J Vet Sci.
2009 Mar;10(1):43-51. 10.4142/jvs.2009.10.1.43.
A multiplex real-time PCR for differential detection and quantification of Salmonella spp., Salmonella enterica serovar Typhimurium and Enteritidis in meats
- Affiliations
-
- 1National Veterinary Research and Quarantine Service, Anyang 430-824, Korea.
- 2Department of Infectious Diseases, College of Veterinary Medicine, KRF Priority Zoonotic Disease Research Institute and BK21 Program for Veterinary Science, Seoul National University, Seoul 151-742, Korea. yoohs@snu.ac.kr
- KMID: 1089345
- DOI: http://doi.org/10.4142/jvs.2009.10.1.43
Abstract
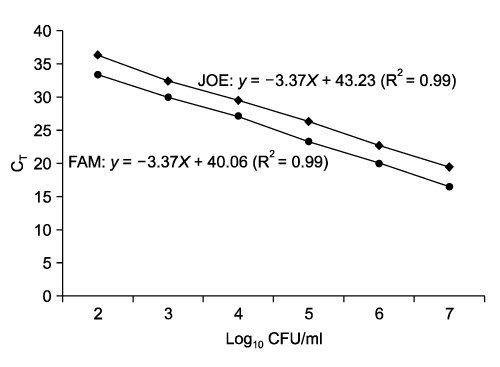
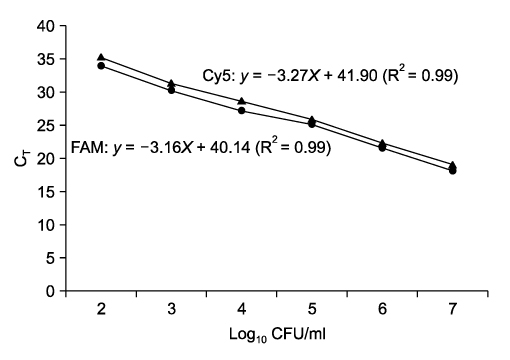
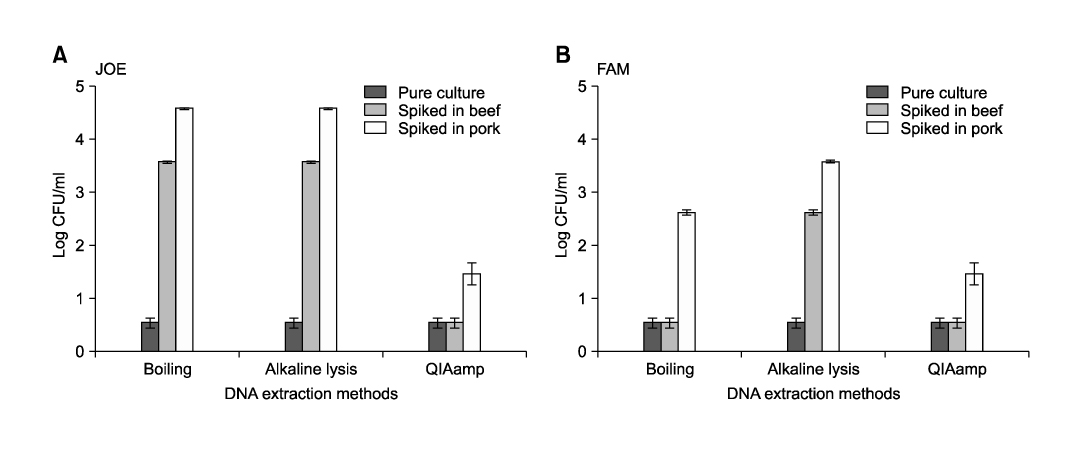
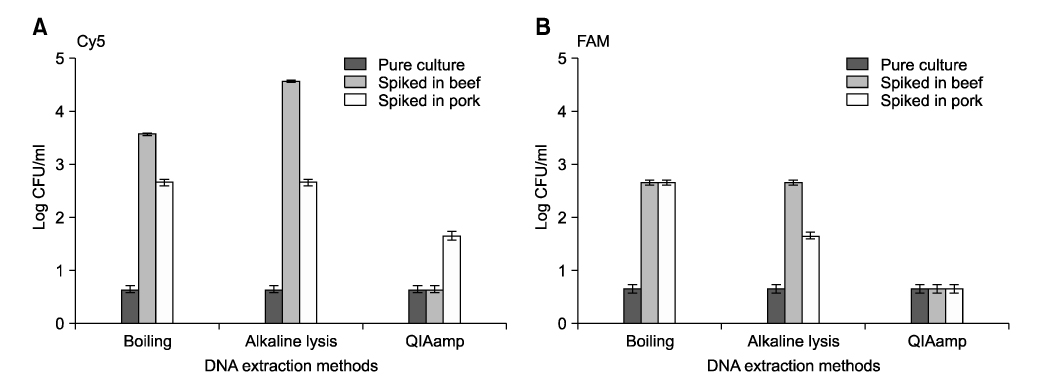
- Salmonella (S.) Typhimurium and S. Enteritidis are the major causative agents of food-borne illnesses worldwide. Currently, a rapid detection system using multiplex real-time polymerase chain reaction (PCR) has been applied for other food-borne pathogens such as Escherichia coli, Staphylococcus aureus and Streptococcus spp. A multiplex real-time PCR was developed for the simultaneous detection of Salmonella spp., especially S. Typhimurium and S. Enteritidis, in beef and pork. For the specific and sensitive multiplex real-time PCR, three representative primers and probes were designed based on sequence data from Genbank. Among the three DNA extraction methods (boiling, alkaline lysis, and QIAamp DNA Mini Kit), the QIAamp DNA Mini Kit was the most sensitive in this study. The optimized multiplex real-time PCR was applied to artificially inoculated beef or pork. The detection sensitivity of the multiplex real-time PCR was increased. The specificity of the multiplex real-time PCR assay, using 128 pure-cultured bacteria including 110 Salmonella isolates and 18 non-Salmonella isolates, was 100%, 100% and 99.1% for Salmonella spp., S. Typhimurium and S. Enteritidis, respectively. The sensitivity was 100%, 100% and 91.7% for Salmonella spp., S. Typhimurium and S. Enteritidis, respectively. The multiplex real-time PCR assay developed in this study could detect up to 0.54 +/- 0.09 and 0.65 +/- 0.07 log10 CFU/ml for S. Typhimurium and S. Enteritidis for beef, 1.45 +/- 0.21 and 1.65 +/- 0.07 log10 CFU/ml for S. Typhimurium and S. Enteritidis for pork, respectively, with all conditions optimized. Our results indicated that the multiplex real-time PCR assay developed in this study could sensitively detect Salmonella spp. and specifically differentiate S. Typhimurium from S. Enteritidis in meats.
MeSH Terms
Figure
Cited by 1 articles
-
