Endocrinol Metab.
2023 Oct;38(5):482-492. 10.3803/EnM.2023.1776.
The Impact of Taurine on Obesity-Induced Diabetes Mellitus: Mechanisms Underlying Its Effect
- Affiliations
-
- 1Interdisciplinary Program in Senior Human Ecology, Changwon National University, Changwon, Korea
- 2Department of Food and Nutrition, Changwon National University, Changwon, Korea
- KMID: 2546979
- DOI: http://doi.org/10.3803/EnM.2023.1776
Abstract
- This review explores the potential benefits of taurine in ameliorating the metabolic disorders of obesity and type 2 diabetes (T2D), highlighting the factors that bridge these associations. Relevant articles and studies were reviewed to conduct a comprehensive analysis of the relationship between obesity and the development of T2D and the effect of taurine on those conditions. The loss of normal β-cell function and development of T2D are associated with obesity-derived insulin resistance. The occurrence of diabetes has been linked to the low bioavailability of taurine, which plays critical roles in normal β-cell function, anti-oxidation, and anti-inflammation. The relationships among obesity, insulin resistance, β-cell dysfunction, and T2D are complex and intertwined. Taurine may play a role in ameliorating these metabolic disorders through different pathways, but further research is needed to fully understand its effects and potential as a therapeutic intervention.
