Cancer Res Treat.
2023 Jan;55(1):167-178. 10.4143/crt.2022.094.
Molecular and Immune Profiling of Syngeneic Mouse Models Predict Response to Immune Checkpoint Inhibitors in Gastric Cancer
- Affiliations
-
- 1Department of Surgery, Ajou University School of Medicine, Suwon, Korea
- 2Cancer Biology Graduate Program, Ajou University Graduate School of Medicine, Suwon, Korea
- 3Inflamm-Aging Translational Research Center, Ajou University School of Medicine, Suwon, Korea
- 4Department of Hospital Pathology, College of Medicine, The Catholic University of Korea, Seoul, Korea
- 5Department of Gastrointestinal Surgery, Graduate School of Medicine, The University of Tokyo, Tokyo, Japan
- KMID: 2538002
- DOI: http://doi.org/10.4143/crt.2022.094
Abstract
- Purpose
Appropriate preclinical mouse models are needed to evaluate the response to immunotherapeutic agents. Immunocompetent mouse models have rarely been reported for gastric cancer. Thus, we investigated immunophenotypes and responses to immune checkpoint inhibitor (ICI) in immunocompetent mouse models using various murine gastric cancer cell lines.
Materials and Methods
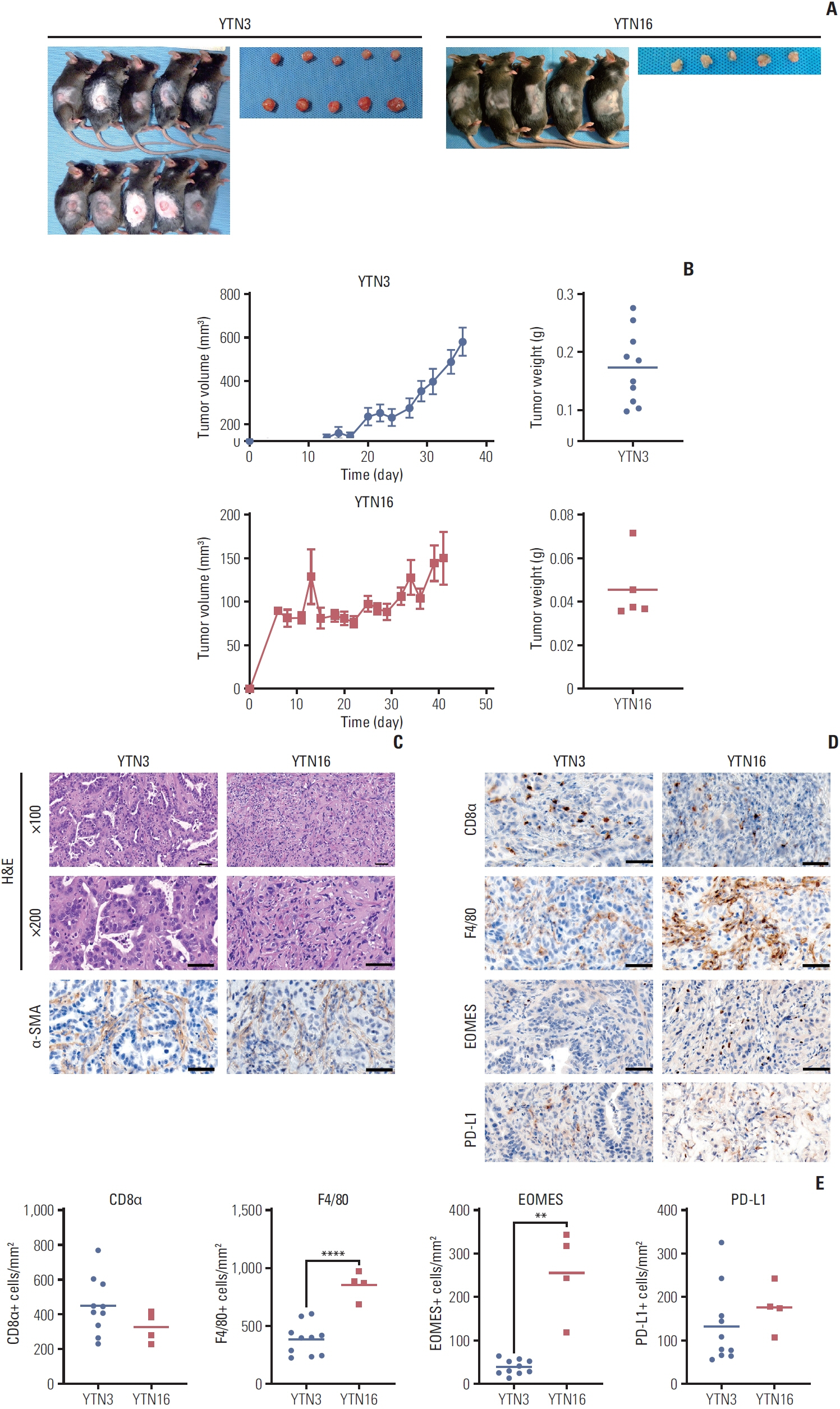
We constructed subcutaneous syngeneic tumors with murine gastric cancer cell lines, YTN3 and YTN16, in C57BL/6J mice. Mice were intraperitoneally treated with IgG isotype control or an anti–programmed death-ligand 1 (PD-L1) neutralizing antibody. We used immunohistochemistry to evaluate the tumor-infiltrating immune cells of formalin-fixed paraffin-embedded mouse tumor tissues. We compared the protein and RNA expression between YTN3 and YTN16 cell lines using a mouse cytokine array and RNA sequencing.
