J Vet Sci.
2010 Jun;11(2):143-149. 10.4142/jvs.2010.11.2.143.
Evaluation of PCR inhibitory effect of enrichment broths and comparison of DNA extraction methods for detection of Salmonella Enteritidis using real-time PCR assay
- Affiliations
-
- 1Department of Public Health, College of Veterinary Medicine, Konkuk University, Seoul 143-701, Korea. bracstu3@konkuk.ac.kr
- 2Department of Infectious Diseases, College of Veterinary Medicine, Konkuk University, Seoul 143-701, Korea.
- 3Korea Food and Drug Administration, Seoul 122-704, Korea.
- 4Department of Animal Biotechnology, College of Animal Bioscience and Technology, Konkuk University, Seoul 143-701, Korea.
- KMID: 1110861
- DOI: http://doi.org/10.4142/jvs.2010.11.2.143
Abstract
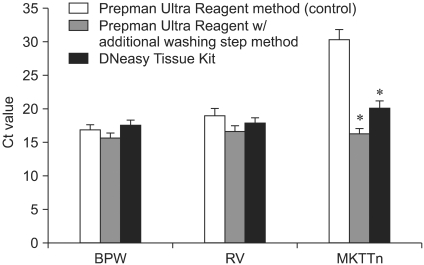
- The best enrichment broth and DNA extraction scheme was determined for rapid and sensitive detection of Salmonella Enteritidis in steamed pork using real-time PCR. The inhibitory effect of commonly used Salmonella enrichment broths, Rappaport-Vassiliadis (RV) and Muller-Kauffmann tetrathionate with novobiocin (MKTTn), on real-time PCR was confirmed. The inhibition of PCR was statistically significant (p < 0.05) in RV and MKTTn, as compared with buffered peptone water (BPW) or phosphate-buffered saline. The inhibitory effect of the selective enrichment media was successfully removed by using a modified DNA extraction, PrepMan Ultra Reagent with an additional washing step or the DNeasy Tissue Kit. In three experiments, when applied to detection of Salmonella Enteritidis in steamed pork, the real-time PCR coupled with single 24 h enrichment with BPW performed better than double 48 h enrichment with BPW plus RV or MKTTn. The simple real-time PCR assay using BPW proved to be a rapid and sensitive test for detection of low concentrations of Salmonella Enteritidis in steamed pork samples as compared with the conventional culture method.
Keyword
MeSH Terms
Figure
Reference
-
1. Al-Soud WA, Ouis IS, Li DQ, Ljungh S, Wadström T. Characterization of the PCR inhibitory effect of bile to optimize real-time PCR detection of Helicobacter species. FEMS Immunol Med Microbiol. 2005; 44:177–182. PMID: 15866213.2. Bansal NS, Gray V, McDonell F. Validated PCR assay for the routine detection of Salmonella in food. J Food Prot. 2006; 69:282–287. PMID: 16496566.3. Catarame TMG, O'Hanlon KA, McDowell DA, Blair IS, Duffy G. Comparison of a real-time polymerase chain reaction assay with a culture method for the detection of Salmonella in retail meat samples. J Food Saf. 2006; 26:1–15.4. Culp SJ, Beland FA, Heflich RH, Benson RW, Blankenship LR, Webb PJ, Mellick PW, Trotter RW, Shelton SD, Greenlees KJ, Manjanatha MG. Mutagenicity and carcinogenicity in relation to DNA adduct formation in rats fed leucomalachite green. Mutat Res. 2002; 506-507:55–63. PMID: 12351145.