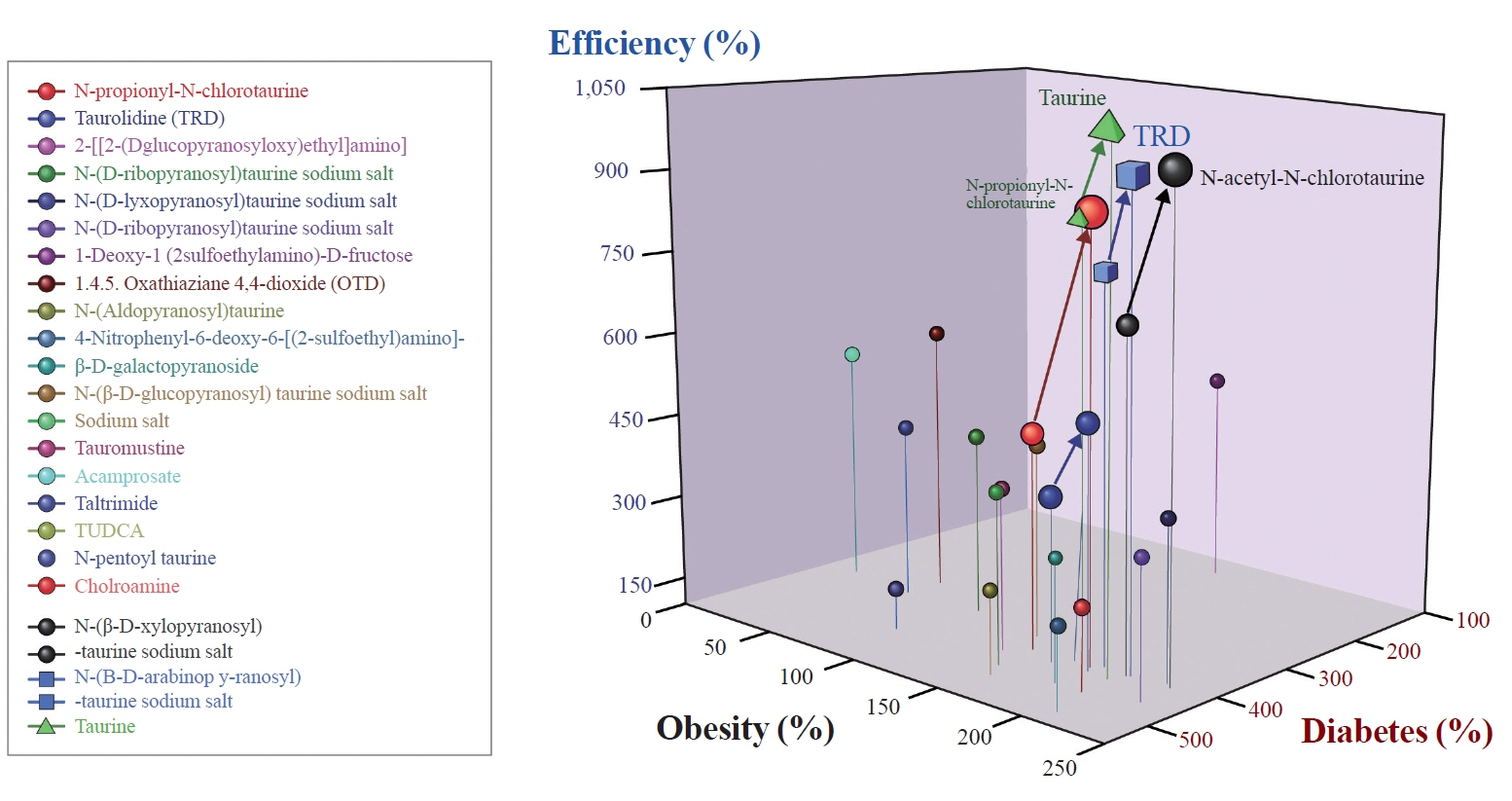
Figure
Reference
-
1. Chooi YC, Ding C, Magkos F. The epidemiology of obesity. Metabolism. 2019; 92:6–10.
Article2. Kaveeshwar SA, Cornwall J. The current state of diabetes mellitus in India. Australas Med J. 2014; 7:45–8.
Article3. Kahn SE, Hull RL, Utzschneider KM. Mechanisms linking obesity to insulin resistance and type 2 diabetes. Nature. 2006; 444:840–6.
Article4. Xiao C, Giacca A, Lewis GF. Sodium phenylbutyrate, a drug with known capacity to reduce endoplasmic reticulum stress, partially alleviates lipid-induced insulin resistance and beta-cell dysfunction in humans. Diabetes. 2011; 60:918–24.5. Uyanga VA, Oke EO, Amevor FK, Zhao J, Wang X, Jiao H, et al. Functional roles of taurine, L-theanine, L-citrulline, and betaine during heat stress in poultry. J Anim Sci Biotechnol. 2022; 13:23.
Article6. Jakaria M, Azam S, Haque ME, Jo SH, Uddin MS, Kim IS, et al. Taurine and its analogs in neurological disorders: focus on therapeutic potential and molecular mechanisms. Redox Biol. 2019; 24:101223.
Article7. Sheri FH, Dawood H, Hassan JK, Aljazaeari QA. Study the effects of taurine oral supplement used for type 2 diabetic patients on body weight; glycemic control and some bone mineralization biochemical markers. Bull Fac Pharm Cairo Univ. 2021; 59:27–32.
Article8. Tenner TE Jr, Zhang XJ, Lombardini JB. Hypoglycemic effects of taurine in the alloxan-treated rabbit, a model for type 1 diabetes. Adv Exp Med Biol. 2003; 526:97–104.
Article9. Brons C, Spohr C, Storgaard H, Dyerberg J, Vaag A. Effect of taurine treatment on insulin secretion and action, and on serum lipid levels in overweight men with a genetic predisposition for type II diabetes mellitus. Eur J Clin Nutr. 2004; 58:1239–47.
Article10. Imae M, Asano T, Murakami S. Potential role of taurine in the prevention of diabetes and metabolic syndrome. Amino Acids. 2014; 46:81–8.
Article11. Hansen SH. The role of taurine in diabetes and the development of diabetic complications. Diabetes Metab Res Rev. 2001; 17:330–46.
Article12. Chen W, Guo J, Zhang Y, Zhang J. The beneficial effects of taurine in preventing metabolic syndrome. Food Funct. 2016; 7:1849–63.
Article13. Li M, Reynolds CM, Sloboda DM, Gray C, Vickers MH. Effects of taurine supplementation on hepatic markers of inflammation and lipid metabolism in mothers and offspring in the setting of maternal obesity. PLoS One. 2013; 8:e76961.
Article14. Vernon RG. Lipid metabolism during lactation: a review of adipose tissue-liver interactions and the development of fatty liver. J Dairy Res. 2005; 72:460–9.
Article15. Abranches MV, Oliveira FC, Conceicao LL, Peluzio MD. Obesity and diabetes: the link between adipose tissue dysfunction and glucose homeostasis. Nutr Res Rev. 2015; 28:121–32.