Results
The mouse tumors revealed distinct histological and molecular characteristics. YTN16 cells showed upregulation of genes and proteins related to immunosuppression, such as Ccl2 (CCL2) and Csf1 (M-CSF). Macrophages and exhausted T cells were more enriched in YTN16 tumors than in YTN3 tumors. Several YTN3 tumors were completely regressed by the PD-L1 inhibitor, whereas YTN16 tumors were unaffected. Although treatment with a PD-L1 inhibitor increased infiltration of T cells in both the tumors, the proportion of exhausted immune cells did not decrease in the non-responder group.
Conclusion
We confirmed the histological and molecular features of cancer cells with various responses to ICI. Our models can be used in preclinical research on ICI resistance mechanisms to enhance clinical efficacy.
Keyword
Figure
Reference
-
References
1. Sung H, Ferlay J, Siegel RL, Laversanne M, Soerjomataram I, Jemal A, et al. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2021; 71:209–49.2. Kang YK, Boku N, Satoh T, Ryu MH, Chao Y, Kato K, et al. Nivolumab in patients with advanced gastric or gastro-oesophageal junction cancer refractory to, or intolerant of, at least two previous chemotherapy regimens (ONO-4538-12, ATTRACTION-2): a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet. 2017; 390:2461–71.3. Wu T, Dai Y. Tumor microenvironment and therapeutic response. Cancer Lett. 2017; 387:61–8.4. Hirata E, Sahai E. Tumor microenvironment and differential responses to therapy. Cold Spring Harb Perspect Med. 2017; 7:a026781.5. Roma-Rodrigues C, Raposo LR, Cabral R, Paradinha F, Baptista PV, Fernandes AR. Tumor microenvironment modulation via gold nanoparticles targeting malicious exosomes: implications for cancer diagnostics and therapy. Int J Mol Sci. 2017; 18:162.6. Chulpanova DS, Kitaeva KV, Rutland CS, Rizvanov AA, Solovyeva VV. Mouse tumor models for advanced cancer immunotherapy. Int J Mol Sci. 2020; 21:4118.7. Hsu HP, Wang CY, Hsieh PY, Fang JH, Chen YL. Knockdown of serine/threonine-protein kinase 24 promotes tumorigenesis and myeloid-derived suppressor cell expansion in an orthotopic immunocompetent gastric cancer animal model. J Cancer. 2020; 11:213–28.8. Park JW, Um H, Yang H, Ko W, Kim DY, Kim HK. Proteogenomic analysis of NCC-S1M, a gastric cancer stem cell-like cell line that responds to anti-PD-1. Biochem Biophys Res Commun. 2017; 484:631–5.9. Yamamoto M, Nomura S, Hosoi A, Nagaoka K, Iino T, Yasuda T, et al. Established gastric cancer cell lines transplantable into C57BL/6 mice show fibroblast growth factor receptor 4 promotion of tumor growth. Cancer Sci. 2018; 109:1480–92.10. Huang da W, Sherman BT, Lempicki RA. Systematic and integrative analysis of large gene lists using DAVID bioinformatics resources. Nat Protoc. 2009; 4:44–57.11. Saraiva M, Vieira P, O’Garra A. Biology and therapeutic potential of interleukin-10. J Exp Med. 2020; 217:e20190418.12. Castro F, Cardoso AP, Goncalves RM, Serre K, Oliveira MJ. Interferon-gamma at the crossroads of tumor immune surveillance or evasion. Front Immunol. 2018; 9:847.13. Perrier S, Darakhshan F, Hajduch E. IL-1 receptor antagonist in metabolic diseases: Dr Jekyll or Mr Hyde? FEBS Lett. 2006; 580:6289–94.14. Nagaoka K, Shirai M, Taniguchi K, Hosoi A, Sun C, Kobayashi Y, et al. Deep immunophenotyping at the single-cell level identifies a combination of anti-IL-17 and checkpoint blockade as an effective treatment in a preclinical model of data-guided personalized immunotherapy. J Immunother Cancer. 2020; 8:e001358.15. Cancer Genome Atlas Research Network. Comprehensive molecular characterization of gastric adenocarcinoma. Nature. 2014; 513:202–9.16. De Rosa S, Sahnane N, Tibiletti MG, Magnoli F, Vanoli A, Sessa F, et al. EBV(+) and MSI gastric cancers harbor high PD-L1/PD-1 expression and high CD8(+) intratumoral lymphocytes. Cancers (Basel). 