Korean J Physiol Pharmacol.
2022 Sep;26(5):347-355. 10.4196/kjpp.2022.26.5.347.
Activating transcription factor 4 aggravates angiotensin IIinduced cell dysfunction in human vascular aortic smooth muscle cells via transcriptionally activating fibroblast growth factor 21
- Affiliations
-
- 1Department of General Surgery, Changshu Hospital Affiliated to Soochow University, Changshu 215500, China
- 2Department of Vascular Surgery, The Affiliated Suzhou Hospital of Nanjing Medical University, Suzhou 215008, China
- KMID: 2532764
- DOI: http://doi.org/10.4196/kjpp.2022.26.5.347
Abstract
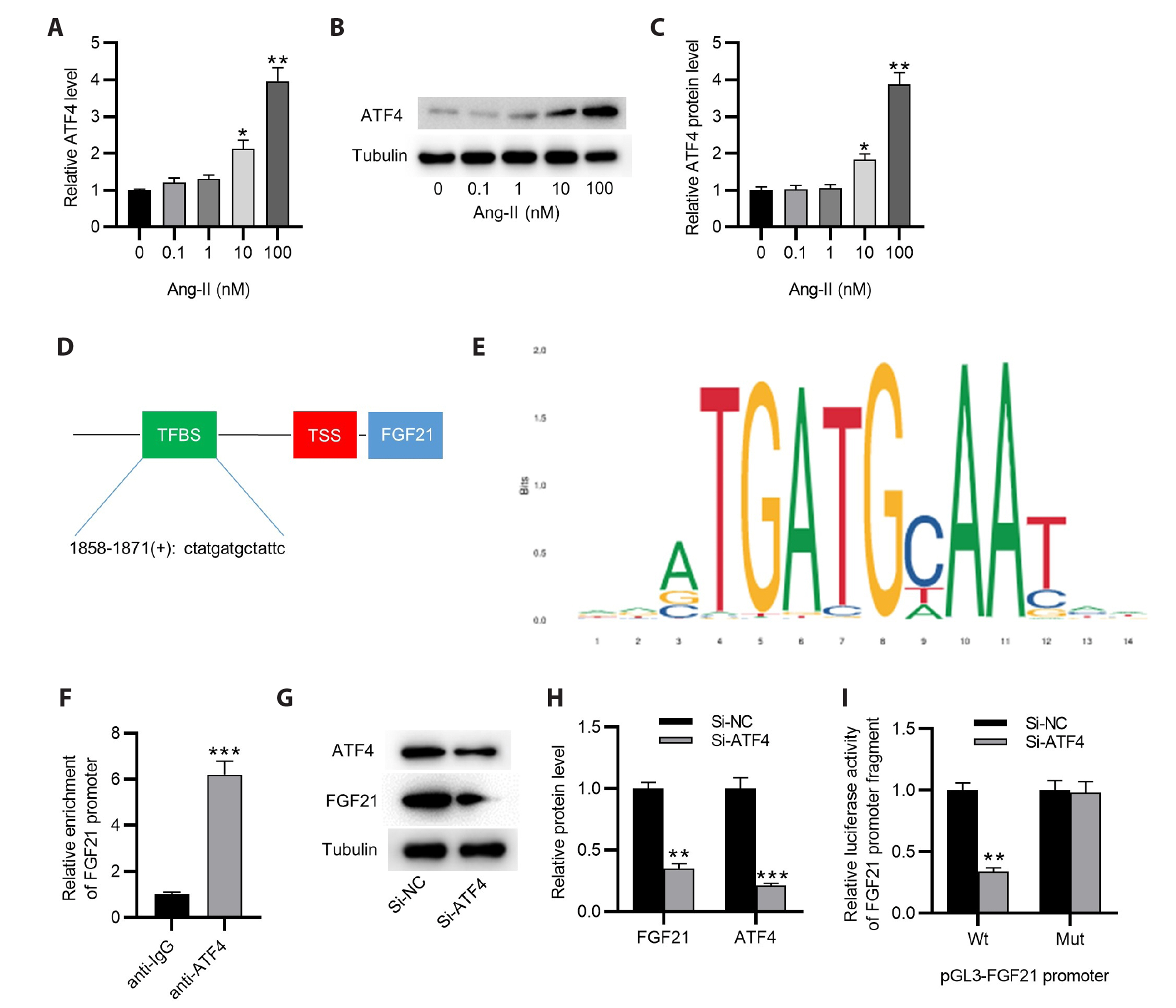
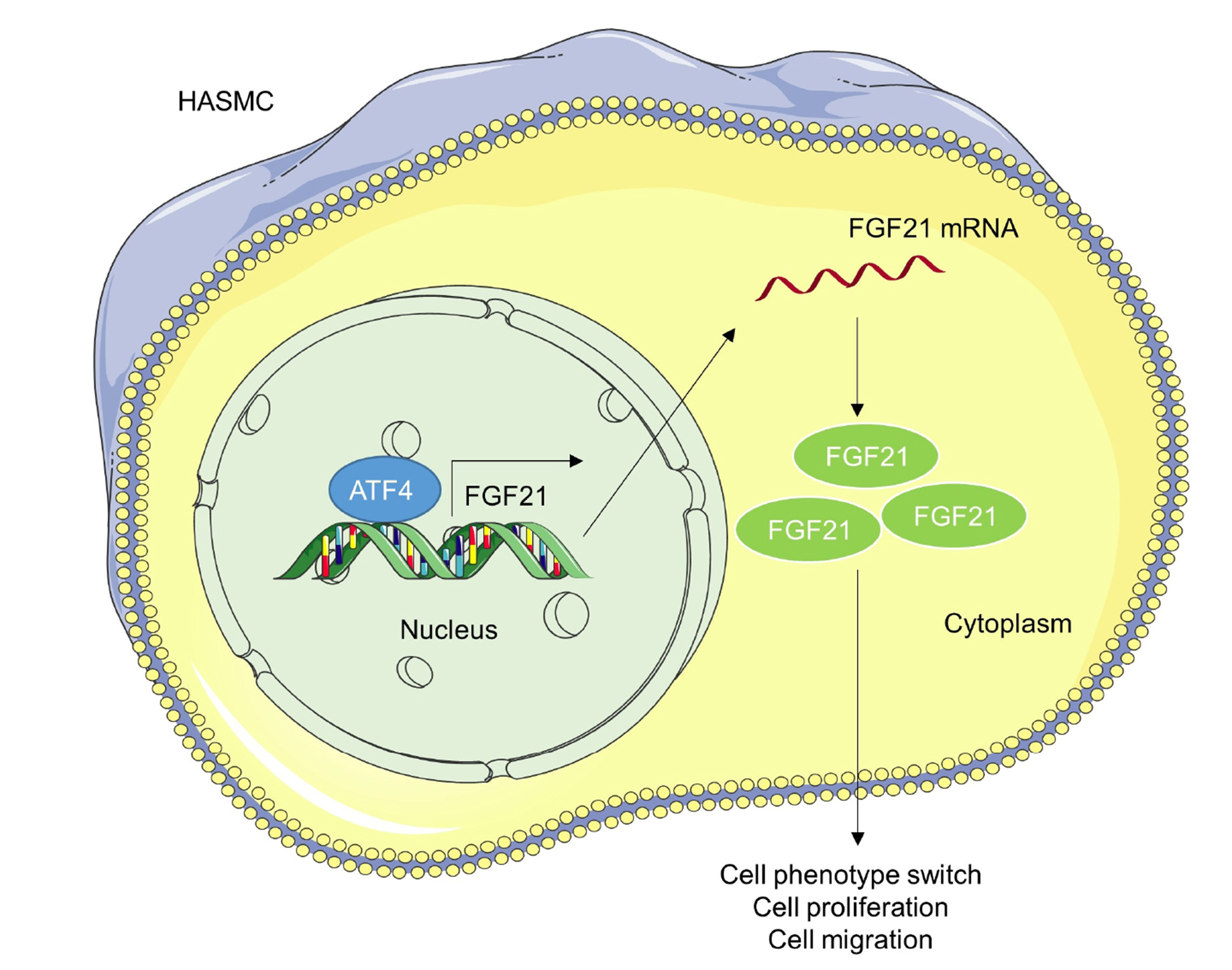
- Abdominal aortic aneurysm (AAA) is a life-threatening disorder worldwide. Fibroblast growth factor 21 (FGF21) was shown to display a high level in the plasma of patients with AAA; however, its detailed functions underlying AAA pathogenesis are unclear. An in vitro AAA model was established in human aortic vascular smooth muscle cells (HASMCs) by angiotensin II (Ang-II) stimulation. Cell counting kit-8, wound healing, and Transwell assays were utilized for measuring cell proliferation and migration. RT-qPCR was used for detecting mRNA expression of FGF21 and activating transcription factor 4 (ATF4). Western blotting was utilized for assessing protein levels of FGF21, ATF4, and markers for the contractile phenotype of HASMCs. ChIP and luciferase reporter assays were implemented for identifying the binding relation between AFT4 and FGF21 promoters. FGF21 and ATF4 were both upregulated in Ang-II-treated HASMCs. Knocking down FGF21 attenuated Ang-IIinduced proliferation, migration, and phenotype switch of HASMCs. ATF4 activated FGF21 transcription by binding to its promoter. FGF21 overexpression reversed AFT4 silencing-mediated inhibition of cell proliferation, migration, and phenotype switch. ATF4 transcriptionally upregulates FGF21 to promote the proliferation, migration, and phenotype switch of Ang-II-treated HASMCs.