Article5. Heller LC, Davis CR, Peak KK, Wingfield D, Cannons AC, Amuso PT, Cattani J. Comparison of methods for DNA isolation from food samples for detection of Shiga toxin-producing Escherichia coli by real-time PCR. Appl Environ Microbiol. 2003; 69:1844–1846. PMID: 12620880.6. Hughes D, Dailianis AE, Hill L, Curiale MS, Gangar V. Salmonella in foods: new enrichment procedure for TECRA Salmonella Visual Immunoassay using a single rv(R10) only, TT only, or dual rv(R10) and TT selective enrichment broths (AOAC official method 998.09): collaborative study. J AOAC Int. 2003; 86:775–790. PMID: 14509439.7. Maher N, Dillon HK, Vermund SH, Unnasch TR. Magnetic bead capture eliminates PCR inhibitors in samples collected from the airborne environment, permitting detection of Pneumocystis carinii DNA. Appl Environ Microbiol. 2001; 67:449–452. PMID: 11133478.8. Malorny B, Hoorfar J. Toward standardization of diagnostic PCR testing of fecal samples: lessons from the detection of Salmonellae in pigs. J Clin Microbiol. 2005; 43:3033–3037. PMID: 16000411.
Article9. Malorny B, Paccassoni E, Fach P, Bunge C, Martin A, Helmuth R. Diagnostic real-time PCR for detection of Salmonella in food. Appl Environ Microbiol. 2004; 70:7046–7052. PMID: 15574899.10. Monteiro L, Bonnemaison D, Vekris A, Petry KG, Bonnet J, Vidal R, Cabrita J, Mégraud F. Complex polysaccharides as PCR inhibitors in feces: Helicobacter pylori model. J Clin Microbiol. 1997; 35:995–998. PMID: 9157172.11. Myint MS, Johnson YJ, Tablante NL, Heckert RA. The effect of pre-enrichment protocol on the sensitivity and specificity of PCR for detection of naturally contaminated Salmonella in raw poultry compared to conventional culture. Food Microbiol. 2006; 23:599–604. PMID: 16943057.12. Oliveira SD, Rodenbusch CR, Cé MC, Rocha SL, Canal CW. Evaluation of selective and non-selective enrichment PCR procedures for Salmonella detection. Lett Appl Microbiol. 2003; 36:217–221. PMID: 12641714.13. Rodríguez-Lázaro D, D'Agostino M, Pla M, Cook N. Construction strategy for an internal amplification control for real-time diagnostic assays using nucleic Acid sequence-based amplification: development and clinical application. J Clin Microbiol. 2004; 42:5832–5836. PMID: 15583319.
Article14. Rossen L, Nørskov P, Holmstrøm K, Rasmussen OF. Inhibition of PCR by components of food samples, microbial diagnostic assays and DNA-extraction solutions. Int J Food Microbiol. 1992; 17:37–45. PMID: 1476866.
Article15. Uyttendaele M, Vanwildemeersch K, Debevere J. Evaluation of real-time PCR vs automated ELISA and a conventional culture method using a semi-solid medium for detection of Salmonella. Lett Appl Microbiol. 2003; 37:386–391. PMID: 14633109.16. Uyttendaele MR, Debevere JM, Lips RM, Neyts KD. Prevalence of Salmonella in poultry carcasses and their products in Belgium. Int J Food Microbiol. 1998; 40:1–8. PMID: 9600604.17. Wiedbrauk DL, Werner JC, Drevon AM. Inhibition of PCR by aqueous and vitreous fluids. J Clin Microbiol. 1995; 33:2643–2646. PMID: 8567898.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- A multiplex real-time PCR for differential detection and quantification of Salmonella spp., Salmonella enterica serovar Typhimurium and Enteritidis in meats
- Application of SYBR Green real-time PCR assay for the specific detection of Salmonella spp
- Multidrug-resistant Salmonella typhimurium and Salmonella enteritidis Identified by Multiplex PCR in Korea
- Evaluation of the AdvanSure TB/NTM Plus Real-Time PCR Assay for the Simultaneous Detection of Mycobacterium tuberculosis and Nontuberculous Mycobacteria from Clinical Specimens
- Performance Evaluation of the Real-Q Cytomegalovirus (CMV) Quantification Kit Using Two Real-Time PCR Systems for Quantifying CMV DNA in Whole Blood