Article16. Badoiu SC, Miricescu D, Stanescu-Spinu II, Ripszky Totan A, Badoiu SE, Costagliola M, et al. Glucose metabolism in burns: what happens? Int J Mol Sci. 2021; 22:5159.17. Yoon MJ, Lee GY, Chung JJ, Ahn YH, Hong SH, Kim JB. Adiponectin increases fatty acid oxidation in skeletal muscle cells by sequential activation of AMP-activated protein kinase, p38 mitogen-activated protein kinase, and peroxisome proliferator-activated receptor alpha. Diabetes. 2006; 55:2562–70.18. Ni Y, Ni L, Zhuge F, Xu L, Fu Z, Ota T. Adipose tissue macrophage phenotypes and characteristics: the key to insulin resistance in obesity and metabolic disorders. Obesity (Silver Spring). 2020; 28:225–34.
Article19. Mendez-Sanchez N, Valencia-Rodriguez A, Coronel-Castillo C, Vera-Barajas A, Contreras-Carmona J, Ponciano-Rodriguez G, et al. The cellular pathways of liver fibrosis in non-alcoholic steatohepatitis. Ann Transl Med. 2020; 8:400.
Article20. Li M, Chi X, Wang Y, Setrerrahmane S, Xie W, Xu H. Trends in insulin resistance: insights into mechanisms and therapeutic strategy. Signal Transduct Target Ther. 2022; 7:216.
Article21. Suba K. Longitudinal imaging of pancreatic islets transplanted into the anterior chamber of the eye [dissertation]. London: Imperial College London;2020.22. van Vliet S, Koh HE, Patterson BW, Yoshino M, LaForest R, Gropler RJ, et al. Obesity is associated with increased basal and postprandial β-cell insulin secretion even in the absence of insulin resistance. Diabetes. 2020; 69:2112–9.
Article23. Halberg N, Wernstedt-Asterholm I, Scherer PE. The adipocyte as an endocrine cell. Endocrinol Metab Clin North Am. 2008; 37:753–68.
Article24. Engin A. Adiponectin-resistance in obesity. Adv Exp Med Biol. 2017; 960:415–41.
Article25. Abdul-Ghani MA, Tripathy D, DeFronzo RA. Contributions of beta-cell dysfunction and insulin resistance to the pathogenesis of impaired glucose tolerance and impaired fasting glucose. Diabetes Care. 2006; 29:1130–9.26. Eizirik DL, Pasquali L, Cnop M. Pancreatic β-cells in type 1 and type 2 diabetes mellitus: different pathways to failure. Nat Rev Endocrinol. 2020; 16:349–62.
Article27. Cho JH, Kim JW, Shin JA, Shin J, Yoon KH. β-Cell mass in people with type 2 diabetes. J Diabetes Investig. 2011; 2:6–17.28. Devedjian JC, George M, Casellas A, Pujol A, Visa J, Pelegrin M, et al. Transgenic mice overexpressing insulin-like growth factor-II in beta cells develop type 2 diabetes. J Clin Invest. 2000; 105:731–40.29. Petersen MC, Shulman GI. Mechanisms of insulin action and insulin resistance. Physiol Rev. 2018; 98:2133–223.
Article30. da Silva Rosa SC, Nayak N, Caymo AM, Gordon JW. Mechanisms of muscle insulin resistance and the cross-talk with liver and adipose tissue. Physiol Rep. 2020; 8:e14607.
Article31. Bovolini A, Garcia J, Andrade MA, Duarte JA. Metabolic syndrome pathophysiology and predisposing factors. Int J Sports Med. 2021; 42:199–214.
Article32. Kim KS, Jang MJ, Fang S, Yoon SG, Kim IY, Seong JK, et al. Anti-obesity effect of taurine through inhibition of adipogenesis in white fat tissue but not in brown fat tissue in a high-fat diet-induced obese mouse model. Amino Acids. 2019; 51:245–54.
Article33. Wen C, Li F, Zhang L, Duan Y, Guo Q, Wang W, et al. Taurine is involved in energy metabolism in muscles, adipose tissue, and the liver. Mol Nutr Food Res. 2019; 63:e1800536.
Article34. Guo YY, Li BY, Peng WQ, Guo L, Tang QQ. Taurine-mediated browning of white adipose tissue is involved in its antiobesity effect in mice. J Biol Chem. 2019; 294:15014–24.
Article35. Caetano LC, Bonfleur ML, Ribeiro RA, Nardelli TR, Lubaczeuski C, do Nascimento da Silva J, et al. Taurine supplementation regulates Iκ-Bα protein expression in adipose tissue and serum IL-4 and TNF-α concentrations in MSG obesity. Eur J Nutr. 2017; 56:705–13.
