J Korean Diabetes Assoc.
2007 May;31(3):220-229. 10.4093/jkda.2007.31.3.220.
Effects of Troglitazone on the Expression of VEGF and TGF-beta in Cultured Rat Mesangial Cells
- Affiliations
-
- 1Department of Internal Medicine, Konkuk University School of Medicine, Korea.
- 2Department of Internal Medicine, College of Medicine, Korea University, Korea.
- KMID: 1523010
- DOI: http://doi.org/10.4093/jkda.2007.31.3.220
Abstract
-
BACKGROUND: Clinical study reported that troglitazone ameliorated microalbuminuria in diabetic nephropathy. However, the mechanism of action is not fully understood. Vascular endothelial growth factor (VEGF) is known as vascular permeability factor and it is considered the most likely cause of glomerular hyperfiltration and proteinuria in diabetic nephropathy. Transforming growth factor-beta (TGF-beta) is a potent inducer of extracellular matrix production and fibrosis in renal cells and one of the important cytokine in the pathogenesis of diabetic nephropathy. To determine whether troglitazone affects VEGF and TGF-beta production in diabetic nephropathy, we examined the effects of troglitazone on the VEGF and TGF-beta expression in cultured rat mesangial cells exposed to high glucose concentration.
METHODS
Rat mesangial cells were cultured in media with D-glucose 5.5 mM (NG) or D-glucose 30 mM (HG), or D-glucose 30 mM/troglitazone 20 micrometer(HTz) and for 6, 24, or 72 hours, respectively. VEGF and TGF-beta expression were assessed by semiquantitative RT-PCR and western blot analysis.
RESULTS
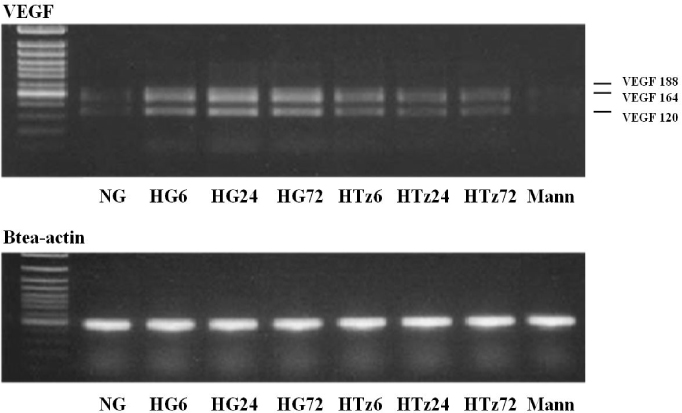
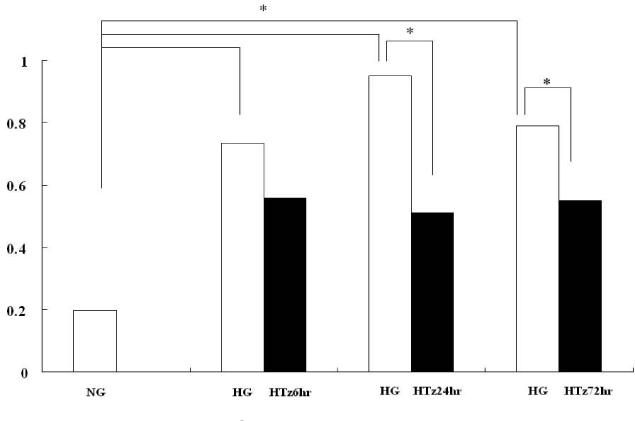
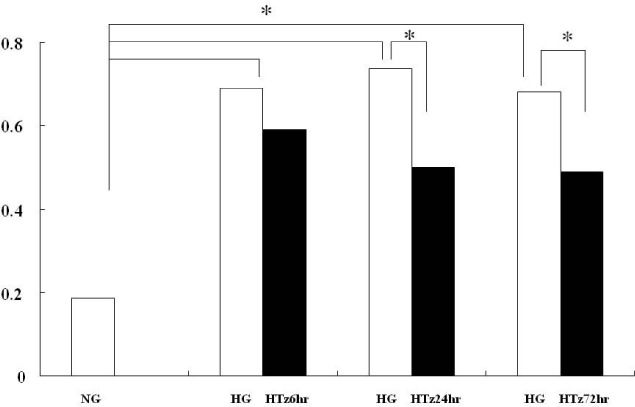
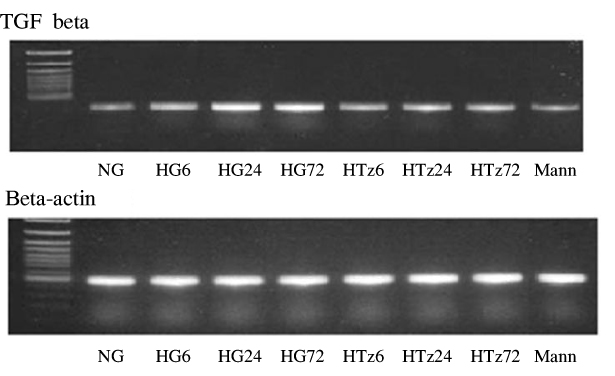
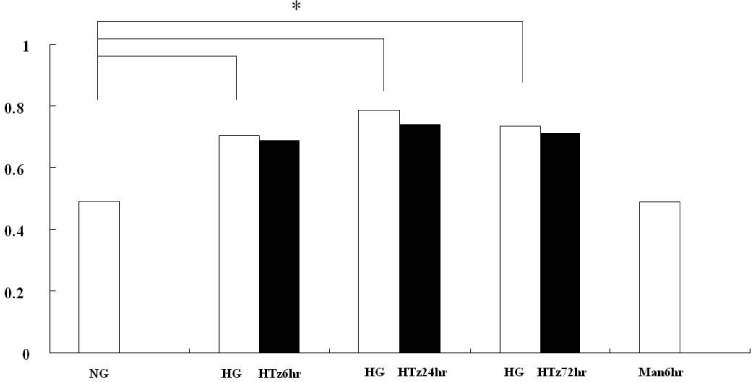
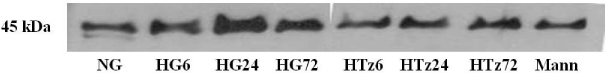
Troglitazone decreased the VEGF164 and VEGF120 mRNA expressions in cultured rat mesangial cells exposed to high glucose concentration with incubation for 24 and 72 hours, respectively. VEGF protein was also decreased in experimental group treated with troglitazone (HTz) than in those with HG for 24 and 72 hours. However troglitazone had no effect on the expression of TGF-beta mRNA in mesangial cells.
CONCLUSION
This study suggested that troglitazone may modulate the development and progression of diabetic nephropathy by reducing the expression of VEGF in mesangial cells
Keyword
MeSH Terms
Figure
Reference
-