An overview of calf diarrhea - infectious etiology, diagnosis, and intervention
Yong-il Cho, Kyoung-Jin Yoon
J Vet Sci. 2014;15(1):1-17. doi: 10.4142/jvs.2014.15.1.1.
Reference
-
1. Andrews WH, Hammack TS. Merker RL, editor. Salmonella. Bacteriological Analytical Manual. 1998. 8th ed. Gaitherburg: AOAC International;5.01–5.20. Chapter 5.2. Baay MF, Huis in't Veld JH. Alternative antigens reduce cross-reactions in an ELISA for the detection of Salmonella enteritidis in poultry. J Appl Bacteriol. 1993. 74:243–247.
Article3. Barrow PA. Serological diagnosis of Salmonella serotype Enteritidis infections in poultry by ELISA and other tests. Int J Food Microbiol. 1994. 21:55–68.
Article4. Bennett AR, Greenwood D, Tennant C, Banks JG, Betts RP. Rapid and definitive detection of Salmonella in foods by PCR. Lett Appl Microbiol. 1998. 26:437–441.5. Chiu CH, Ou JT. Rapid identification of Salmonella serovars in feces by specific detection of virulence genes, invA and spvC, by an enrichment broth culture-multiplex PCR combination assay. J Clin Microbiol. 1996. 34:2619–2622.
Article6. Clouthier SC, Müller KH, Doran JL, Collinson SK, Kay WW. Characterization of three fimbrial genes, sefABC, of Salmonella enteritidis. J Bacteriol. 1993. 175:2523–2533.
Article7. De Medici D, Croci L, Delibato E, Di Pasquale S, Filetici E, Toti L. Evaluation of DNA extraction methods for use in combination with SYBR green I real-time PCR to detect Salmonella enterica serotype Enteritidis in poultry. Appl Environ Microbiol. 2003. 69:3456–3461.
Article8. Fey A, Eichler S, Flavier S, Christen R, Höfle MG, Guzmán CA. Establishment of a real-time PCR-based approach for accurate quantification of bacterial RNA targets in water, using Salmonella as a model organism. Appl Environ Microbiol. 2004. 70:3618–3623.
Article9. Gillespie BE, Oliver SP. Simultaneous detection of mastitis pathogens, Staphylococcus aureus, Streptococcus uberis, and Streptococcus agalactiae by multiplex real-time polymerase chain reaction. J Dairy Sci. 2005. 88:3510–3518.
Article10. Grant MA, Hu J, Jinneman KC. Multiplex real-time PCR detection of heat-labile and heat-stable toxin genes in enterotoxigenic Escherichia coli. J Food Prot. 2006. 69:412–416.
Article11. Hein I, Flekna G, Krassnig M, Wagner M. Real-time PCR for the detection of Salmonella spp. in food: An alternative approach to a conventional PCR system suggested by the FOOD-PCR project. J Microbiol Methods. 2006. 66:538–547.
Article12. Holicka J, Guy RA, Kapoor A, Shepherd D, Horgen PA. A rapid (one day), sensitive real-time polymerase chain reaction assay for detecting Escherichia coli O157:H7 in ground beef. Can J Microbiol. 2006. 52:992–998.
