Lab Anim Res.
2012 Sep;28(3):205-208. 10.5625/lar.2012.28.3.205.
Development of a type II diabetic mellitus animal model using Micropig(R)
- Affiliations
-
- 1Medikinetics Co., Ltd., Pyeong-Taek, Korea.
- 2Center for Agricultural Biomaterials, Seoul National University, Seoul, Korea.
- 3Qu-BEST Consulting Co., Ltd., Seongnam, Korea.
- 4Laboratory of Developmental Genetics, College of Medicine, Inha Univerisity, Incheon, Korea. wklee@inha.ac.kr
- 5Center for Advanced Medical Education by the BK21 Project, College of Medicine, Inha Univerisity, Incheon, Korea.
- KMID: 1436726
- DOI: http://doi.org/10.5625/lar.2012.28.3.205
Abstract
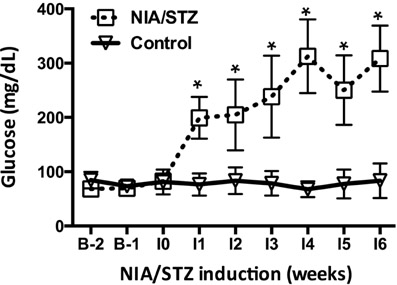
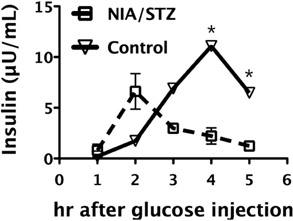
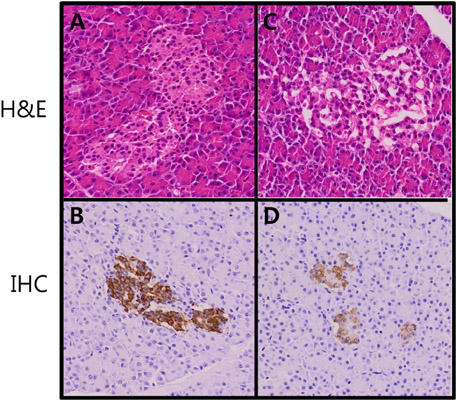
- Diabetes, which has shown an explosive increase in terms of its incidence, is regarded as a serious disease that must be overcome for the sake of human life. Among animal models used for testing of drug efficacy, the mini-pig model has shown a rapid upload due to its many similarities with human, particularly concerning the pharmacokinetics of compounds after subcutaneous administration, the structure and function of the gastrointestinal tract, the morphology of the pancreas, and overall metabolic status. Based on these various advantages, we sought to develop an animal model of type II diabetic mellitus using the Micro-pig, which differs from other miniature pigs. We used six male Micro-pigs for induction of a moderate insulin deficient model with nicotinamide (NIA)/streptozotocin (STZ) treatment and three animals for control. For evaluation of incidence of type II diabetes, we measured blood glucose level, and performed oral glucose tolerance test and immunohistochemistry on pancreatic tissue using insulin antibody. Compared to control animals, all animals treated with NIA/STZ showed high levels of glucose and low levels of insulin. In addition, we observed the partially destroyed beta cell population from tissue of the pancreas in treated animals. Based on these results, we report that the Micro-pig model developed in this study can be used for testing of the efficacy of therapeutic agents for treatment of Type 2 diabetic mellitus.
Keyword
MeSH Terms
Figure
Cited by 2 articles
-
Proteomic analysis of pancreas in miniature pigs according to developmental stages using two-dimensional electrophoresis and matrix-assisted laser desorption/ionization-time of flight mass spectrometry
Sun Shin Yi, Il Yong Kim, Sae Jin Oh, Hye Jung Yeom, Su Cheong Yeom, Seung Yong Hwang, Je Kyung Seong
Lab Anim Res. 2014;30(1):1-7. doi: 10.5625/lar.2014.30.1.1.Early changes in retinal structure and BMP2 expression in the retina and crystalline lens of streptozotocin-induced diabetic pigs
Jae Seung Jeong, Woon-Kyu Lee, Yeon Sung Moon, Na Rae Kim
Lab Anim Res. 2017;33(3):216-222. doi: 10.5625/lar.2017.33.3.216.