1. Rossing P. Diabetic nephropathy: worldwide epidemic and effects of current treatment on natural history. Curr Diab Rep. 2006. 6:479–483.2. The Diabetes Control and Complications Trial Research Group. The effect of intensive treatment of diabetes on the development and progression of long-term complications in insulin-dependent diabetes mellitus. N Engl J Med. 1993. 329:977–986.3. Fujiwara T, Yoshioka S, Yoshioka T, Ushiyama I, Horikoshi H. Characterization of new oral antidiabetic agent CS-045.Studies inKK andob/ob mice and Zucker fatty rats. Diabetes. 1988. 37:1549–1558.4. lwamoto Y, Kuzuya T, Matsuda A, Awata T, Kumakura S, Inooka G, Shiraishi I. Effect of new oral antidiabetic agent CS-045 on glucose tolerance and insulin secretion in patients with NIDDM. Diabetes Care. 1991. 14:1083–1086.5. Nolan JJ, Ludvik B, Beerdsen P, Joyce M, Olefsky J. Improvement in glucose tolerance andinsulin resistance in obese subjects treated with troglitazone. N Engl J Med. 1994. 331:1188–1193.6. Imano E, Kanda T, Nakatani Y, Nishida T, Arai K, Motomura M, Kajimoto Y, Yamasaki Y, Hori M. Effect of troglitazone on microalbuminuria in patients with incipient diabetic nephropathy. Diabetes care. 1998. 21:2135–2139.7. Ziyadeh FN. Renal tubular basement membrane and collagen type IV in diabetes mellitus. Kidney Int. 1993. 43:114–120.8. Ziyadeh FN, Goldfarb S. The renal tubulointerstitium in diabetes melltus. Kidney Int. 1991. 39:464–475.9. Ziyadeh FN. The extracellular matrix in diabetic nephropathy. Am Kidney Dis. 1993. 22:736–744.10. UK Prospective Diabetes Study (UKPDS) Group. Intensive blood-glucose control with sulphonylureas or insulin compared with conventional treatment and risk of complications in patients with type 2 diabetes (UKPDS 33). Lancet. 1998. 352:837–853.11. Senger DR, Van Der Water L, Brown LF, Nagy JA, Yeo KT, Yeo TK, Berse B, Jackson RW, Dvorak AM. Vascular permeability factor in tumor biology. Cancer and Metastasis Rev. 1993. 12:303–324.12. Iijima K, Yoshikawa N, Connolly DT, Nakamura H. Human mesangial cells and peripheral blood mononuclear cells produce vascular permeability factor. Kidney Int. 1993. 44:959–966.13. Sharma K, Ziyadeh FN. The emerging role of transforming growth factor-β in kidney diseases. Am J Physiol. 1994. 35:F829–F842.14. Marre M, Leblance H, Suarez L, Guyenne TT, Menard J, Passa P. Converting enzyme inhibition and kidney function in normotensive diabetic patients with persistent microalbuminuria. Br Med J. 1987. 294:1448–1452.15. Fujii M, Takenura R, Yamaguchi M, Hasegawa G, Shigeta H, Nakano K, Kondo M. Troglitazone (CS-045) ameliorates albuminuria in streptozotocin-induced diabetic rats. Metabolism. 1997. 46:981–983.16. Nicholas SB, Kawano Y, Wakino S, Collins AR, Hsueh WA. Expression and function of peroxysome proliferator-activated receptor-γ in mesangial cells. Hypertension. 2001. 37(2):722–727.17. Ferrara N, Houck K, Jakeman L, Leung DW. Molecular and biological properties of the vascular endothelial growth factor. Endocrinol Rev. 1992. 13:18–32.18. Kim NH, Jung HH, Cha DR, Choi DS. Expression of VEGF in response to high glucose in rat mesangial cells. J Endocrinol. 