Article36. Lin S, Hirai S, Yamaguchi Y, Goto T, Takahashi N, Tani F, et al. Taurine improves obesity-induced inflammatory responses and modulates the unbalanced phenotype of adipose tissue macrophages. Mol Nutr Food Res. 2013; 57:2155–65.
Article37. Leroux M, Lemery T, Boulet N, Briot A, Zakaroff A, Bouloumie A, et al. Effects of the amino acid derivatives, βhydroxy-β-methylbutyrate, taurine, and N-methyltyramine, on triacylglycerol breakdown in fat cells. J Physiol Biochem. 2019; 75:263–73.
Article38. Batista TM, Ribeiro RA, da Silva PM, Camargo RL, Lollo PC, Boschero AC, et al. Taurine supplementation improves liver glucose control in normal protein and malnourished mice fed a high-fat diet. Mol Nutr Food Res. 2013; 57:423–34.
Article39. Rafiee Z, Garcia-Serrano AM, Duarte JM. Taurine supplementation as a neuroprotective strategy upon brain dysfunction in metabolic syndrome and diabetes. Nutrients. 2022; 14:1292.
Article40. Bae M, Ahmed K, Yim JE. Beneficial effects of taurine on metabolic parameters in animals and humans. J Obes Metab Syndr. 2022; 31:134–46.
Article41. Saad SY, Al-Rikabi AC. Protection effects of taurine supplementation against cisplatin-induced nephrotoxicity in rats. Chemotherapy. 2002; 48:42–8.
Article42. Cappelli AP, Zoppi CC, Barbosa-Sampaio HC, Costa JM Jr, Protzek AO, Morato PN, et al. Taurine-induced insulin signalling improvement of obese malnourished mice is associated with redox balance and protein phosphatases activity modulation. Liver Int. 2014; 34:771–83.
Article43. Larsen LH, Orstrup LK, Hansen SH, Grunnet N, Quistorff B, Mortensen OH. The effect of long-term taurine supplementation and fructose feeding on glucose and lipid homeostasis in Wistar rats. Adv Exp Med Biol. 2013; 776:39–50.
Article44. Qiu T, Pei P, Yao X, Jiang L, Wei S, Wang Z, et al. Taurine attenuates arsenic-induced pyroptosis and nonalcoholic steatohepatitis by inhibiting the autophagic-inflammasomal pathway. Cell Death Dis. 2018; 9:946.
Article45. Carneiro EM, Latorraca MQ, Araujo E, Beltra M, Oliveras MJ, Navarro M, et al. Taurine supplementation modulates glucose homeostasis and islet function. J Nutr Biochem. 2009; 20:503–11.
Article46. Han J, Bae JH, Kim SY, Lee HY, Jang BC, Lee IK, et al. Taurine increases glucose sensitivity of UCP2-overexpressing beta-cells by ameliorating mitochondrial metabolism. Am J Physiol Endocrinol Metab. 2004; 287:E1008–18.47. Wu G, San J, Pang H, Du Y, Li W, Zhou X, et al. Taurine attenuates AFB1-induced liver injury by alleviating oxidative stress and regulating mitochondria-mediated apoptosis. Toxicon. 2022; 215:17–27.
Article48. Yang J, Zong X, Wu G, Lin S, Feng Y, Hu J. Taurine increases testicular function in aged rats by inhibiting oxidative stress and apoptosis. Amino Acids. 2015; 47:1549–58.
Article49. Roy A, Sil PC. Tertiary butyl hydroperoxide induced oxidative damage in mice erythrocytes: protection by taurine. Pathophysiology. 2012; 19:137–48.
Article50. Jong CJ, Azuma J, Schaffer SW. Role of mitochondrial permeability transition in taurine deficiency-induced apoptosis. Exp Clin Cardiol. 2011; 16:125–8.51. Wu N, Lu Y, He B, Zhang Y, Lin J, Zhao S, et al. Taurine prevents free fatty acid-induced hepatic insulin resistance in association with inhibiting JNK1 activation and improving insulin signaling in vivo. Diabetes Res Clin Pract. 2010; 90:288–96.