Keyword
Figure
Reference
-
1. Mason BL, Minhajuddin A, Czysz AH, Jha MK, Gadad BS, Mayes TL, Trivedi MH. 2022; Fibroblast growth factor 21 (FGF21) is increased in MDD and interacts with body mass index (BMI) to affect depression trajectory. Transl Psychiatry. 12:16. DOI: 10.1038/s41398-021-01679-y. PMID: 35017468. PMCID: PMC8752780. PMID: 741a0a0f9d014ac49f40f05279d94e70.
Article2. Kakoty V, K C S, Tang RD, Yang CH, Dubey SK, Taliyan R. 2020; Fibroblast growth factor 21 and autophagy: a complex interplay in Parkinson disease. Biomed Pharmacother. 127:110145. DOI: 10.1016/j.biopha.2020.110145. PMID: 32361164.
Article3. Lu H, Jia C, Wu D, Jin H, Lin Z, Pan J, Li X, Wang W. 2021; Fibroblast growth factor 21 (FGF21) alleviates senescence, apoptosis, and extracellular matrix degradation in osteoarthritis via the SIRT1-mTOR signaling pathway. Cell Death Dis. 12:865. DOI: 10.1038/s41419-021-04157-x. PMID: 34556628. PMCID: PMC8460788. PMID: 78876a2127d747b0a53e5dd0597c8086.
Article4. Shi Y, Wang S, Peng H, Lv Y, Li W, Cheng S, Liu J. 2019; Fibroblast growth factor 21 attenuates vascular calcification by alleviating endoplasmic reticulum stress mediated apoptosis in rats. Int J Biol Sci. 15:138–147. DOI: 10.7150/ijbs.28873. PMID: 30662354. PMCID: PMC6329919.
Article5. Huang WP, Chen CY, Lin TW, Kuo CS, Huang HL, Huang PH, Lin SJ. 2022; Fibroblast growth factor 21 reverses high-fat diet-induced impairment of vascular function via the anti-oxidative pathway in ApoE knockout mice. J Cell Mol Med. 26:2451–2461. DOI: 10.1111/jcmm.17273. PMID: 35307922. PMCID: PMC8995458.
Article6. Pan X, Shao Y, Wu F, Wang Y, Xiong R, Zheng J, Tian H, Wang B, Wang Y, Zhang Y, Han Z, Qu A, Xu H, Lu A, Yang T, Li X, Xu A, Du J, Lin Z. 2018; FGF21 prevents angiotensin II-induced hypertension and vascular dysfunction by activation of ACE2/angiotensin-(1-7) axis in mice. Cell Metab. 27:1323–1337.e5. DOI: 10.1016/j.cmet.2018.04.002. PMID: 29706566.
Article7. Xie T, Yin L, Guo D, Zhang Z, Chen Y, Liu B, Wang W, Zheng Y. 2021; The potential role of plasma fibroblast growth factor 21 as a diagnostic biomarker for abdominal aortic aneurysm presence and development. Life Sci. 274:119346. DOI: 10.1016/j.lfs.2021.119346. PMID: 33713667.
Article8. Ebert SM, Rasmussen BB, Judge AR, Judge SM, Larsson L, Wek RC, Anthony TG, Marcotte GR, Miller MJ, Yorek MA, Vella A, Volpi E, Stern JI, Strub MD, Ryan Z, Talley JJ, Adams CM. 2022; Biology of activating transcription factor 4 (ATF4) and its role in skeletal muscle atrophy. J Nutr. 152:926–938. DOI: 10.1093/jn/nxab440. PMID: 34958390. PMCID: PMC8970988.
