Intest Res.
2023 Jul;21(3):283-294. 10.5217/ir.2023.00020.
Artificial intelligence in inflammatory bowel disease: implications for clinical practice and future directions
- Affiliations
-
- 1Bristol Myers Squibb, Princeton, NJ, USA
- 2Translational Gastroenterology Unit, Oxford NIHR Biomedical Research Centre, University of Oxford, Oxford, UK
- 3Inflammatory Bowel Disease Clinic, University of Calgary, Calgary, AB, Canada
- 4Division of Gastroenterology, Department of Medicine, University of British Columbia, Vancouver, BC, Canada
- 5Satisfai Health, Vancouver, BC, Canada
- KMID: 2544752
- DOI: http://doi.org/10.5217/ir.2023.00020
Abstract
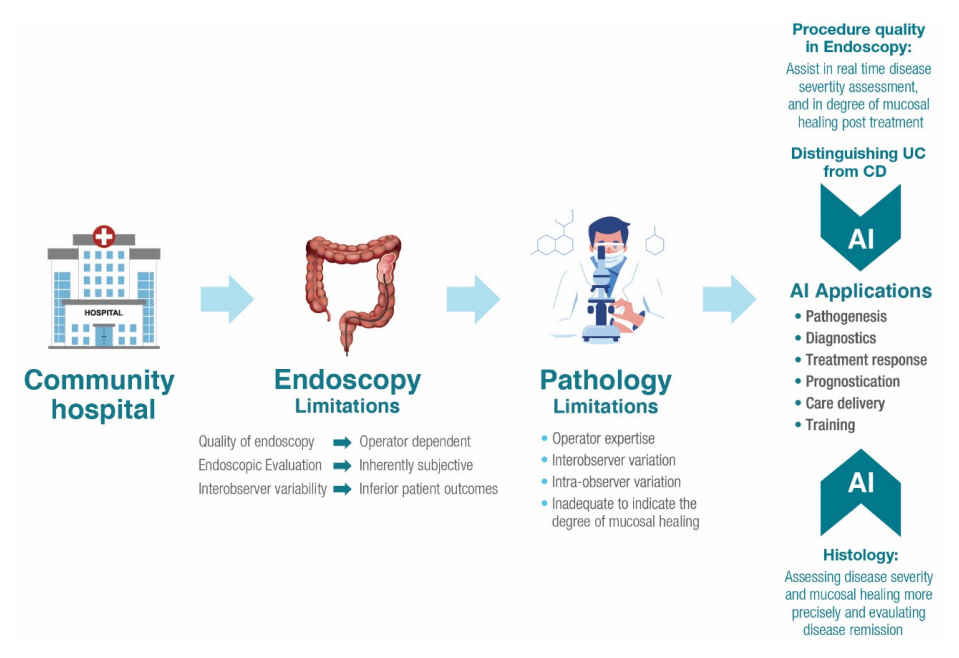
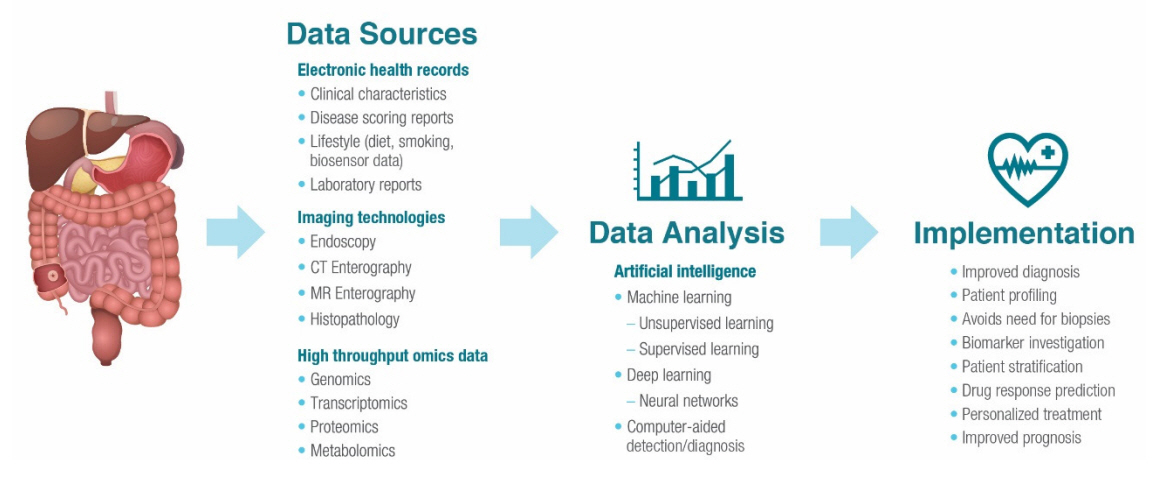
- Inflammatory bowel disease encompasses Crohn’s disease and ulcerative colitis and is characterized by uncontrolled, relapsing, and remitting course of inflammation in the gastrointestinal tract. Artificial intelligence represents a new era within the field of gastroenterology, and the amount of research surrounding artificial intelligence in patients with inflammatory bowel disease is on the rise. As clinical trial outcomes and treatment targets evolve in inflammatory bowel disease, artificial intelligence may prove as a valuable tool for providing accurate, consistent, and reproducible evaluations of endoscopic appearance and histologic activity, thereby optimizing the diagnosis process and identifying disease severity. Furthermore, as the applications of artificial intelligence for inflammatory bowel disease continue to expand, they may present an ideal opportunity for improving disease management by predicting treatment response to biologic therapies and for refining the standard of care by setting the basis for future treatment personalization and cost reduction. The purpose of this review is to provide an overview of the unmet needs in the management of inflammatory bowel disease in clinical practice and how artificial intelligence tools can address these gaps to transform patient care.
