Cancer Res Treat.
2023 Apr;55(2):488-497. 10.4143/crt.2022.359.
Real World Characteristics and Clinical Outcomes of HER2-Mutant Non–Small Cell Lung Cancer Patients Detected by Next-Generation Sequencing
- Affiliations
-
- 1Division of Medical Oncology, Department of Internal Medicine, Yonsei Cancer Center, Yonsei University College of Medicine, Seoul, Korea
- 2Center for Lung Cancer, Research Institute and Hospital, National Cancer Center, Goyang, Korea
- KMID: 2541235
- DOI: http://doi.org/10.4143/crt.2022.359
Abstract
- Purpose
This study was conducted to investigate the clinical characteristics of patients with advanced non–small cell lung cancer (NSCLC) harboring human epidermal growth factor receptor 2 (HER2) mutations and to evaluate response to standard treatment and HER2-targeted agents.
Materials and Methods
Using tissue and/or blood next-generation sequencing, we identified 44 patients with NSCLC harboring HER2 mutations who were treated at Severance Hospital between December 2016 and February 2021. Clinical data, including patient characteristics, mutation status, incidence of metastasis for distant lesions, and response to chemotherapy, were retrospectively analyzed.
Results
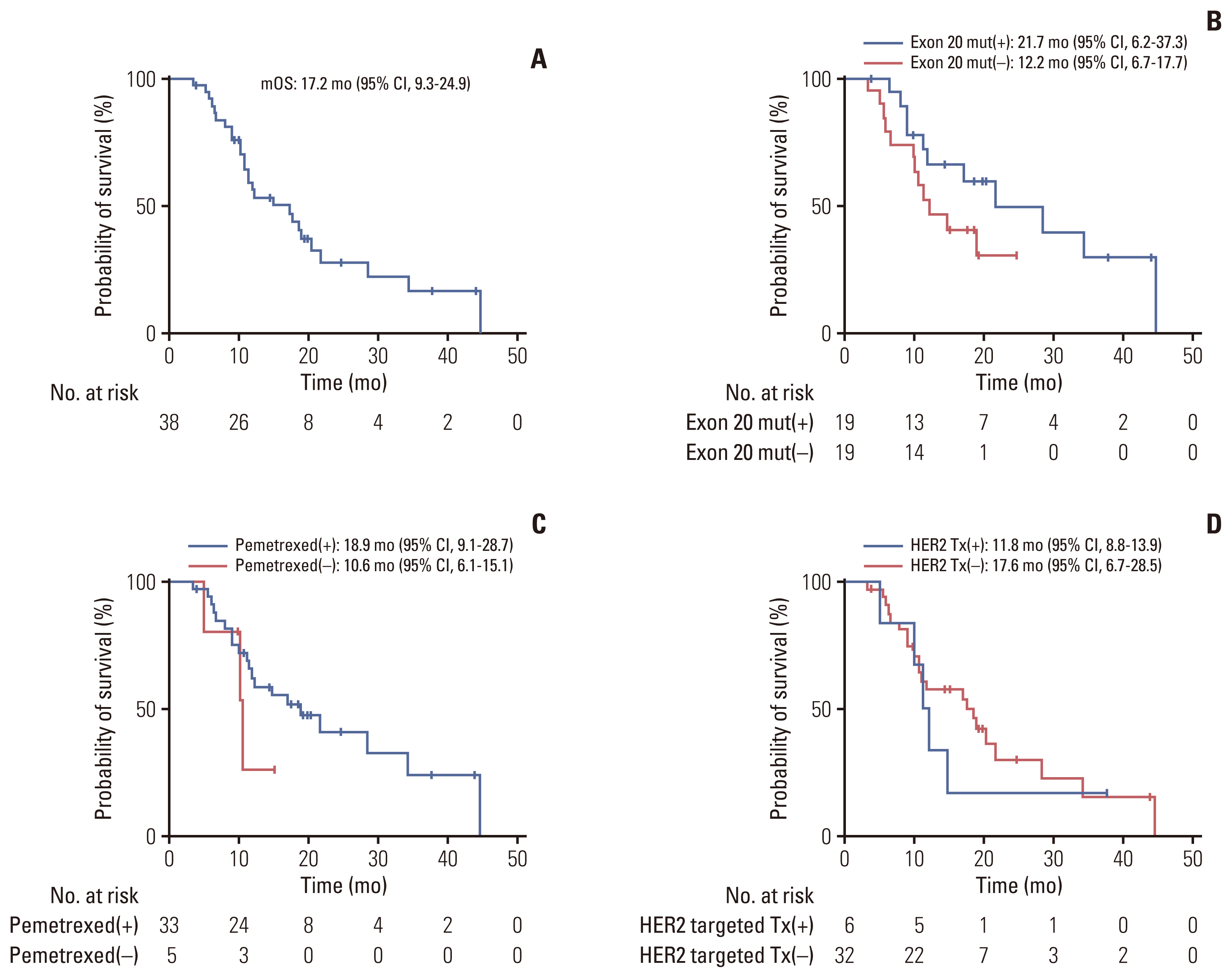
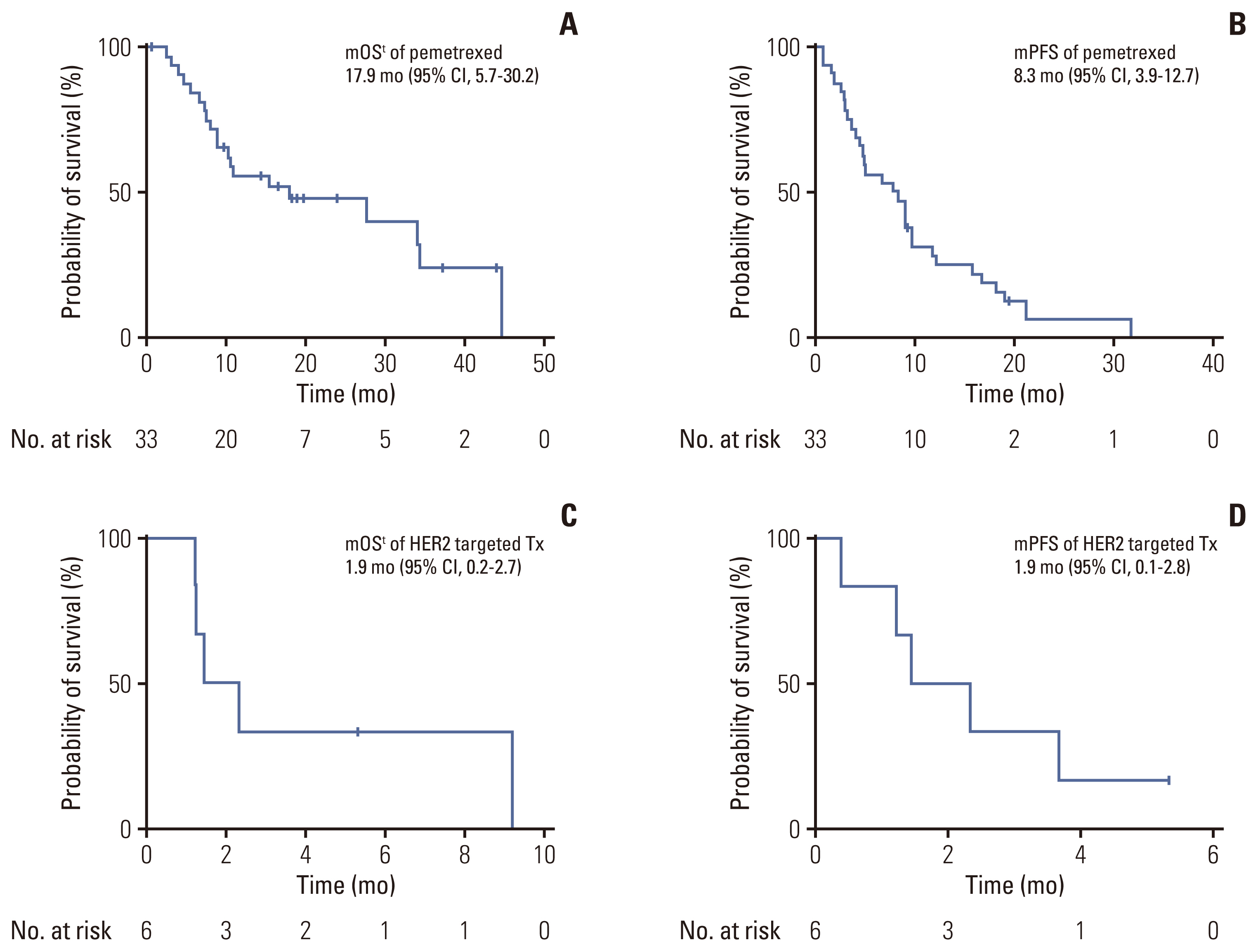
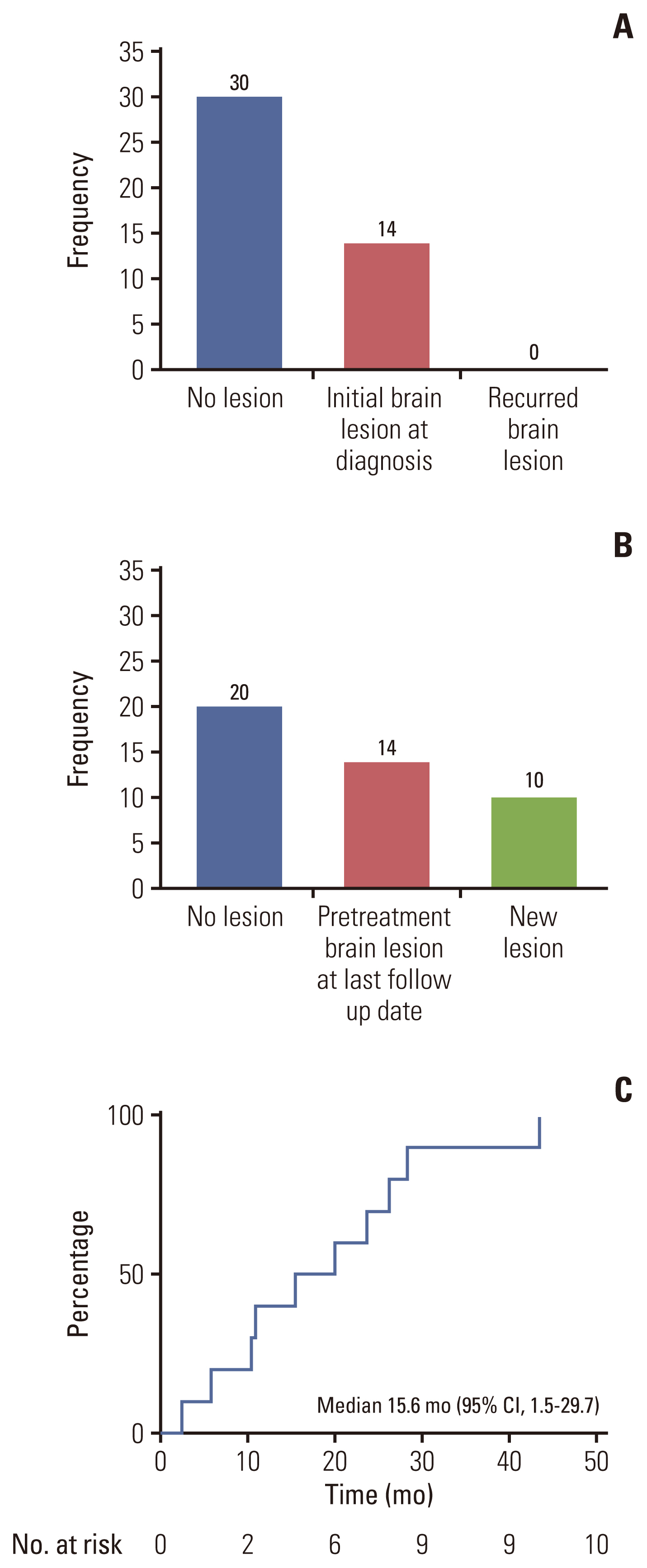
The median age was 58 years, and 61% of the patients were female. Most patients (64%) were never-smokers. Adenocarcinoma was the most predominant subtype (98%). A total of 66% of the patients had extrathoracic metastatic lesions, and 32% had intracranial lesions at initial presentation. The median time to the development of brain metastasis was 15.6 months (range, 2.4 to 43.7). The most common type of HER2 mutation was 12 base pair in-frame insertion in exon 20, A775_G776insYVMA. Of the 44 patients, two had concomitant driver mutations, one with epidermal growth factor receptor (EGFR) mutation (V769M), and one with BRAF mutation (V600E). Patients treated with pemetrexed-based chemotherapy (75%) had an overall response rate (ORR) and progression-free survival (PFS) of 30% and 8.3 months (95% confidence interval [CI], 3.9 to 12.7), respectively. The ORR and PFS of HER2-targeted agent treated patients (14%) were 0.0% and 1.9 months (95% CI, 0.1 to 2.8), respectively.