Article9. Kim KH, Jeong YT, Kim SH, Jung HS, Park KS, Lee HY, Lee MS. 2013; Metformin-induced inhibition of the mitochondrial respiratory chain increases FGF21 expression via ATF4 activation. Biochem Biophys Res Commun. 440:76–81. DOI: 10.1016/j.bbrc.2013.09.026. PMID: 24041694.
Article10. Maruyama R, Shimizu M, Li J, Inoue J, Sato R. 2016; Fibroblast growth factor 21 induction by activating transcription factor 4 is regulated through three amino acid response elements in its promoter region. Biosci Biotechnol Biochem. 80:929–934. DOI: 10.1080/09168451.2015.1135045. PMID: 27010621.
Article11. Ni XQ, Lu WW, Zhang JS, Zhu Q, Ren JL, Yu YR, Liu XY, Wang XJ, Han M, Jing Q, Du J, Tang CS, Qi YF. 2018; Inhibition of endoplasmic reticulum stress by intermedin1-53 attenuates angiotensin II-induced abdominal aortic aneurysm in ApoE KO Mice. Endocrine. 62:90–106. DOI: 10.1007/s12020-018-1657-6. PMID: 29943223.
Article12. Zhou Z, Zhou H, Zou X, Wang X. 2022; RUNX3 is up-regulated in abdominal aortic aneurysm and regulates the function of vascular smooth muscle cells by regulating TGF-β1. J Mol Histol. 53:1–11. DOI: 10.1007/s10735-021-10035-9. PMID: 34813022.
Article13. Liu X, Chen X, Xu C, Lou J, Weng Y, Tang L. 2022; Platelet protects angiotensin II-driven abdominal aortic aneurysm formation through inhibition of inflammation. Exp Gerontol. 159:111703. DOI: 10.1016/j.exger.2022.111703. PMID: 35038567.
Article14. Gao P, Zhang H, Zhang Q, Fang X, Wu H, Wang M, Lu Z, Wei X, Yang G, Yan Z, Liu D, Zhu Z. 2019; Caloric restriction exacerbates angiotensin II-induced abdominal aortic aneurysm in the absence of p53. Hypertension. 73:547–560. DOI: 10.1161/HYPERTENSIONAHA.118.12086. PMID: 30686087.
Article15. Ma M, Yang X, Han F, Wang H. 2022; Circ_0092291 attenuates angiotensin II-induced cell damages in human aortic vascular smooth muscle cells via mediating the miR-626/COL4A1 signal axis. J Physiol Biochem. 78:245–256. DOI: 10.1007/s13105-021-00859-0. PMID: 34997455.
Article16. Chiang MT, Chen IM, Hsu FF, Chen YH, Tsai MS, Hsu YW, Leu HB, Huang PH, Chen JW, Liu FT, Chen YH, Chau LY. 2021; Gal-1 (galectin-1) upregulation contributes to abdominal aortic aneurysm progression by enhancing vascular inflammation. Arterioscler Thromb Vasc Biol. 41:331–345. DOI: 10.1161/ATVBAHA.120.315398. PMID: 33147994.
Article17. Horimatsu T, Blomkalns AL, Ogbi M, Moses M, Kim D, Patel S, Gilreath N, Reid L, Benson TW, Pye J, Ahmadieh S, Thompson A, Robbins N, Mann A, Edgell A, Benjamin S, Stansfield BK, Huo Y, Fulton DJ, Agarwal G, et al. 2020; Niacin protects against abdominal aortic aneurysm formation via GPR109A independent mechanisms: role of NAD+/nicotinamide. Cardiovasc Res. 116:2226–2238. DOI: 10.1093/cvr/cvz303. PMID: 31710686. PMCID: PMC7695356.
Article18. Doyle BJ, Bappoo N, Syed MBJ, Forsythe RO, Powell JT, Conlisk N, Hoskins PR, McBride OMB, Shah ASV, Norman PE, Newby DE. 2020; Biomechanical assessment predicts aneurysm related events in patients with abdominal aortic aneurysm. Eur J Vasc Endovasc Surg. 60:365–373. DOI: 10.1016/j.ejvs.2020.02.023. PMID: 32253165.
Article19. Bernal S, Lopez-Sanz L, Jimenez-Castilla L, Prieto I, Melgar A, La Manna S, Martin-Ventura JL, Blanco-Colio LM, Egido J, Gomez-Guerrero C. 2021; Protective effect of suppressor of cytokine signalling 1-based therapy in experimental abdominal aortic aneurysm. Br J Pharmacol. 178:564–581. DOI: 10.1111/bph.15330. PMID: 33227156.