Figure
Reference
-
1. GBD 2017 Inflammatory Bowel Disease Collaborators. The global, regional, and national burden of inflammatory bowel disease in 195 countries and territories, 1990-2017: a systematic analysis for the Global Burden of Disease Study 2017. Lancet Gastroenterol Hepatol. 2020; 5:17–30.2. Maaser C, Sturm A, Vavricka SR, et al. ECCO-ESGAR Guideline for Diagnostic Assessment in IBD Part 1: initial diagnosis, monitoring of known IBD, detection of complications. J Crohns Colitis. 2019; 13:144–164.
Article3. Cohen-Mekelburg S, Berry S, Stidham RW, Zhu J, Waljee AK. Clinical applications of artificial intelligence and machine learning-based methods in inflammatory bowel disease. J Gastroenterol Hepatol. 2021; 36:279–285.
Article4. Gajendran M, Loganathan P, Catinella AP, Hashash JG. A comprehensive review and update on Crohn’s disease. Dis Mon. 2018; 64:20–57.
Article5. Gajendran M, Loganathan P, Jimenez G, et al. A comprehensive review and update on ulcerative colitis. Dis Mon. 2019; 65:100851.
Article6. Dal Buono A, Roda G, Argollo M, Peyrin-Biroulet L, Danese S. Histological healing: should it be considered as a new outcome for ulcerative colitis? Expert Opin Biol Ther. 2020; 20:407–412.
Article7. Turner D, Ricciuto A, Lewis A, et al. STRIDE-II: an update on the selecting therapeutic targets in inflammatory bowel disease (STRIDE) initiative of the International Organization for the Study of IBD (IOIBD): determining therapeutic goals for treat-to-target strategies in IBD. Gastroenterology. 2021; 160:1570–1583.
Article8. Schett G, McInnes IB, Neurath MF. Reframing immune-mediated inflammatory diseases through signature cytokine hubs. N Engl J Med. 2021; 385:628–639.
Article9. Le Berre C, Sandborn WJ, Aridhi S, et al. Application of artificial intelligence to gastroenterology and hepatology. Gastroenterology. 2020; 158:76–94.
Article10. Seyed Tabib NS, Madgwick M, Sudhakar P, Verstockt B, Korcsmaros T, Vermeire S. Big data in IBD: big progress for clinical practice. Gut. 2020; 69:1520–1532.
Article11. Pannala R, Krishnan K, Melson J, et al. Artificial intelligence in gastrointestinal endoscopy. VideoGIE. 2020; 5:598–613.
Article12. Chahal D, Byrne MF. A primer on artificial intelligence and its application to endoscopy. Gastrointest Endosc. 2020; 92:813–820.
Article13. Sinonquel P, Eelbode T, Bossuyt P, Maes F, Bisschops R. Artificial intelligence and its impact on quality improvement in upper and lower gastrointestinal endoscopy. Dig Endosc. 2021; 33:242–253.
Article14. Fernandes SR, Pinto JS, Marques da Costa P, Correia L; GEDII. Disagreement among gastroenterologists using the Mayo and Rutgeerts Endoscopic Scores. Inflamm Bowel Dis. 2018; 24:254–260.