Article13. Imre A, Olasz F, Nagy B. Development of a PCR system for the characterisation of Salmonella flagellin genes. Acta Vet Hung. 2005. 53:163–172.
Article14. International Organization for Standardization (ISO). Microbiology of food and animal feeding stuffs. Horizontal method for the detection of Salmonella (ISO 6579:2003). 2003. Geneva: ISO.15. Kent PT, Thomason BM, Morris GK. Salmonellae in Foods and Feeds. 1981. Atlanta: USA : Department of Health and Human Services;29.16. Korea Food & Drug Administration (KFDA). Food & Drug Statistical Yearbook. 2008. Seoul: KFDA;142.17. Lin CK, Hung CL, Hsu SC, Tsai CC, Tsen HY. An improved PCR primer pair based on 16S rDNA for the specific detection of Salmonella serovars in food samples. J Food Prot. 2004. 67:1335–1343.
Article18. Mackinnon A. A spreadsheet for the calculation of comprehensive statistics for the assessment of diagnostic tests and inter-rater agreement. Comput Biol Med. 2000. 30:127–134.
Article19. Malorny B, Paccassoni E, Fach P, Bunge C, Martin A, Helmuth R. Diagnostic real-time PCR for detection of Salmonella in food. Appl Environ Microbiol. 2004. 70:7046–7052.
Article20. Mead PS, Slutsker L, Dietz V, McCaig LF, Bresee JS, Shapiro C, Griffin PM, Tauxe RV. Food-related illness and death in the United States. Emerg Infect Dis. 1999. 5:607–625.
Article21. Miyamoto T, Tian HZ, Okabe T, Trevanich S, Asoh K, Tomoda S, Honjoh K, Hatano S. Application of random amplified polymorphic DNA analysis for detection of Salmonella spp. in foods. J Food Prot. 1998. 61:785–791.
Article22. Pang T, Bhutta ZA, Finlay BB, Altwegg M. Typhoid fever and other salmonellosis: a continuing challenge. Trends Microbiol. 1995. 3:253–255.
Article23. Quinn C, Ward J, Griffin M, Yearsley D, Egan J. A comparison of conventional culture and three rapid methods for the detection of Salmonella in poultry feeds and environmental samples. Lett Appl Microbiol. 1995. 20:89–91.
Article24. Rossmanith P, Krassnig M, Wagner M, Hein I. Detection of Listeria monocytogenes in food using a combined enrichment/real-time PCR method targeting the prfA gene. Res Microbiol. 2006. 157:763–771.
Article25. Sails AD, Fox AJ, Bolton FJ, Wareing DR, Greenway DL. A real-time PCR assay for the detection of Campylobacter jejuni in foods after enrichment culture. Appl Environ Microbiol. 2003. 69:1383–1390.
Article26. Seo KH, Valentin-Bon IE, Brackett RE. Detection and enumeration of Salmonella Enteritidis in homemade ice cream associated with an outbreak: comparison of conventional and real-time PCR methods. J Food Prot. 2006. 69:639–643.
Article27. Seo KH, Valentin-Bon IE, Brackett RE, Holt PS. Rapid, specific detection of Salmonella Enteritidis in pooled eggs by real-time PCR. J Food Prot. 2004. 67:864–869.
Article28. Soumet C, Ermel G, Rose V, Rose N, Drouin P, Salvat G, Colin P. Identification by a multiplex PCR-based assay of Salmonella Typhimurium and Salmonella Enteritidis strains from environmental swabs of poultry houses. Lett Appl Microbiol. 1999. 29:1–6.
Article29. Szmolka A, Kaszanyitzky E, Nagy B. Improved diagnostic and real-time PCR in rapid screening for Salmonella in the poultry food chain. Acta Vet Hung. 2006. 54:297–312.
Article30. Tan W, Shelef LA. Automated detection of Salmonella spp. in foods. J Microbiol Methods. 1999. 37:87–91.31. Trkov M, Avguštin G. An improved 16S rRNA based PCR method for the specific detection of Salmonella enterica. Int J Food Microbiol. 2003. 80:67–75.
Article32. Wilson MA, Rimler RB, Hoffman LJ. Comparison of DNA fingerprints and somatic serotypes of serogroup B and E Pasteurella multocida isolates. J Clin Microbiol. 1992. 30:1518–1524.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Multidrug-resistant Salmonella typhimurium and Salmonella enteritidis Identified by Multiplex PCR in Korea
- A Virulent Salmonella enterica Serovar Enteritidis Phage SE2 with a Strong Bacteriolytic Activity of Planktonic and Biofilmed Cells
- Salmonella Serovars from Foodborne and Waterborne Diseases in Korea, 1998-2007: Total Isolates Decreasing Versus Rare Serovars Emerging
- Protection Against Salmonella Typhimurium, Salmonella Gallinarum, and Salmonella Enteritidis Infection in Layer Chickens Conferred by a Live Attenuated Salmonella Typhimurium Strain
- Application of SYBR Green real-time PCR assay for the specific detection of Salmonella spp