Article20. Memon AA, Zarrouk M, Ågren-Witteschus S, Sundquist J, Gottsäter A, Sundquist K. 2020; Identification of novel diagnostic and prognostic biomarkers for abdominal aortic aneurysm. Eur J Prev Cardiol. 27:132–142. DOI: 10.1177/2047487319873062. PMID: 31466471.
Article21. Riches-Suman K, Hussain A. 2022; Identifying and targeting the molecular signature of smooth muscle cells undergoing early vascular ageing. Biochim Biophys Acta Mol Basis Dis. 1868:166403. DOI: 10.1016/j.bbadis.2022.166403. PMID: 35367337.
Article22. Hsu CY, Vo TTT, Lee CW, Chen YL, Lin WN, Cheng HC, Vo QC, Lee IT. 2022; Carbon monoxide releasing molecule-2 attenuates angiotensin II-induced IL-6/Jak2/Stat3-associated inflammation by inhibiting NADPH oxidase- and mitochondria-derived ROS in human aortic smooth muscle cells. Biochem Pharmacol. 198:114978. DOI: 10.1016/j.bcp.2022.114978. PMID: 35218740.
Article23. Jiang B, Wang M, Li X, Ren P, Li G, Wang Y, Wang L, Li X, Yang D, Qin L, Xin S. 2022; Hexarelin attenuates abdominal aortic aneurysm formation by inhibiting SMC phenotype switch and inflammasome activation. Microvasc Res. 140:104280. DOI: 10.1016/j.mvr.2021.104280. PMID: 34856183.
Article24. Yao D, He Q, Sun J, Cai L, Wei J, Cai G, Liu J, Lin Y, Wang L, Huang X. 2021; FGF21 attenuates hypoxia-induced dysfunction and inflammation in HPAECs via the microRNA-27b-mediated PPARγ pathway. Int J Mol Med. 47:116. DOI: 10.3892/ijmm.2021.4949. PMID: 33907846. PMCID: PMC8083827.
Article25. Zeng Z, Zheng Q, Chen J, Tan X, Li Q, Ding L, Zhang R, Lin X. 2020; FGF21 mitigates atherosclerosis via inhibition of NLRP3 inflammasome-mediated vascular endothelial cells pyroptosis. Exp Cell Res. 393:112108. DOI: 10.1016/j.yexcr.2020.112108. PMID: 32445748.
Article26. Yuan S, Liang X, He W, Liang M, Jin J, He Q. 2021; ATF4-dependent heme-oxygenase-1 attenuates diabetic nephropathy by inducing autophagy and inhibiting apoptosis in podocyte. Ren Fail. 43:968–979. DOI: 10.1080/0886022X.2021.1936040. PMID: 34157937. PMCID: PMC8231401.
Article27. Feng L, Li M, Hu X, Li Y, Zhu L, Chen M, Wei Q, Xu W, Zhou Q, Wang W, Chen D, Wang X, Jin H. 2021; CK1δ stimulates ubiquitination-dependent proteasomal degradation of ATF4 to promote chemoresistance in gastric Cancer. Clin Transl Med. 11:e587. DOI: 10.1002/ctm2.587. PMID: 34709767. PMCID: PMC8516343. PMID: 0e859d4b9c61470e9f235edfc56ecf03.
Article28. Malabanan KP, Khachigian LM. 2010; Activation transcription factor-4 and the acute vascular response to injury. J Mol Med (Berl). 88:545–552. DOI: 10.1007/s00109-010-0615-4. PMID: 20306012.
Article29. He L, Yuan J, Xu Q, Chen R, Chen L, Fang M. 2016; miRNA-1283 regulates the PERK/ATF4 pathway in vascular injury by targeting ATF4. PLoS One. 11:e0159171. DOI: 10.1371/journal.pone.0159171. PMID: 27537404. PMCID: PMC4990291.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Angiotensin II type 1 receptor antibodies in kidney transplantation
- Kahweol from Coffee Induces Apoptosis by Upregulating Activating Transcription Factor 3 in Human Colorectal Cancer Cells
- The effects on the production of platelet activating factor in the cultured human endothelial cells by interleukin-6 and granulocyte macrophage colony stimulating factor
- The effects of carnitine and platelet activating factor on the motility parameters of human spermatozoa
- Human Leptin Protein Induces Proliferation of A549 Cells via Inhibition of PKR-Like ER Kinase and Activating Transcription Factor-6 Mediated Apoptosis