2018; 10:102.17. Rodriquenz MG, Roviello G, D’Angelo A, Lavacchi D, Roviello F, Polom K. MSI and EBV positive gastric cancer’s subgroups and their link with novel immunotherapy. J Clin Med. 2020; 9:1427.18. Cristescu R, Lee J, Nebozhyn M, Kim KM, Ting JC, Wong SS, et al. Molecular analysis of gastric cancer identifies subtypes associated with distinct clinical outcomes. Nat Med. 2015; 21:449–56.19. Kersten K, Coffelt SB, Hoogstraat M, Verstegen NJ, Vrijland K, Ciampricotti M, et al. Mammary tumor-derived CCL2 enhances pro-metastatic systemic inflammation through upregulation of IL1beta in tumor-associated macrophages. Oncoimmunology. 2017; 6:e1334744.20. Lee GT, Kwon SJ, Kim J, Kwon YS, Lee N, Hong JH, et al. WNT5A induces castration-resistant prostate cancer via CCL2 and tumour-infiltrating macrophages. Br J Cancer. 2018; 118:670–8.21. Van Overmeire E, Stijlemans B, Heymann F, Keirsse J, Morias Y, Elkrim Y, et al. M-CSF and GM-CSF receptor signaling differentially regulate monocyte maturation and macrophage polarization in the tumor microenvironment. Cancer Res. 2016; 76:35–42.22. Peranzoni E, Lemoine J, Vimeux L, Feuillet V, Barrin S, Kantari-Mimoun C, et al. Macrophages impede CD8 T cells from reaching tumor cells and limit the efficacy of anti-PD-1 treatment. Proc Natl Acad Sci U S A. 2018; 115:E4041–50.23. Jeong HY, Ham IH, Lee SH, Ryu D, Son SY, Han SU, et al. Spatially distinct reprogramming of the tumor microenvironment based on tumor invasion in diffuse-type gastric cancers. Clin Cancer Res. 2021; 27:6529–42.24. Sharma P, Wagner K, Wolchok JD, Allison JP. Novel cancer immunotherapy agents with survival benefit: recent successes and next steps. Nat Rev Cancer. 2011; 11:805–12.25. Huang RR, Jalil J, Economou JS, Chmielowski B, Koya RC, Mok S, et al. CTLA4 blockade induces frequent tumor infiltration by activated lymphocytes regardless of clinical responses in humans. Clin Cancer Res. 2011; 17:4101–9.26. Ng Tang D, Shen Y, Sun J, Wen S, Wolchok JD, Yuan J, et al. Increased frequency of ICOS+ CD4 T cells as a pharmacodynamic biomarker for anti-CTLA-4 therapy. Cancer Immunol Res. 2013; 1:229–34.27. Chen PL, Roh W, Reuben A, Cooper ZA, Spencer CN, Prieto PA, et al. Analysis of immune signatures in longitudinal tumor samples yields insight into biomarkers of response and mechanisms of resistance to immune checkpoint blockade. Cancer Discov. 2016; 6:827–37.28. Im SJ, Hashimoto M, Gerner MY, Lee J, Kissick HT, Burger MC, et al. Defining CD8+ T cells that provide the proliferative burst after PD-1 therapy. Nature. 2016; 537:417–21.29. Koyama S, Akbay EA, Li YY, Herter-Sprie GS, Buczkowski KA, Richards WG, et al. Adaptive resistance to therapeutic PD-1 blockade is associated with upregulation of alternative immune checkpoints. Nat Commun. 2016; 7:10501.30. Weulersse M, Asrir A, Pichler AC, Lemaitre L, Braun M, Carrie N, et al. Eomes-dependent loss of the co-activating receptor CD226 restrains CD8(+) T cell anti-tumor functions and limits the efficacy of cancer immunotherapy. Immunity. 2020; 53:824–39.31. Bang YJ, Ruiz EY, Van Cutsem E, Lee KW, Wyrwicz L, Schenker M, et al. Phase III, randomised trial of avelumab versus physician’s choice of chemotherapy as third-line treatment of patients with advanced gastric or gastro-oesophageal junction cancer: primary analysis of JAVELIN Gastric 300. Ann Oncol. 2018; 29:2052–60.32. Kang W, Maher L, Michaud M, Bae SW, Kim S, Lee HS, et al. Development of a novel orthotopic gastric cancer mouse model. Biol Proced Online. 2021; 23:1.33. Song S, Xu Y, Huo L, Zhao S, Wang R, Li Y, et al. Patient-derived cell lines and orthotopic mouse model of peritoneal carcinomatosis recapitulate molecular and phenotypic features of human gastric adenocarcinoma. J Exp Clin Cancer Res. 2021; 40:207.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Gastrointestinal cancer treatment with immune checkpoint inhibitors
- Gut microbiome on immune checkpoint inhibitor therapy and consequent immune-related colitis: a review
- Immune-Checkpoint Inhibitors in the Era of Precision Medicine: What Radiologists Should Know
- Treatment of advanced urogenital cancers with immune checkpoint inhibitors
- Current status of cancer immunotherapy with immune checkpoint inhibitors