J Vet Sci.
2008 Mar;9(1):45-50. 10.4142/jvs.2008.9.1.45.
Serum immunoglobulin fused interferon-alpha inhibited tumor growth in athymic mice bearing colon 26 adenocarcinoma cells
- Affiliations
-
- 1Laboratory of Toxicology, College of Veterinary Medicine, Seoul National University, Seoul 151-742, Korea. mchotox@snu.ac.kr
- 2Interdisciplinary Program in Nano-Science and Technology, Seoul National University, Seoul 151-742, Korea.
- 3Hanmi Pharmaceutical Research Center, Hwaseong 445-813, Korea.
- KMID: 1102953
- DOI: http://doi.org/10.4142/jvs.2008.9.1.45
Abstract
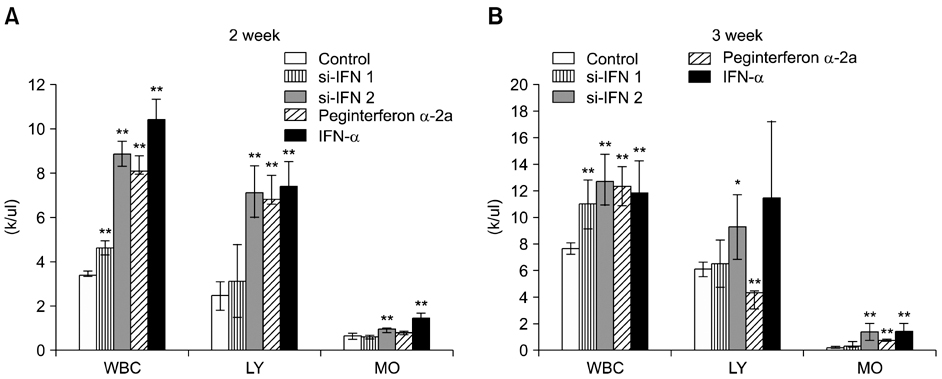
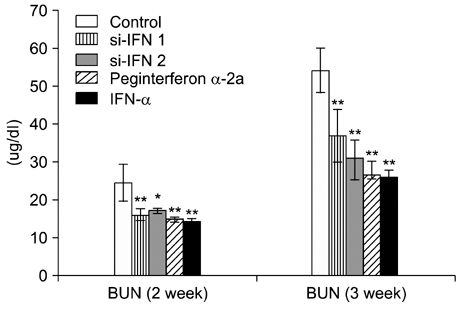
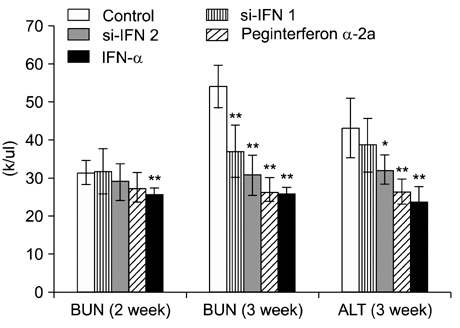
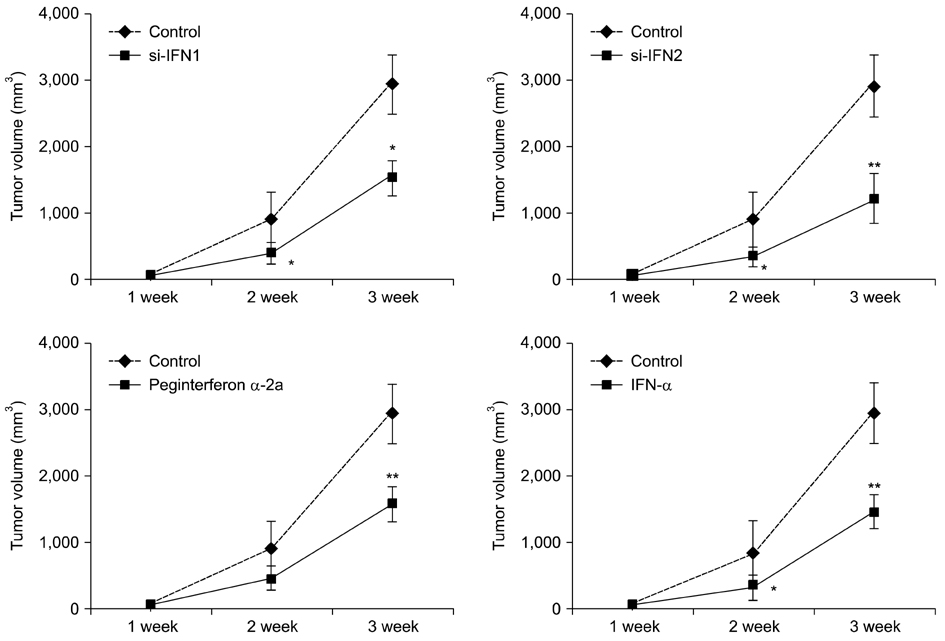
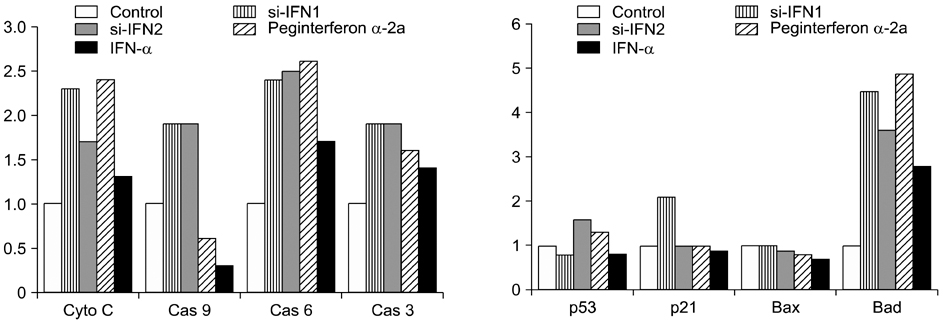
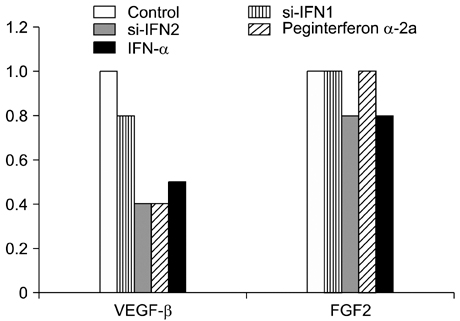
- Interferon (IFN) has therapeutic potential for a wide range of infectious and proliferative disorders. However, the half-life of IFN is too short to have a stable therapeutic effect. To overcome this problem, serum immunoglobulin has been fused to IFN. In this study, the efficacy of serum immunoglobulin fused INFs (si-IFN1 and si-IFN2) was evaluated on athymic mice bearing colon 26 adenocarcinoma cells. Seven days after the implantation of tumor cells, each group of mice was injected once a week with si-IFN1 and si-IFN2 at two different concentrations (10 x : 30 microgram/kg and 50 x : 150 microgram/kg). A slight anti-tumoral effect was observed in all 10 x groups compared to the control. In the 50 x groups, however, si-IFN1 and si-IFN2 showed significant anti- tumoral effects compared to the control. To gain more information on the mechanisms associated with the decrease of tumor size, a Western blot assay of apoptosis-related molecules was performed. The protein expression of cytochrome c, caspase 9, 6, and 3 were increased by si-IFN1 and si-IFN2. These 2 IFNs also increased the expressions of p53, p21, Bax and Bad. Interestingly, si-IFN1 and si-IFN2 decreased the expression of VEGF-beta. Taken together, serum immunoglobulin fused IFNs increased therapeutic efficacy under current experimental condition.