Article15. Stidham R, Yao H, Soroushmehr R, et al. 796 Computer vision measurement of disease severity distribution outperforms traditional endoscopic scoring for detecting therapeutic response in a clinical trial of ustekinumab for ulcerative colitis. Gastroenterology. 2022; 162:S–193.
Article16. Maeda Y, Kudo SE, Mori Y, et al. Fully automated diagnostic system with artificial intelligence using endocytoscopy to identify the presence of histologic inflammation associated with ulcerative colitis (with video). Gastrointest Endosc. 2019; 89:408–415.
Article17. Kiesslich R, Duckworth CA, Moussata D, et al. Local barrier dysfunction identified by confocal laser endomicroscopy predicts relapse in inflammatory bowel disease. Gut. 2012; 61:1146–1153.
Article18. Vespa E, D’Amico F, Sollai M, et al. Histological scores in patients with inflammatory bowel diseases: the state of the art. J Clin Med. 2022; 11:939.
Article19. Berzin TM, Parasa S, Wallace MB, Gross SA, Repici A, Sharma P. Position statement on priorities for artificial intelligence in GI endoscopy: a report by the ASGE Task Force. Gastrointest Endosc. 2020; 92:951–959.
Article20. Dekker E, Nass KJ, Iacucci M, et al. Performance measures for colonoscopy in inflammatory bowel disease patients: European Society of Gastrointestinal Endoscopy (ESGE) Quality Improvement Initiative. Endoscopy. 2022; 54:904–915.
Article21. Wu L, Zhang J, Zhou W, et al. Randomised controlled trial of WISENSE, a real-time quality improving system for monitoring blind spots during esophagogastroduodenoscopy. Gut. 2019; 68:2161–2169.
Article22. Freedman D, Blau Y, Katzir L, et al. Detecting deficient coverage in colonoscopies. IEEE Trans Med Imaging. 2020; 39:3451–3462.
Article23. Zhou J, Wu L, Wan X, et al. A novel artificial intelligence system for the assessment of bowel preparation (with video). Gastrointest Endosc. 2020; 91:428–435.
Article24. Thakkar S, Carleton NM, Rao B, Syed A. Use of artificial intelligence-based analytics from live colonoscopies to optimize the quality of the colonoscopy examination in real time: proof of concept. Gastroenterology. 2020; 158:1219–1221.
Article25. Ali S, Zhou F, Bailey A, et al. A deep learning framework for quality assessment and restoration in video endoscopy. Med Image Anal. 2021; 68:101900.
Article26. Smolander J, Dehmer M, Emmert-Streib F. Comparing deep belief networks with support vector machines for classifying gene expression data from complex disorders. FEBS Open Bio. 2019; 9:1232–1248.
Article27. Bielecki C, Bocklitz TW, Schmitt M, et al. Classification of inflammatory bowel diseases by means of Raman spectroscopic imaging of epithelium cells. J Biomed Opt. 2012; 17:076030.
Article28. Tong Y, Lu K, Yang Y, et al. Can natural language processing help differentiate inflammatory intestinal diseases in China? Models applying random forest and convolutional neural network approaches. BMC Med Inform Decis Mak. 2020; 20:248.
Article29. Majtner T, Brodersen JB, Herp J, Kjeldsen J, Halling ML, Jensen MD. A deep learning framework for autonomous detection and classification of Crohn’s disease lesions in the small bowel and colon with capsule endoscopy. Endosc Int Open. 2021; 9:E1361–E1370.
Article30. Huang TY, Zhan SQ, Chen PJ, Yang CW, Lu HH. Accurate diagnosis of endoscopic mucosal healing in ulcerative colitis using deep learning and machine learning. J Chin Med Assoc. 2021; 84:678–681.
Article31. Yao H, Najarian K, Gryak J, et al. Fully automated endoscopic disease activity assessment in ulcerative colitis. Gastrointest Endosc. 2021; 93:728–736.
Article32. Gutierrez Becker B, Arcadu F, Thalhammer A, et al. Training and deploying a deep learning model for endoscopic severity grading in ulcerative colitis using multicenter clinical trial data. Ther Adv Gastrointest Endosc. 2021; 14:2631774521990623.
Article33. Takenaka K, Ohtsuka K, Fujii T, et al. Development and validation of a deep neural network for accurate evaluation of endoscopic images from patients with ulcerative colitis. Gastroenterology. 2020; 158:2150–2157.
Article34. Bossuyt P, Nakase H, Vermeire S, et al. Automatic, computer-aided determination of endoscopic and histological inflammation in patients with mild to moderate ulcerative colitis based on red density. Gut. 2020; 69:1778–1786.
