J Vet Sci.
2012 Sep;13(3):219-222. 10.4142/jvs.2012.13.3.219.
Tissue distribution of sialic acid-linked influenza virus receptors in beagle dogs
- Affiliations
-
- 1College of Veterinary Medicine, South China Agricultural University, Guangzhou 510642, China. guihongzh@scau.edu.cn
- KMID: 1389759
- DOI: http://doi.org/10.4142/jvs.2012.13.3.219
Abstract
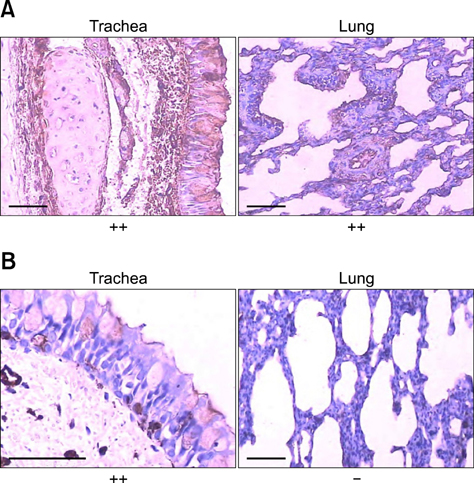
- Reports of influenza A virus infections in dogs has received considerable attention from veterinarians, virologists, and epidemiologists. Interaction between influenza viral hemagglutinin and cell oligosaccharides containing sialic acid residues results in infection. Sialic acids have an alpha-2,3-linkage to the penultimate galactose in the avian influenza virus receptor and an alpha-2,6-linkage in the human receptor. To date, there are no detailed data on the tissue distribution or histological features of either type of sialic acid-linked influenza virus receptors in beagle dogs, which are common laboratory animals and pets. We conducted the current study to visualize the in situ tissue distribution of both sialic acid-linked influenza virus receptors in various organs of beagle dogs using Maackia amurensis lectin II and Sambucus nigra agglutinin. Both alpha-2,3- and alpha-2,6-sialic acid-linked receptors were detected in the endothelial cells of the respiratory tract and other organs. Endothelial cells of most gastrointestinal organs were negative for alpha-2,3-sialic acid-linked receptors in the dogs. Our results suggested that these canine organs may be affected by influenza virus infection. The findings from our study will also help evaluate the occurrence and development of influenza virus infections in dogs.
Keyword
MeSH Terms
-
Animals
Dog Diseases/metabolism
Dogs/metabolism/*virology
Female
Influenza A Virus, H5N1 Subtype/*metabolism
Maackia/chemistry
Male
N-Acetylneuraminic Acid/metabolism
Organ Specificity
Orthomyxoviridae Infections/metabolism/transmission/veterinary
Plant Lectins/metabolism
Receptors, Cell Surface/analysis/chemistry/metabolism
Receptors, Virus/analysis/chemistry/*metabolism
Sambucus nigra/chemistry
Figure
Reference
-
1. Amonsin A, Songserm T, Chutinimitkul S, Jam-on R, Sae-Heng N, Pariyothorn N, Payungporn S, Theamboonlers A, Poovorawan Y. Genetic analysis of influenza A virus (H5N1) derived from domestic cat and dog in Thailand. Arch Virol. 2007. 152:1925–1933.
Article2. Cardona CJ, Xing Z, Sandrock CE, Davis CE. Avian influenza in birds and mammals. Comp Immunol Microbiol Infect Dis. 2009. 32:255–273.
Article3. Cleaveland S, Meslin FX, Breiman R. Dogs can play useful role as sentinel hosts for disease. Nature. 2006. 440:605.
Article4. Crawford PC, Dubovi EJ, Castleman WL, Stephenson I, Gibbs EPJ, Chen L, Smith C, Hill RC, Ferro P, Pompey J, Bright RA, Medina MJ, Johnson CM, Olsen CW, Cox NJ, Klimov AI, Katz JM, Donis RO. Influenza Genomics Group. Transmission of equine influenza virus to dogs. Science. 2005. 310:482–485.
Article5. Daly JM, Blunden AS, Macrae S, Miller J, Bowman SJ, Kolodziejek J, Nowotny N, Smith KC. Transmission of equine influenza virus to English foxhounds. Emerg Infect Dis. 2008. 14:461–464.
Article6. Deshpande MS, Abdelmagid O, Tubbs A, Jayappa H, Wasmoen T. Experimental reproduction of canine influenza virus H3N8 infection in young puppies. Vet Ther. 2009. 10:29–39.7. Giese M, Harder TC, Teifke JP, Klopfleisch R, Breithaupt A, Mettenleiter TC, Vahlenkamp TW. Experimental infection and natural contact exposure of dogs with avian influenza virus (H5N1). Emerg Infect Dis. 2008. 14:308–310.