MeSH Terms
-
Adenocarcinoma/*drug therapy
Alanine Transaminase/blood
Animals
Antineoplastic Agents/chemistry/pharmacology
Blood Urea Nitrogen
Dose-Response Relationship, Drug
Immunoglobulins/*chemistry/*pharmacology
Interferon Alfa-2a/chemistry/pharmacology
Interferon-alpha/*chemistry/*pharmacology
Mice
Mice, Nude
Neoplasms, Experimental/*drug therapy
Polyethylene Glycols/chemistry/pharmacology
Recombinant Fusion Proteins/chemistry/pharmacology
Figure
Reference
-
1. Chen SA, Sawchuk RJ, Brundage RC, Horvath C, Mendenhall HV, Gunther RA, Braeckman RA. Plasma and lymph pharmacokinetics of recombinant human interleukin-2 and polyethylene glycol-modified interleukin-2 in pigs. J Pharmacol Exp Ther. 2000. 293:248–259.2. de Luján Alvarez M, Ronco MT, Ochoa JE, Monti JA, Carnovale CE, Pisani GB, Lugano MC, Carrillo MC. Interferon α-induced apoptosis on rat preneoplastic liver is mediated by hepatocytic transforming growth factor β1. Hepatology. 2004. 40:394–402.
Article3. Fidler IJ. Regulation of neoplastic angiogenesis. J Natl Cancer Inst Monogr. 2000. 28:10–14.
Article4. Gutterman JU. Cyt okine therapeutics: lessons from interferon α. Proc Natl Acad Sci USA. 1994. 91:1198–1205.5. Lee CW, Hong DH, Han SB, Jung SH, Kim HC, Fine RL, Lee SH, Kim HM. A novel stereo-selective sulfonylurea, 1-[1-(4-aminobenzoyl)-2,3-dihydro-1H-indol-6-sulfonyl]-4-phenyl-imidazolidin-2-one, has antitumor efficacy in in vitro and in vivo tumor models. Biochem Pharmacol. 2002. 64:473–480.
Article6. Morinaga Y, Suga Y, Ehara S, Harada K, Nihei Y, Suzuki M. Combination effect of AC-7700, a novel combretastatin A-4 derivative, and cisplatin against murine and human tumors in vivo. Cancer Sci. 2003. 94:200–204.
Article7. Takaoka A, Hayakawa S, Yanai H, Stoiber D, Negishi H, Kikuchi H, Sasaki S, Imai K, Shibue T, Honda K, Taniguchi T. Integration of interferon-α/β signalling to p53 responses in tumour suppression and antiviral defence. Nature. 2003. 424:516–523.
Article8. Tsunoda S, Ishikawa T, Yamamoto Y, Kamada H, Koizumi K, Matsui J, Tsutsumi Y, Hirano T, Mayumi T. Enhanced antitumor potency of polyethylene glycolylated tumor necrosis factor-α: a novel polymer-conjugation technique with a reversible amino-protective reagent. J Pharmacol Exp Ther. 1999. 290:368–372.9. von Marschall Z, Scholz A, Cramer T, Schäfer G, Schirner M, Oberg K, Wiedenmann B, Höcker M, Rosewicz S. Effects of interferon alpha on vascular endothelial growth factor gene transcription and tumor angiogenesis. J Natl Cancer Inst. 2003. 95:437–448.
Article10. Zhang F, Lu W, Dong Z. Tumor-infiltrating macrophages are involved in suppressing growth and metastasis of human prostate cancer cells by INF-beta gene therapy in nude mice. Clin Cancer Res. 2002. 8:2942–2951.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- The Effects of Korean Traditional Medicine Mixture on Cytokine Level, Food Intake and Nutrition Metabolism of the Cachexia Induced-Mice
- The Effect of Interferon-alpha and bFGF on the Proliferation of Cultured Leiomyoma and Myometrial Cells
- Antitumor effects of recombinant human interferon-alpha and gamma on human malignant melanoma xenograft in nude mice and alteration in morphology and immunologic parameters
- Perivascular epithelioid cell tumor (PEComa) of the ascending colon: the implication of IFN-alpha2b treatment
- Anticancer activity of subfractions containing pure compounds of Chaga mushroom (Inonotus obliquus) extract in human cancer cells and in Balbc/c mice bearing Sarcoma-180 cells