Conclusion
Given its distinct characteristics and treatment responses, novel treatment strategies for HER2-mutant NSCLC should be developed promptly to improve survival outcomes of patients.
Figure
Reference
-
References
1. Rosell R, Carcereny E, Gervais R, Vergnenegre A, Massuti B, Felip E, et al. Erlotinib versus standard chemotherapy as first-line treatment for European patients with advanced EGFR mutation-positive non-small-cell lung cancer (EURTAC): a multicentre, open-label, randomised phase 3 trial. Lancet Oncol. 2012; 13:239–46.2. Solomon BJ, Mok T, Kim DW, Wu YL, Nakagawa K, Mekhail T, et al. First-line crizotinib versus chemotherapy in ALK-positive lung cancer. N Engl J Med. 2014; 371:2167–77.
Article3. Shaw AT, Ou SH, Bang YJ, Camidge DR, Solomon BJ, Salgia R, et al. Crizotinib in ROS1-rearranged non-small-cell lung cancer. N Engl J Med. 2014; 371:1963–71.
Article4. Kris MG, Camidge DR, Giaccone G, Hida T, Li BT, O’Connell J, et al. Targeting HER2 aberrations as actionable drivers in lung cancers: phase II trial of the pan-HER tyrosine kinase inhibitor dacomitinib in patients with HER2-mutant or amp-lified tumors. Ann Oncol. 2015; 26:1421–7.
Article5. Li BT, Michelini F, Misale S, Cocco E, Baldino L, Cai Y, et al. HER2-mediated internalization of cytotoxic agents in ERBB2 amplified or mutant lung cancers. Cancer Discov. 2020; 10:674–87.6. Patil T, Mushtaq R, Marsh S, Azelby C, Pujara M, Davies KD, et al. Clinicopathologic characteristics, treatment outcomes, and acquired resistance patterns of atypical EGFR mutations and HER2 alterations in stage IV non-small-cell lung cancer. Clin Lung Cancer. 2020; 21:e191–204.
Article7. Zhou J, Ding N, Xu X, Zhang Y, Ye M, Li C, et al. Clinical outcomes of patients with HER2-mutant advanced lung cancer: chemotherapies versus HER2-directed therapies. Ther Adv Med Oncol. 2020; 12:1758835920936090.
Article8. Mazieres J, Barlesi F, Filleron T, Besse B, Monnet I, Beau-Faller M, et al. Lung cancer patients with HER2 mutations treated with chemotherapy and HER2-targeted drugs: results from the European EUHER2 cohort. Ann Oncol. 2016; 27:281–6.
Article9. Odegaard JI, Vincent JJ, Mortimer S, Vowles JV, Ulrich BC, Banks KC, et al. Validation of a plasma-based comprehensive cancer genotyping assay utilizing orthogonal tissue- and plasma-based methodologies. Clin Cancer Res. 2018; 24:3539–49.
Article10. Li MM, Datto M, Duncavage EJ, Kulkarni S, Lindeman NI, Roy S, et al. Standards and guidelines for the interpretation and reporting of sequence variants in cancer: a Joint Consensus Recommendation of the Association for Molecular Pathology, American Society of Clinical Oncology, and College of American Pathologists. J Mol Diagn. 2017; 19:4–23.
Article11. Han Y, Lim SM, Kim HR, Hong MH, Cho BC, Ahn B. P50.09 Characteristics and clinical outcomes of HER2 mutated non-small cell lung cancer patients detected by NGS in routine clinical practice. J Thorac Oncol. 2021; 16(10 Suppl):S1117–8.
Article12. Mazieres J, Peters S, Lepage B, Cortot AB, Barlesi F, Beau-Faller M, et al. Lung cancer that harbors an HER2 mutation: epidemiologic characteristics and therapeutic perspectives. J Clin Oncol. 2013; 31:1997–2003.13. Lee K, Jung HA, Sun JM, Lee SH, Ahn JS, Park K, et al. Clinical characteristics and outcomes of non-small cell lung cancer patients with HER2 alterations in Korea. Cancer Res Treat. 2020; 52:292–300.
Article14. Oh IJ, Hur JY, Park CK, Kim YC, Kim SJ, Lee MK, et al. Clinical activity of Pan-HER inhibitors against HER2-mutant lung adenocarcinoma. Clin Lung Cancer. 2018; 19:e775–81.