Reference
-
1. Georg P, Ludvik B. Lipids and Diabetes. J Clin Basic Cardiol. 2000. 3:159–162.2. Nyholm B, Pørksen N, Juhl CB, Gravholt CH, Butler PC, Weeke J, Veldhuis JD, Pincus S, Schmitz O. Assessment of insulin secretion in relatives of patients with type 2 (non-insulin-dependent) diabetes mellitus: evidence of early beta-cell dysfunction. Metabolism. 2000. 49(7):896–905.3. Harris MI. Definition and classification of diabetes mellitus and the criteria for diagnosis. Diabetes Mellitus: A Fundamental and Clinical Text. 2004. Philladelphia: Lippincott Williams & Wilkins;457–467.4. Sparre T, Larsen MR, Heding PE, Karlsen AE, Jensen ON, Pociot F. Unraveling the pathogenesis of type 1 diabetes with proteomics: present and future directions. Mol Cell Proteomics. 2005. 4(4):441–457.5. Bellinger DA, Merricks EP, Nichols TC. Swine models of type 2 diabetes mellitus: insulin resistance, glucose tolerance, and cardiovascular complications. ILAR J. 2006. 47(3):243–258.6. Miller ER, Ullrey DE. The pig as a model for human nutrition. Annu Rev Nutr. 1987. 7:361–382.7. Barth CA, Pfeuffer M, Scholtissek J. Animal models for the study of lipid metabolism, with particular reference to the Göttingen minipig. Adv Anim Physiol Anim Nutr. 1990. S20:39–49.8. Swindle MM, Smith AC. Comparative anatomy and physiology of the pig. Scand J Lab Anim Sci. 1998. 25:11–21.9. Larsen MO, Wilken M, Gotfredsen CF, Carr RD, Svendsen O, Rolin B. Mild streptozotocin diabetes in the Göttingen minipig. A novel model of moderate insulin deficiency and diabetes. Am J Physiol Endocrinol Metab. 2002. 282(6):E1342–E1351.10. Lee PY, Park SG, Kim EY, Lee MS, Chung SJ, Lee SC, Yu DY, Bae KH. Proteomic analysis of pancreata from mini-pigs treated with streptozotocin as a type I diabetes models. J Microbiol Biotechnol. 2010. 20(4):817–820.11. Koopmans SJ, Dekker R, Ackermans MT, Sauerwein HP, Serlie MJ, van Beusekom HM, van den Heuvel M, van der Giessen WJ. Dietary saturated fat/cholesterol, but not unsaturated fat or starch, induces C-reactive protein associated early atherosclerosis and ectopic fat deposition in diabetic pigs. Cardiovasc Diabetol. 2011. 10:64.12. Koopmans SJ, Mroz Z, Dekker R, Corbijn H, Ackermans M, Sauerwein H. Association of insulin resistance with hyperglycemia in streptozotocin-diabetic pigs: effects of metformin at isoenergetic feeding in a type 2-like diabetic pig model. Metabolism. 2006. 55(7):960–971.13. Pascoe WS, Storlien LH. Inducement by fat feeding of basal hyperglycemia in rats with abnormal beta-cell function. Model for study of etiology and pathogenesis of NIDDM. Diabetes. 1990. 39(2):226–233.14. Herr RR, Jahnke JK, Argoudelis AD. The structure of streptozotocin. J Am Chem Soc. 1967. 89(18):4808–4809.15. Yamamoto H, Uchigata Y, Okamoto H. Streptozotocin and alloxan induce DNA strand breaks and poly(ADP-ribose) synthetase in pancreatic islets. Nature. 1981. 294(5838):284–286.16. Schein PS, Cooney DA, McMenamin MG, Anderson T. Streptozotocin diabetes--further studies on the mechanism of depression of nicotinamide adenine dinucleotide concentrations in mouse pancreatic islets and liver. Biochem Pharmacol. 1973. 22(20):2625–2631.17. Masiello P, Bergamini E. Nicotinamide and streptozotocin diabetes in the rat. Factors influencing the effectiveness of the protection. Experientia. 1977. 33(9):1246–1247.18. Masiello P, Broca C, Gross R, Roye M, Manteghetti M, Hillaire-Buys D, Novelli M, Ribes G. Experimental NIDDM: development of a new model in adult rats administered streptozotocin and nicotinamide. Diabetes. 1998. 47(2):224–229.19. Fararh KM, Atoji Y, Shimizu Y, Takewaki T. Isulinotropic properties of Nigella sativa oil in Streptozotocin plus Nicotinamide diabetic hamster. Res Vet Sci. 2002. 73(3):279–282.20. Engle MJ, Perelman RH, McMahon KE, Langan SM, Farrell PM. Relationship between the severity of experimental diabetes and altered lung phospholipid metabolism. Proc Soc Exp Biol Med. 1984. 176(3):261–267.21. Rodrigues B, Poucheret P, Battel ML, McNeill JH. McNeill JH, editor. Streptozotocin-induced diabetes: induction, mechanism(s) and dose dependency. Experimental Models of Diabetes. 1999. Boca Raton, FL: CRC;3–19.22. Larsen MO, Rolin B, Wilken M, Carr RD, Gotfredsen CF. Measurements of insulin secretory capacity and glucose tolerance to predict pancreatic beta-cell mass in vivo in the nicotinamide/streptozotocin Göttingen minipig, a model of moderate insulin deficiency and diabetes. Diabetes. 2003. 52(1):118–123.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Clinical Analysis of Diabetic Retinopathy According to the Type of Diabetes Mellitus
- Development of osteoporosis animal model using micropigs
- Minimal Change Nephrotic Syndrome in a Patient with Type II Diabetic Mellitus and Review of the Literature
- Diabetic Nephropathy in Childhood and Adolescence (I) : Clinical Features
- Kruppel-Like Factor 2 Suppression by High Glucose as a Possible Mechanism of Diabetic Vasculopathy