Article8. Harder TC, Vahlenkamp TW. Influenza virus infections in dogs and cats. Vet Immunol Immunopathol. 2010. 134:54–60.
Article9. Ibricevic A, Pekosz A, Walter MJ, Newby C, Battaile JT, Brown EG, Holtzman MJ, Brody SL. Influenza virus receptor specificity and cell tropism in mouse and human airway epithelial cells. J Virol. 2006. 80:7469–7480.
Article10. Kirkland PD, Finlaison DS, Crispe E, Hurt AC. Influenza virus transmission from horses to dogs, Australia. Emerg Infect Dis. 2010. 16:699–702.
Article11. Kishimoto C, Hiraoka Y, Takada H, Kurokawa M, Ochiai H. Effects of immunoglobulin on murine myocarditis caused by influenza A virus: experimental study. J Cardiol. 2005. 45:247–255.12. Kitaura Y, Deguchi H, Terasaki F, Ukimura A, Morita H, Tatsumi T. Influenza myocarditis--pathophysiology and developmental mechanism of myocarditis. Nihon Rinsho. 2003. 61:1945–1952.13. Matrosovich MN, Gambaryan AS, Teneberg S, Piskarev VE, Yamnikova SS, Lvov DK, Robertson JS, Karlsson KA. Avian influenza A viruses differ from human viruses by recognition of sialyloligosaccharides and gangliosides and by a higher conservation of the HA receptor-binding site. Virology. 1997. 233:224–234.
Article14. Nelli RK, Kuchipudi SV, White GA, Perez BB, Dunham SP, Chang KC. Comparative distribution of human and avian type sialic acid influenza receptors in the pig. BMC Vet Res. 2010. 6:4.
Article15. Ning ZY, Luo MY, Qi WB, Yu B, Jiao PR, Liao M. Detection of expression of influenza virus receptors in tissues of BALB/c mice by histochemistry. Vet Res Commun. 2009. 33:895–903.
Article16. Payungporn S, Crawford PC, Kouo TS, Chen LM, Pompey J, Castleman WL, Dubovi EJ, Katz JM, Donis RO. Influenza A virus (H3N8) in dogs with respiratory disease, Florida. Emerg Infect Dis. 2008. 14:902–908.
Article17. Shinya K, Ebina M, Yamada S, Ono M, Kasai N, Kawaoka Y. Avian flu: influenza virus receptors in the human airway. Nature. 2006. 440:435–436.18. Shu Y, Li CK, Li Z, Gao R, Liang Q, Zhang Y, Dong L, Zhou J, Dong J, Wang D, Wen L, Wang M, Bai T, Li D, Dong X, Yu H, Yang W, Wang Y, Feng Z, McMichael AJ, Xu XN. Avian influenza A (H5N1) viruses can directly infect and replicate in human gut tissues. J Infect Dis. 2010. 201:1173–1177.
Article19. Song D, Kang B, Lee C, Jung K, Ha G, Kang D, Park S, Park B, Oh J. Transmission of avian influenza virus (H3N2) to dogs. Emerg Infect Dis. 2008. 14:741–746.
Article20. Song D, Lee C, Kang B, Jung K, Oh T, Kim H, Park B, Oh J. Experimental infection of dogs with avian-origin canine influenza A virus (H3N2). Emerg Infect Dis. 2009. 15:56–58.
Article21. Songserm T, Amonsin A, Jam-on R, Sae-Heng N, Pariyothorn N, Payungporn S, Theamboonlers A, Chutinimitkul S, Thanawongnuwech R, Poovorawan Y. Fatal avian influenza A H5N1 in a dog. Emerg Infect Dis. 2006. 12:1744–1747.
Article22. Stevens J, Blixt O, Tumpey TM, Taubenberger JK, Paulson JC, Wilson IA. Structure and receptor specificity of the hemagglutinin from an H5N1 influenza virus. Science. 2006. 312:404–410.
Article23. Suzuki Y. Sialobiology of influenza: molecular mechanism of host range variation of influenza viruses. Biol Pharm Bull. 2005. 28:399–408.
Article24. Thompson CI, Barclay WS, Zambon MC, Pickles RJ. Infection of human airway epithelium by human and avian strains of influenza A virus. J Virol. 2006. 80:8060–8068.
Article25. Yao L, Korteweg C, Hsueh W, Gu J. Avian influenza receptor expression in H5N1-infected and noninfected human tissues. FASEB J. 2008. 22:733–740.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Expression patterns of influenza virus receptors in the respiratory tracts of four species of poultry
- Experimental infection of dogs with highly pathogenic avian influenza virus (H5N8)
- An Universal Approach to Getting Ahead for Influenza B Vaccines
- Influenza virus vaccine for neglected hosts: horses and dogs
- Binding of the Streptococcus gordonii Surface Glycoprotein Hsa to alpha(2-3) Linked Sialic Acid Residues on Fibronectin