J Vet Sci.
2006 Sep;7(3):233-239. 10.4142/jvs.2006.7.3.233.
Unique features of bovine lymphocytes exposed to a staphylococcal enterotoxin
- Affiliations
-
- 1Department of Microbiology, College of Veterinary Medicine, Seoul National University, Seoul 151-742, Korea.
- 2Department of Microbiology, Molecular Biology and Biochemistry, University of Idaho, Moscow, Idaho 83844, USA. gbohach@uidaho.edu
- 3Department of Veterinary Microbiology and Pathology, Washington State University, Pullman, WA 99164, USA.
- 4Department of Pathology and Microbiology, University of Nebraska Medical Center, Omaha, NE 68198, USA.
- 5Animal Health Research, LG Life Sciences Ltd., Daejeon 305-380, Korea.
- KMID: 1089907
- DOI: http://doi.org/10.4142/jvs.2006.7.3.233
Abstract
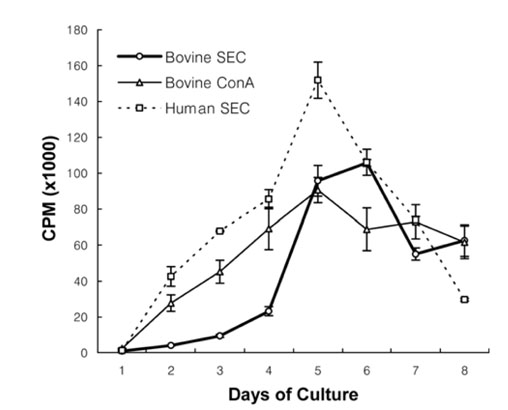
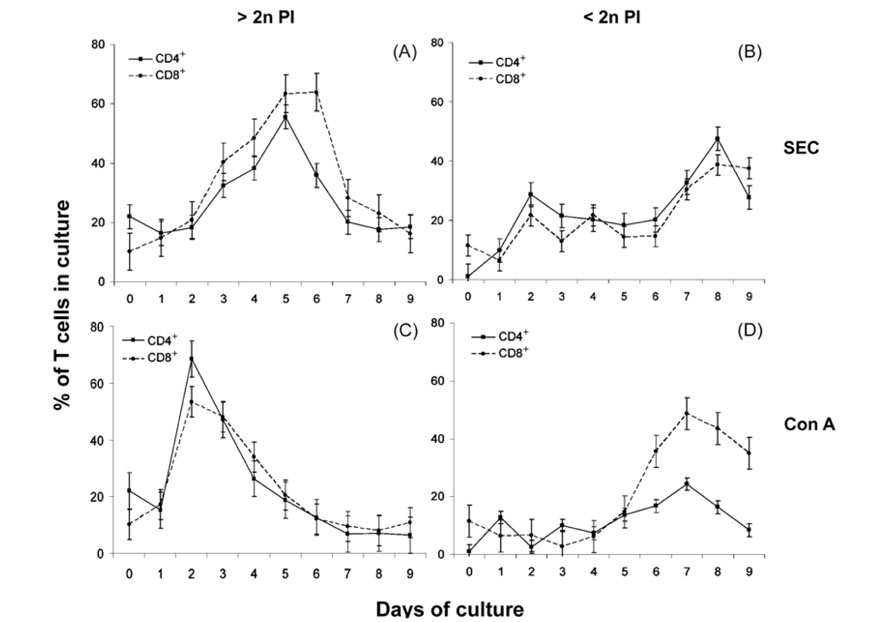
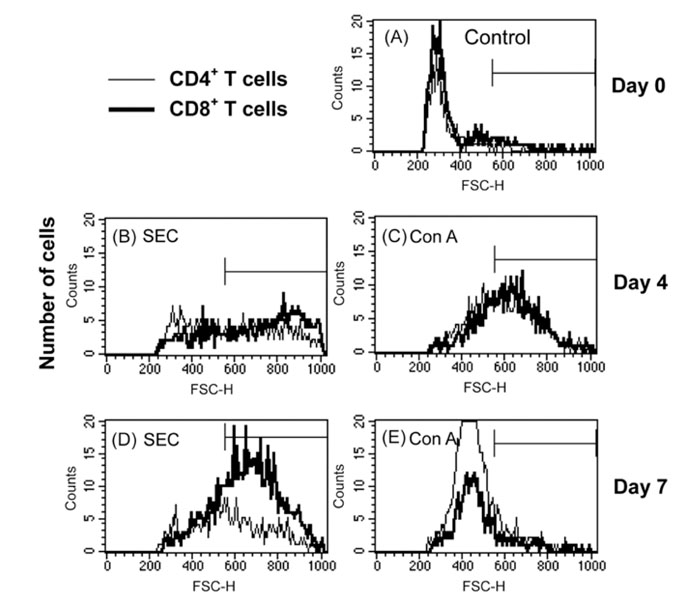
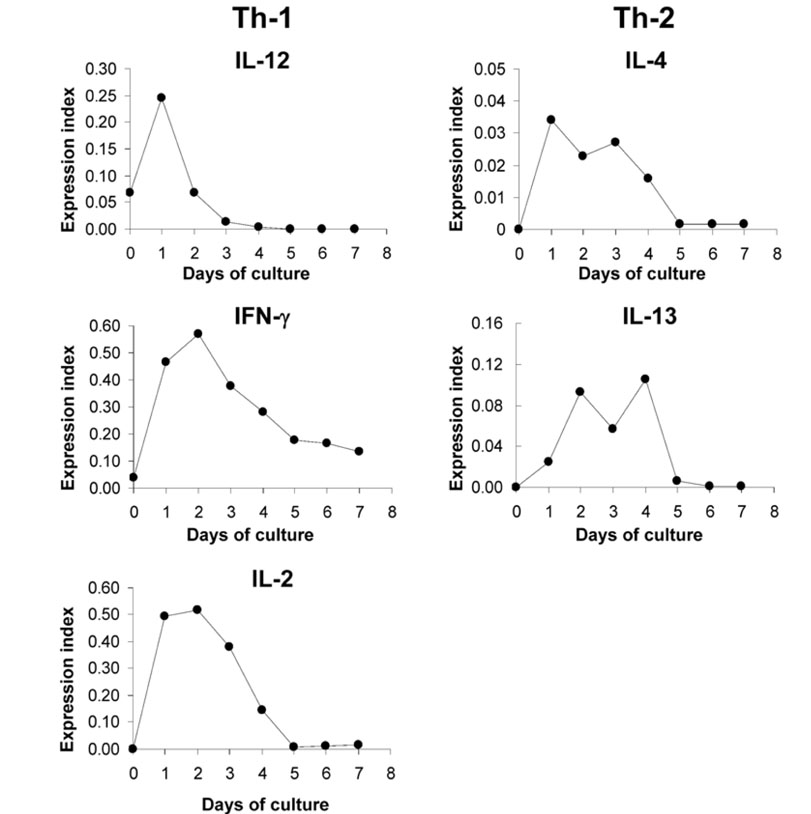
- We previously demonstrated that stimulation of bovine peripheral blood mononuclear cells (PBMCs) with staphylococcal enterotoxin C (SEC), led to an inversion of the CD4(+):CD8(+) T cell ratio and generation of an atypical CD8(+) T cell subpopulation expressing CD26. In the present study, we examined T cell apoptosis and proliferation profiles of PBMC subpopulations in cultures stimulated with SEC. Unlike when stimulated with concanavalin A, nucleic acid synthesis in bovine PBMC cultures stimulated with SEC was low during the first four days but increased greatly on day 5. In contrast, nucleic acid synthesis in human PBMC cultures stimulated with SEC increased continuously. To investigate the mechanism of delayed bovine T cell proliferation, various cell phenotypes were monitored. The inversion of the bovine CD4(+):CD8(+) T cell ratio in PBMC cultures stimulated by SEC was associated with higher proliferation and lower apoptosis of CD8(+) T cells compared to CD4(+) T cells. The mRNA levels for interleukin (IL)-4 and IL-13 were sustained over 4 days but IL-12 mRNA levels dropped to background on day 2. These data suggest that SEC induces a prolonged Th-2- biased microenvironment, and together with the inversion of the bovine CD4(+):CD8(+) T cell ratios in bovine PBMC cultures with SEC, may in part explain the inability of the mammary immune system to establish an effective response to Staphylococcus aureus infections.