Article15. Hainsworth JD, Meric-Bernstam F, Swanton C, Hurwitz H, Spigel DR, Sweeney C, et al. Targeted therapy for advanced solid tumors on the basis of molecular profiles: results from MyPathway, an open-label, phase IIa multiple basket study. J Clin Oncol. 2018; 36:536–42.
Article16. Rolfo C, Russo A. HER2 mutations in non-small cell lung cancer: a herculean effort to hit the target. Cancer Discov. 2020; 10:643–5.17. Suzawa K, Toyooka S, Sakaguchi M, Morita M, Yamamoto H, Tomida S, et al. Antitumor effect of afatinib, as a human epidermal growth factor receptor 2-targeted therapy, in lung cancers harboring HER2 oncogene alterations. Cancer Sci. 2016; 107:45–52.
Article18. De Greve J, Teugels E, Geers C, Decoster L, Galdermans D, De Mey J, et al. Clinical activity of afatinib (BIBW 2992) in patients with lung adenocarcinoma with mutations in the kinase domain of HER2/neu. Lung Cancer. 2012; 76:123–7.
Article19. Chan A, Delaloge S, Holmes FA, Moy B, Iwata H, Harvey VJ, et al. Neratinib after trastuzumab-based adjuvant therapy in patients with HER2-positive breast cancer (ExteNET): a multicentre, randomised, double-blind, placebo-controlled, phase 3 trial. Lancet Oncol. 2016; 17:367–77.
Article20. Li B, Gandhi L, Besse B, Jhaveri K, Mazieres J, Boni V, et al. FP14.15 Neratinib-based combination therapy in HER2- mutant lung adenocarcinomas: findings from two internatio-nal phase 2 studies. J Thorac Oncol. 2021; 16(3 Suppl):S234.21. Peters S, Stahel R, Bubendorf L, Bonomi P, Villegas A, Kowalski DM, et al. Trastuzumab emtansine (T-DM1) in patients with previously treated HER2-overexpressing metastatic non-small cell lung cancer: efficacy, safety, and biomarkers. Clin Cancer Res. 2019; 25:64–72.
Article22. Tsurutani J, Iwata H, Krop I, Janne PA, Doi T, Takahashi S, et al. Targeting HER2 with trastuzumab deruxtecan: a dose-expansion, phase I study in multiple advanced solid tumors. Cancer Discov. 2020; 10:688–701.
Article23. Li BT, Smit EF, Goto Y, Nakagawa K, Udagawa H, Mazieres J, et al. Trastuzumab deruxtecan in HER2-mutant non-small-cell lung cancer. N Engl J Med. 2022; 386:241–51.
Article24. Mazieres J, Drilon A, Lusque A, Mhanna L, Cortot AB, Mezquita L, et al. Immune checkpoint inhibitors for patients with advanced lung cancer and oncogenic driver alterations: results from the IMMUNOTARGET registry. Ann Oncol. 2019; 30:1321–8.
Article25. Chen K, Pan G, Cheng G, Zhang F, Xu Y, Huang Z, et al. Immune microenvironment features and efficacy of PD-1/PD-L1 blockade in non-small cell lung cancer patients with EGFR or HER2 exon 20 insertions. Thorac Cancer. 2021; 12:218–26.
Article26. Rybarczyk-Kasiuchnicz A, Ramlau R, Stencel K. Treatment of brain metastases of non-small cell lung carcinoma. Int J Mol Sci. 2021; 22:593.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Clinical Characteristics and Outcomes of Non-small Cell Lung Cancer Patients with HER2 Alterations in Korea
- Efficacy of Afatinib in a Previously-Treated Patient with Non-Small Cell Lung Cancer Harboring HER2 Mutation: Case Report
- Comparison of Clinicopathogenomic Features and Treatment Outcomes of EGFR and HER2 Exon 20 Insertion Mutations in Non–Small Cell Lung Cancer: Single-Institution Experience
- Real-World Evidence of Trastuzumab, Pertuzumab, and Docetaxel Combination as a First-Line Treatment for Korean Patients with HER2-Positive Metastatic Breast Cancer
- KRAS Mutation Detection in Non-small Cell Lung Cancer Using a Peptide Nucleic Acid-Mediated Polymerase Chain Reaction Clamping Method and Comparative Validation with Next-Generation Sequencing