2000. 165(3):617–624.19. Aiello LP, Avery RL, Arrigg PG, Keyt BA, Jampel HD, Shah ST, Pasquale LR, Thieme H, Iwamoto MA, Park JE, et al. Vascular endothelial growth factor in ocular fluid of patients with diabetic retinopathy and other retinal disorders. N Engl J Med. 1994. 331:1480–1487.20. Aiello LP, Pierce EA, Foley ED, Takagi H, Chen H, Riddle L, Ferrara N, King GL, Smith LE. Suppression of retinal neovascularization in vivo by inhibition of vascular endothelial growth factor (VEGF) using soluble VEGF-receptor chimeric proteins. Proc Natl Acad Sci USA. 1995. 92:10457–10461.21. Williams B, Baker AQ, Gallacher B, Lodwick D. Angiotensin II increases vascular permeability factor gene expression by human vascular smooth muscle cells. Hypertension. 1995. 25:913–917.22. Jakeman LB, Winer J, Benner GL, Altar CA, Ferrara N. Binding sites for vascular endothelial growth factor are localized on endothelial cells in adult rat tissues. J Clin Invest. 1992. 89:244–253.23. Singh AK, Gudehithlu KP, Pegoraro AA, Singh GK, Basheerudin K, Robey RB, Arruda JA, Dunea G. Vascular factors altered in glucose-treated mesangial cells and diabetic glomeruli. Changes in vascular factors impair endothelial cell growth and matrix. Lab Invest. 2004. 84:597–606.24. Shrma K, Ziyadeh FN. Hyperglycemia and diabetic kidney disease. The case for transforming growth factor-β as a key mediator. Diabetes. 1995. 44:1139–1146.25. Wolf G, Sharma K, Chen Y, Ericksen M, Ziyadeh FN. Highglucose-induced proliferation in mesangial cells is reversedautocrine TGF-β. Kidney Int. 1992. 42:647–666.26. Ziyadeh FN, Sharma K, Ericksen M, Wolf G. Stimulation of collagen gene expression and protein synthesis in murine mesangial cells by high glucose is mediated by activation of transformaing growth factor-β. J Clin Invest. 1994. 93:536–542.27. Nakamura T, Fukui M, Ebihara I, Osada S, Nagaoka I, Tomino Y, Koide H. mRNA expression of growth factors in glomeruli from diabetic rats. Diabetes. 1993. 42:450–456.28. Yamamoto T, Nakamura T, Noble NA, Ruoslahti E, Border WA. Expression of transforming growth factor beta is elevated in human and experimental diabetic nephropathy. Proc Natl Acad Sci USA. 1993. 90:1814–1818.29. Iwano M, Kubo A, Nishino T, Sato H, Nishioka H, Akai Y, Kurioka H, Fujii Y, Kanauchi M, Shiki H, Dohi K. Quantification of glomerular TGF-β1 mRNA in patients with diabetes mellitus. Kidney Int. 1996. 49:1120–1126.30. Sharma K, Ziyadeh FN, Alzahabi B, McGowan TA, Kapoor S, Kurnik BRC, Kurnik PB, Weisberg LS. Increased renal production of transforming growth factor-β1 in patients with type II diabetes. Diabetes. 1997. 46:854–859.31. Zheng F, Fornoni A, Elliot SJ, Guan Y, Breyer MD, Striker LJ, Striker GE. Upregulation of type I collagen by TGF-β in mesangial cells blocked by PPAR activation. Am J Physiol Renal Physiol. 