Article52. Kulakowski EC, Maturo J. Hypoglycemic properties of taurine: not mediated by enhanced insulin release. Biochem Pharmacol. 1984; 33:2835–8.
Article53. Park EJ, Bae JH, Kim SY, Lim JG, Baek WK, Kwon TK, et al. Inhibition of ATP-sensitive K+ channels by taurine through a benzamido-binding site on sulfonylurea receptor 1. Biochem Pharmacol. 2004; 67:1089–96.
Article54. Jong CJ, Azuma J, Schaffer S. Mechanism underlying the antioxidant activity of taurine: prevention of mitochondrial oxidant production. Amino Acids. 2012; 42:2223–32.
Article55. Park E, Quinn MR, Wright CE, Schuller-Levis G. Taurine chloramine inhibits the synthesis of nitric oxide and the release of tumor necrosis factor in activated RAW 264.7 cells. J Leukoc Biol. 1993; 54:119–24.
Article56. Liu Y, Quinn MR. Chemokine production by rat alveolar macrophages is inhibited by taurine chloramine. Immunol Lett. 2002; 80:27–32.
Article57. Yildirim Z, Kilic N, Ozer C, Babul A, Take G, Erdogan D. Effects of taurine in cellular responses to oxidative stress in young and middle-aged rat liver. Ann N Y Acad Sci. 2007; 1100:553–61.
Article58. Vohra BP, Hui X. Taurine protects against carbon tetrachloride toxicity in the cultured neurons and in vivo. Arch Physiol Biochem. 2001; 109:90–4.
Article59. Kim C, Cha YN. Taurine chloramine produced from taurine under inflammation provides anti-inflammatory and cytoprotective effects. Amino Acids. 2014; 46:89–100.
Article60. Schaffer SW, Shimada-Takaura K, Jong CJ, Ito T, Takahashi K. Impaired energy metabolism of the taurine-deficient heart. Amino Acids. 2016; 48:549–58.
Article61. L’Amoreaux WJ, Cuttitta C, Santora A, Blaize JF, Tachjadi J, El Idrissi A. Taurine regulates insulin release from pancreatic beta cell lines. J Biomed Sci. 2010; 17(Suppl 1):S11.
Article62. Khodabandehloo H, Gorgani-Firuzjaee S, Panahi G, Meshkani R. Molecular and cellular mechanisms linking inflammation to insulin resistance and β-cell dysfunction. Transl Res. 2016; 167:228–56.
Article63. Murakami S. The physiological and pathophysiological roles of taurine in adipose tissue in relation to obesity. Life Sci. 2017; 186:80–6.
Article64. Li M, Reynolds CM, Sloboda DM, Gray C, Vickers MH. Maternal taurine supplementation attenuates maternal fructose-induced metabolic and inflammatory dysregulation and partially reverses adverse metabolic programming in offspring. J Nutr Biochem. 2015; 26:267–76.
Article65. Yao HT, Lin P, Chang YW, Chen CT, Chiang MT, Chang L, et al. Effect of taurine supplementation on cytochrome P450 2E1 and oxidative stress in the liver and kidneys of rats with streptozotocin-induced diabetes. Food Chem Toxicol. 2009; 47:1703–9.
Article66. Oprescu AI, Bikopoulos G, Naassan A, Allister EM, Tang C, Park E, et al. Free fatty acid-induced reduction in glucosestimulated insulin secretion: evidence for a role of oxidative stress in vitro and in vivo. Diabetes. 2007; 56:2927–37.67. Ribeiro RA, Vanzela EC, Oliveira CA, Bonfleur ML, Boschero AC, Carneiro EM. Taurine supplementation: involvement of cholinergic/phospholipase C and protein kinase A pathways in potentiation of insulin secretion and Ca2+ handling in mouse pancreatic islets. Br J Nutr. 2010; 104:1148–55.
Article68. Ribeiro RA, Bonfleur ML, Amaral AG, Vanzela EC, Rocco SA, Boschero AC, et al. Taurine supplementation enhances nutrient-induced insulin secretion in pancreatic mice islets. Diabetes Metab Res Rev. 2009; 25:370–9.
Article69. Haber CA, Lam TK, Yu Z, Gupta N, Goh T, Bogdanovic E, et al. N-acetylcysteine and taurine prevent hyperglycemia-induced insulin resistance in vivo: possible role of oxidative stress. Am J Physiol Endocrinol Metab. 2003; 285:E744–53.