Keyword
MeSH Terms
-
Animals
Apoptosis/drug effects/immunology
CD4-CD8 Ratio/veterinary
CD4-Positive T-Lymphocytes/drug effects/*immunology/microbiology
CD8-Positive T-Lymphocytes/drug effects/*immunology/microbiology
Cattle
Concanavalin A/pharmacology
Cytokines/genetics/immunology
Enterotoxins/*pharmacology
Female
Lymphocyte Activation/drug effects
Mastitis, Bovine/immunology/*microbiology
Reverse Transcriptase Polymerase Chain Reaction/veterinary
Staphylococcal Infections/immunology/microbiology/*veterinary
Staphylococcus aureus/*immunology
Figure
Reference
-
1. Almeida RA, Matthews KR, Cifrian E, Guidry AJ, Oliver SP. Staphylococcus aureus invasion of bovine mammary epithelial cells. J Dairy Sci. 1996. 79:1021–1026.2. Bayles KW, Wesson CA, Liou LE, Fox LK, Bohach GA, Trumble WR. Intracellular Staphylococcus aureus escapes the endosome and induces apoptosis in epithelial cells. Infect Immun. 1998. 66:336–342.
Article3. Blank N, Burger R, Duerr B, Bakker F, Wohlfarth A, Dumitriu I, Kalden JR, Herrmann M. MEK inhibitor U0126 interferes with immunofluorescence analysis of apoptotic cell death. Cytometry. 2002. 48:179–184.
Article4. Bohach GA, Fast DJ, Nelson RD, Schlievert PM. Staphylococcal and streptococcal pyrogenic toxins involved in toxic shock syndrome and related illnesses. Crit Rev Microbiol. 1990. 17:251–272.
Article5. Boshell M, McLeod J, Walker L, Hall N, Patel Y, Sansom D. Effects of antigen presentation on superantigen-induced apoptosis mediated by Fas/Fas ligand interactions in human T cells. Immunology. 1996. 87:586–592.
Article6. Chang BS, Bohach GA, Lee SU, Davis WC, Fox LK, Ferens WA, Seo KS, Koo HC, Kwon NH, Park YH. Immunosuppression by T regulatory cells in cows infected with Staphylococcal superantigen. J Vet Sci. 2005. 6:247–250.
Article7. Damle NK, Leytze G, Klussman K, Ledbetter JA. Activation with superantigens induces programmed death in antigen-primed CD4+ class II+ major histocompatibility complex T lymphocytes via a CD11a/CD18-dependent mechanism. Eur J Immunol. 1993. 23:1513–1522.
Article8. Dannecker G, Mahlknecht U, Schultz H, Hoffmann MK, Niethammer D. Activation of human T cells by the superantigen Staphylococcus enterotoxin B: analysis on a cellular level. Immunobiology. 1994. 190:116–126.
Article9. Davis WC, Naessens J, Brown WC, Ellis JA, Hamilton MJ, Cantor GH, Barbosa JI, Ferens W, Bohach GA. Analysis of monoclonal antibodies reactive with molecules upregulated or expressed only on activated lymphocytes. Vet Immunol Immunopathol. 1996. 52:301–311.
Article10. Deringer JR, Ely RJ, Stauffacher CV, Bohach GA. Subtype-specific interactions of type C staphylococcal enterotoxins with the T-cell receptor. Mol Microbiol. 1996. 22:523–534.
Article11. Ferens WA, Davis WC, Hamilton MJ, Park YH, Deobald CF, Fox L, Bohach G. Activation of bovine lymphocyte subpopulations by staphylococcal enterotoxin C. Infect Immun. 1998. 66:573–580.
Article12. Ferens WA, Goff WL, Davis WC, Fox LK, Deobald C, Hamilton MJ, Bohach GA. Induction of type 2 cytokines by a staphylococcal enterotoxin superantigen. J Nat Toxins. 1998. 7:193–213.13. Goff WL, Johnson WC, Parish SM, Barrington GM, Elsasser TH, Davis WC, Valdez RA. IL-4 and IL-10 inhibition of IFN-gamma- and TNF-alpha-dependent nitric oxide production from bovine mononuclear phagocytes exposed to Babesia bovis merozoites. Vet Immunol Immunopathol. 2002. 84:237–251.
Article14. Hoffmann ML, Jablonski LM, Crum KK, Hackett SP, Chi YI, Stauffacher CV, Stevens DL, Bohach GA. Predictions of T-cell receptor- and major histocompatibility complex-binding sites on staphylococcal enterotoxin C1. Infect Immun. 1994. 62:3396–3407.
Article15. Iandolo JJ. Iglewski BH, Clark VL, editors. The genetics of staphylococcal enterotoxins and virulence factors. Molecular Basis of Bacterial Pathogenesis. 1990. San Diego: Academic Press;399–426.16. Kawabe Y, Ochi A. Selective anergy of V beta 8+, CD4+ T cells in Staphylococcus enterotoxin B-primed mice. J Exp Med. 1990. 172:1065–1070.