Article35. Waljee AK, Liu B, Sauder K, et al. Predicting corticosteroid-free endoscopic remission with vedolizumab in ulcerative colitis. Aliment Pharmacol Ther. 2018; 47:763–772.
Article36. Miyoshi J, Maeda T, Matsuoka K, et al. Machine learning using clinical data at baseline predicts the efficacy of vedolizumab at week 22 in patients with ulcerative colitis. Sci Rep. 2021; 11:16440.
Article37. Waljee AK, Wallace BI, Cohen-Mekelburg S, et al. Development and validation of machine learning models in prediction of remission in patients with moderate to severe Crohn disease. JAMA Netw Open. 2019; 2:e193721.
Article38. Waljee AK, Sauder K, Patel A, et al. Machine learning algorithms for objective remission and clinical outcomes with thiopurines. J Crohns Colitis. 2017; 11:801–810.
Article39. Takayama T, Okamoto S, Hisamatsu T, et al. Computer-aided prediction of long-term prognosis of patients with ulcerative colitis after cytoapheresis therapy. PLoS One. 2015; 10:e0131197.
Article40. Atreya R, Neumann H, Neufert C, et al. In vivo imaging using fluorescent antibodies to tumor necrosis factor predicts therapeutic response in Crohn’s disease. Nat Med. 2014; 20:313–318.
Article41. Guidi L, Marzo M, Andrisani G, et al. Faecal calprotectin assay after induction with anti-tumour necrosis factor α agents in inflammatory bowel disease: prediction of clinical response and mucosal healing at one year. Dig Liver Dis. 2014; 46:974–979.
Article42. Popa IV, Burlacu A, Mihai C, Prelipcean CC. A machine learning model accurately predicts ulcerative colitis activity at one year in patients treated with anti-tumour necrosis factor α agents. Medicina (Kaunas). 2020; 56:628.
Article43. Olivera P, Danese S, Jay N, Natoli G, Peyrin-Biroulet L. Big data in IBD: a look into the future. Nat Rev Gastroenterol Hepatol. 2019; 16:312–321.
Article44. Verstockt B, Sudahakar P, Creyns B, et al. DOP70 An integrated multi-omics biomarker predicting endoscopic response in ustekinumab treated patients with Crohn’s disease. J Crohns Colitis. 2019; 13(Supplement_1):S072–S073.
Article45. Borren NZ, Plichta D, Joshi AD, et al. Multi-“-Omics” profiling in patients with quiescent inflammatory bowel disease identifies biomarkers predicting relapse. Inflamm Bowel Dis. 2020; 26:1524–1532.
Article46. Siegel CA, Horton H, Siegel LS, et al. A validated web-based tool to display individualised Crohn’s disease predicted outcomes based on clinical, serologic and genetic variables. Aliment Pharmacol Ther. 2016; 43:262–271.
Article47. D’Haens G, Kelly O, Battat R, et al. Development and validation of a test to monitor endoscopic activity in patients with Crohn’s disease based on serum levels of proteins. Gastroenterology. 2020; 158:515–526.
Article48. Sossenheimer PH, Yvellez OV, Andersen MJ, et al. 539 Wearable devices can predict disease activity in inflammatory bowel disease patients. Gastroenterology. 2019; 156:S–111.49. Wang P, Berzin TM, Glissen Brown JR, et al. Real-time automatic detection system increases colonoscopic polyp and adenoma detection rates: a prospective randomised controlled study. Gut. 2019; 68:1813–1819.
Article50. Korbar B, Olofson AM, Miraflor AP, et al. Deep learning for classification of colorectal polyps on whole-slide images. J Pathol Inform. 2017; 8:30.
Article51. Wei JW, Suriawinata AA, Vaickus LJ, et al. Evaluation of a deep neural network for automated classification of colorectal polyps on histopathologic slides. JAMA Netw Open. 2020; 3:e203398.
Article52. Bédard A, Westerling-Bui T, Zuraw A. Proof of concept for a deep learning algorithm for identification and quantification of key microscopic features in the murine model of DSS-induced colitis. Toxicol Pathol. 2021; 49:897–904.
Article53. Cheng JY, Abel JT, Balis UG, McClintock DS, Pantanowitz L. Challenges in the development, deployment, and regulation of artificial intelligence in anatomic pathology. Am J Pathol. 2021; 191:1684–1692.
Article54. Ehteshami Bejnordi B, Veta M, Johannes van Diest P, et al. Diagnostic assessment of deep learning algorithms for detection of lymph node metastases in women with breast cancer. JAMA. 2017; 318:2199–2210.55. Saha M, Chakraborty C, Racoceanu D. Efficient deep learning model for mitosis detection using breast histopathology images. Comput Med Imaging Graph. 2018; 64:29–40.
