial of paclitaxel and weekly 24 h infusion of 5-fluorouracil/folinic acid in patients with advanced gastric cancer. Anticancer Drugs. 1997; 8:396–399. PMID: 9180395.
Article11. Kollmannsberger C, Quietzsch D, Haag C, Lingenfelser T, Schroeder M, Hartmann JT, et al. A phase II study of paclitaxel, weekly, 24-hour continuous infusion 5-fluorouracil, folinic acid and cisplatin in patients with advanced gastric cancer. Br J Cancer. 2000; 83:458–462. PMID: 10945491.
Article12. Kim MK, Lee KH, Hyun MS, Do YR, Song HS, Lee WS, et al. A multi-center, phase II clinical trial of Padexol (paclitaxel) and cisplatin for patients suffering with advanced gastric cancer. Cancer Res Treat. 2005; 37:349–353.
Article13. Vanhoefer U, Harstrick A, Wilke H, Schleucher N, Walles H, Schroder J, et al. Schedule-dependent antagonism of paclitaxel and cisplatin in human gastric and ovarian carcinoma cell lines in vitro. Eur J Cancer. 1995; 31A:92–97. PMID: 7695986.
Article14. Therasse P, Arbuck SG, Eisenhauer EA, Wanders J, Kaplan RS, Rubinstein L, et al. European Organization for Research and Treatment of Cancer. National Cancer Institute of the United States. National Cancer Institute of Canada. New guidelines to evaluate the response to treatment in solid tumors. J Natl Cancer Inst. 2000; 92:205–216. PMID: 10655437.
Article15. Cascinu S, Graziano F, Cardarelli N, Marcellini M, Giordani P, Menichetti ET, et al. Phase II study of paclitaxel in pretreated advanced gastric cancer. Anticancer Drugs. 1998; 9:307–310. PMID: 9635920.
Article16. Ohtsu A, Boku N, Tamura F, Muro K, Shimada Y, Saigenji K, et al. An early phase II study of a 3-hour infusion of paclitaxel for advanced gastric cancer. Am J Clin Oncol. 1998; 21:416–419. PMID: 9708646.
Article17. Murad AM, Petroianu A, Guimaraes RC, Aragao BC, Cabral LO, Scalabrini-Neto AO. Phase II trial of the combination of paclitaxel and 5-fluorouracil in the treatment of advanced gastric cancer: a novel, safe, and effective regimen. Am J Clin Oncol. 1999; 22:580–586. PMID: 10597742.18. Kornek GV, Raderer M, Schull B, Fiebiger W, Gedlicka C, Lenauer A, et al. Effective combination chemotherapy with paclitaxel and cisplatin with or without human granulocyte colony-stimulating factor and/or erythropoietin in patients with advanced gastric cancer. Br J Cancer. 2002; 86:1858–1863. PMID: 12085176.
Article19. Ajani JA, Baker J, Pisters PW, Ho L, Mansfield PF, Feig BW, et al. Irinotecan/cisplatin in advanced, treated gastric or gastroesophageal junction carcinoma. Oncology. 2002; 16(5):Suppl 5. 16–18. PMID: 12109800.20. Suh SH, Kwon HC, Jo JH, Cho YR, Seo BG, Lee DM, et al. Oxaliplatin with biweekly low dose leucovorin and bolus and continuous infusion of 5-fluorouracil (Modified FOLFOX-4) as a salvage therapy for patients with advanced gastric cancer. Cancer Res Treat. 2005; 37:279–283.21. Assersohn L, Brown G, Cunningham D, Ward C, Oates J, Waters JS, et al. Phase II study of irinotecan and 5-fluorouracil/leucovorin in patients with primary refractory or relapsed advanced oesophageal and gastric carcinoma. Ann Oncol. 2004; 15:64–69. PMID: 14679122.
Article22. Chun JH, Kim HK, Lee JS, Choi JY, Lee HG, Yoon SM, et al. Weekly irinotecan in patients with metastatic gastric cancer failing cisplatin-based chemotherapy. Jpn J Clin Oncol. 2004; 34:8–13. PMID: 15020657.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- A Phase II Trial of Paclitaxel, 5-fluorouracil (5-FU) and Cisplatin in Patients with Metastatic or Recurrent Gastric Cancer
- Phase II Study of Cisplatin, Ifosfamide . Paclitaxel (CIP) as Neoadjuvant Chemotherapy in Patients with Locally Advanced Cervical Carcinoma
- Phase II Study of Paclitaxel, Cisplatin, and 5-Fluorouracil Combination Chemotherapy in Patients with Advanced Gastric Cancer
- Salvage Treatment for Advanced Gastric Cancer Using FEP (5-FU, Etoposide, Cisplatin) Combination Chemotherapy
- Combination chemotherapy with docetaxel and cisplatin as first-line treatment in advanced gastric cancer: is it a new effective chemotherapy?