2002. 282:639–648.32. lssiki K, Haneda M, Koya D, Maeda S, Sugimoto T, Kikkawa R. Thiazolidinedione compounds ameliorate glomerular dysfunction independent of their insulin-sensitizing action in diabetic rats. Diabetes. 2000. 49:1022–1032.33. Izumi T, Enomoto S, Hosiyama K, Sasahara K, Shibukawa A, Nakagawa T, Sugiyama Y. Prediction of the human pharmacokinetics of troglitazone, a new and extensively metabolized antidiabetic agent, after oral administration, with an animal scale-up approach. J Pharmacol Exp Ther. 1996. 277:1630–1641.34. Trachtman H, Futterweit S, Franki N, Singhal PC. Effect of vascular endothelial growth factor on nitric oxide production by cultured rat mesangial cells. Biochem Biophy Res Com. 1998. 245(2):443–446.35. Xin X, Yang S, Kowalski J, Gerritsen ME. Peroxisome proliferator-activated receptor gamma ligands are potent inhibitors of angiogenesis in vitro and in vivo. J Biol Chem. 1999. 274:9116–9121.36. Lehmann JM, Moore LB, Smith-Oliver TA, Wilkison WO, Wilson TM, Kliewer SA. An antidiabetic thiazolidinedione is a high affinity ligand for peroxisome proliferator-activated receptor (PPARγ). J Biol Chem. 1995. 270:12953–12956.37. Craven PA, Davidson CM, DeRuberts FR. Increase in diacylglycerol mass in isolated glomeruli by glucose from de novo synthesis of glycolipids. Diabetes. 1990. 39:667–674.38. Koya D, King GL. Protein kinase C activation and the development of diabetic complications. Diabetes. 1998. 47:859–866.39. Wheelerjones C, Abughazaleh R, Cospedal R, Houliston RA, Martin J, Zachary I. Vascular endothelial growth factor stimulates prostacyclin production and activation of cytosolic phospholipase A2 in endothelial cells via P42/P44 mitogen-activated protein kinase. FEBS Lett. 1997. 420:28–32.40. Amemiya T, Sasamura H, Mifune M, Kitamura Y, Hirahashi J, Hayashi M, Saruta T. Vascular endothelial growth factor activates MAP kinase and enhances collagen synthesis in human mesangial cells. Kidney Int. 1999. 56:2055–2063.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- The Effect of High Glucose and TGF-beta on the Cellular Injury in Cultured Glomerular Epithelial Cells
- The effects of high glucose concentration on angiotensin II- or transforming growth factor-beta-induced DNA synthesis, hypertrophy and collagen synthesis in cultured rat mesangial cells
- Attenuation of the Expression of Fibrogenic Molecules by Transforming Growth Factor-beta1 Antisense in Cultured Rat Mesangial Cells
- The Clinical Significance of VEGF and TGF-beta Expression in Osteosarcoma
- Effect of Protein Kinase C Inhibitors on Expression of TGF-betamRNA in Cultured Mesangial Cells Under High Glucose Concentration