Article17. Kenny K, Reiser RF, Bastida-Corcuera FD, Norcross NL. Production of enterotoxins and toxic shock syndrome toxin by bovine mammary isolates of Staphylococcus aureus. J Clin Microbiol. 1993. 31:706–707.
Article18. Lee SU, Ferens W, Davis WC, Hamilton MJ, Park YH, Fox LK, Naessens J, Bohach GA. Identity of activation molecule 3 on superantigen-stimulated bovine cells is CD26. Infect Immun. 2001. 69:7190–7193.
Article19. MacDonald HR, Baschieri S, Lees RK. Clonal expansion precedes anergy and death of V beta 8+ peripheral T cells responding to staphylococcal enterotoxin B in vivo. Eur J Immunol. 1991. 21:1963–1966.
Article20. Marr JC, Lyon JD, Roberson JR, Lupher M, Davis WC, Bohach GA. Characterization of novel type C staphylococcal enterotoxins: biological and evolutionary implications. Infect Immun. 1993. 61:4254–4262.
Article21. Mercado R, Vijh S, Allen SE, Kerksiek K, Pilip IM, Pamer EG. Early programming of T cell populations responding to bacterial infection. J Immunol. 2000. 165:6833–6839.
Article22. Modlin RL, Mehra V, Wong L, Fujimiya Y, Chang WC, Horwitz DA, Bloom BR, Rea TH, Pattengale PK. Suppressor T lymphocytes from lepromatous leprosy skin lesions. J Immunol. 1986. 137:2831–2834.23. Noble A, Pestano GA, Cantor H. Suppression of immune responses by CD8 cells. I. Superantigen-activated CD8 cells induce unidirectional Fas-mediated apoptosis of antigen-activated CD4 cells. J Immunol. 1998. 160:559–565.24. Park YH, Fox LK, Hamilton MJ, Davis WC. Bovine mononuclear leukocyte subpopulations in peripheral blood and mammary gland secretions during lactation. J Dairy Sci. 1992. 75:998–1006.
Article25. Park YH, Fox LK, Hamilton MJ, Davis WC. Suppression of proliferative response of BoCD4+ T lymphocytes by activated BoCD8+ T lymphocytes in the mammary gland of cows with Staphylococcus aureus mastitis. Vet Immunol Immunopathol. 1993. 36:137–151.
Article26. Poindexter NJ, Schlievert PM. Toxic-shock-syndrome toxin 1-induced proliferation of lymphocytes: comparison of the mitogenic response of human, murine, and rabbit lymphocytes. J Infect Dis. 1985. 151:65–72.
Article27. Renno T, Attinger A, Locatelli S, Bakker T, Vacheron S, MacDonald HR. Cutting edge: apoptosis of superantigen-activated T cells occurs preferentially after a discrete number of cell divisions in vivo. J Immunol. 1999. 162:6312–6315.28. Sutra L, Poutrel B. Virulence factors involved in the pathogenesis of bovine intramammary infections due to Staphylococcus aureus. J Med Microbiol. 1994. 40:79–89.
Article29. Taylor BC, Dellinger JD, Cullor JS, Stott JL. Bovine milk lymphocytes display the phenotype of memory T cells and are predominantly CD8+. Cell Immunol. 1994. 156:245–253.
Article30. Tollersrud T, Kenny K, Caugant DA, Lund A. Characterisation of isolates of Staphylococcus aureus from acute, chronic and subclinical mastitis in cows in Norway. APMIS. 2000. 108:565–572.
Article31. Webb SR, Gascoigne NR. T-cell activation by superantigens. Curr Opin Immunol. 1994. 6:467–475.
Article32. Yamamura M, Uyemura K, Deans RJ, Weinberg K, Rea TH, Bloom BR, Modlin RL. Defining protective responses to pathogens: cytokine profiles in leprosy lesions. Science. 1991. 254:277–279.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- spa typing and enterotoxin gene profile of Staphylococcus aureus isolated from bovine raw milk in Korea
- Staphylococcal enterotoxin specific IgE and asthma: a systematic review and meta-analysis
- Increase of Rhinovirus Replication in Airway Epithelial Cells by Staphylococcal Enterotoxin A and B
- Effects of Enterotoxins of Staphylococcus Aureus on Ciliary Activity of the Nasal Mucosa
- Effect of Staphylococcal Enterotoxin on the Development of Allergic Rhinitis in Mice