Article56. Ribli D, Horváth A, Unger Z, Pollner P, Csabai I. Detecting and classifying lesions in mammograms with deep learning. Sci Rep. 2018; 8:4165.
Article57. Raya-Povedano JL, Romero-Martín S, Elías-Cabot E, Gubern-Mérida A, Rodríguez-Ruiz A, Álvarez-Benito M. AI-based strategies to reduce workload in breast cancer screening with mammography and tomosynthesis: a retrospective evaluation. Radiology. 2021; 300:57–65.
Article58. de Groof AJ, Struyvenberg MR, van der Putten J, et al. Deeplearning system detects neoplasia in patients with Barrett’s esophagus with higher accuracy than endoscopists in a multistep training and validation study with benchmarking. Gastroenterology. 2020; 158:915–929.
Article59. Luo H, Xu G, Li C, et al. Real-time artificial intelligence for detection of upper gastrointestinal cancer by endoscopy: a multicentre, case-control, diagnostic study. Lancet Oncol. 2019; 20:1645–1654.
Article60. Ebigbo A, Mendel R, Rückert T, et al. Endoscopic prediction of submucosal invasion in Barrett’s cancer with the use of artificial intelligence: a pilot study. Endoscopy. 2021; 53:878–883.
Article61. Kudo SE, Misawa M, Mori Y, et al. Artificial intelligence-assisted system improves endoscopic identification of colorectal neoplasms. Clin Gastroenterol Hepatol. 2020; 18:1874–1881.
Article62. U.S. Food and Drug Administration. Food and Drug Administration. FDA authorizes marketing of first device that uses artificial intelligence to help detect potential signs of colon cancer [Internet]. c2021. [cited 2021 Jul 23]. https://www.fda.gov/news-events/press-announcements/fda-authorizes-marketing-first-device-uses-artificialintelligence-help-detect-potential-signs-colon.63. Ahuja AS. The impact of artificial intelligence in medicine on the future role of the physician. PeerJ. 2019; 7:e7702.
Article64. Sarwar S, Dent A, Faust K, et al. Physician perspectives on integration of artificial intelligence into diagnostic pathology. NPJ Digit Med. 2019; 2:28.
Article65. Walradt T, Glissen Brown JR, Alagappan M, Lerner HP, Berzin TM. Regulatory considerations for artificial intelligence technologies in GI endoscopy. Gastrointest Endosc. 2020; 92:801–806.
Article66. U.S. Food and Drug Administration. Food and Drug Administration. Proposed regulatory framework for modifications to artificial intelligence/machine learning (AI/ML)-based software as a medical device (SaMD): discussion paper and request for feedback [Internet]. c2021. [cited 2022 Dec 15]. https://www.fda.gov/files/medical%20devices/published/US-FDA-Artificial-Intelligence-and-Machine-Learning-Discussion-Paper.pdf.67. El Hajjar A, Rey JF. Artificial intelligence in gastrointestinal endoscopy: general overview. Chin Med J (Engl). 2020; 133:326–334.
Article68. Mori Y, Kudo SE, East JE, et al. Cost savings in colonoscopy with artificial intelligence-aided polyp diagnosis: an add-on analysis of a clinical trial (with video). Gastrointest Endosc. 2020; 92:905–911.69. Chen MM, Golding LP, Nicola GN. Who will pay for AI? Radiol Artif Intell. 2021; 3:e210030.
Article70. Rex DK. Making a resect-and-discard strategy work for diminutive colorectal polyps: let’s get real. Endoscopy. 2022; 54:364–366.
Article71. Zand A, Stokes Z, Sharma A, van Deen WK, Hommes D. Artificial intelligence for inflammatory bowel diseases (IBD); accurately predicting adverse outcomes using machine learning. Dig Dis Sci. 2022; 67:4874–4885.
Article72. Schoeb D, Suarez-Ibarrola R, Hein S, et al. Use of artificial intelligence for medical literature search: randomized controlled trial using the Hackathon Format. Interact J Med Res. 2020; 9:e16606.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Artificial intelligence for endoscopy in inflammatory bowel disease
- Artificial Intelligence in Lower Gastrointestinal Endoscopy: The Current Status and Future Perspective
- Real World Data and Artificial Intelligence in Diabetology
- Artificial Intelligence in Medicine: Beginner's Guide
- Application of artificial intelligence for diagnosis of early gastric cancer based on magnifying endoscopy with narrow-band imaging