Korean J Physiol Pharmacol.
2022 Mar;26(2):95-111. 10.4196/kjpp.2022.26.2.95.
Metformin alleviates chronic obstructive pulmonary disease and cigarette smoke extract-induced glucocorticoid resistance by activating the nuclear factor E2-related factor 2/heme oxygenase-1 signaling pathway
- Affiliations
-
- 1College of Pharmacy, Anhui University of Chinese Medicine, Hefei, Anhui 230012, China.
- 2Anhui Province Key Laboratory of Research & Development of Chinese Medicine, Hefei, Anhui 230012, China.
- KMID: 2526729
- DOI: http://doi.org/10.4196/kjpp.2022.26.2.95
Abstract
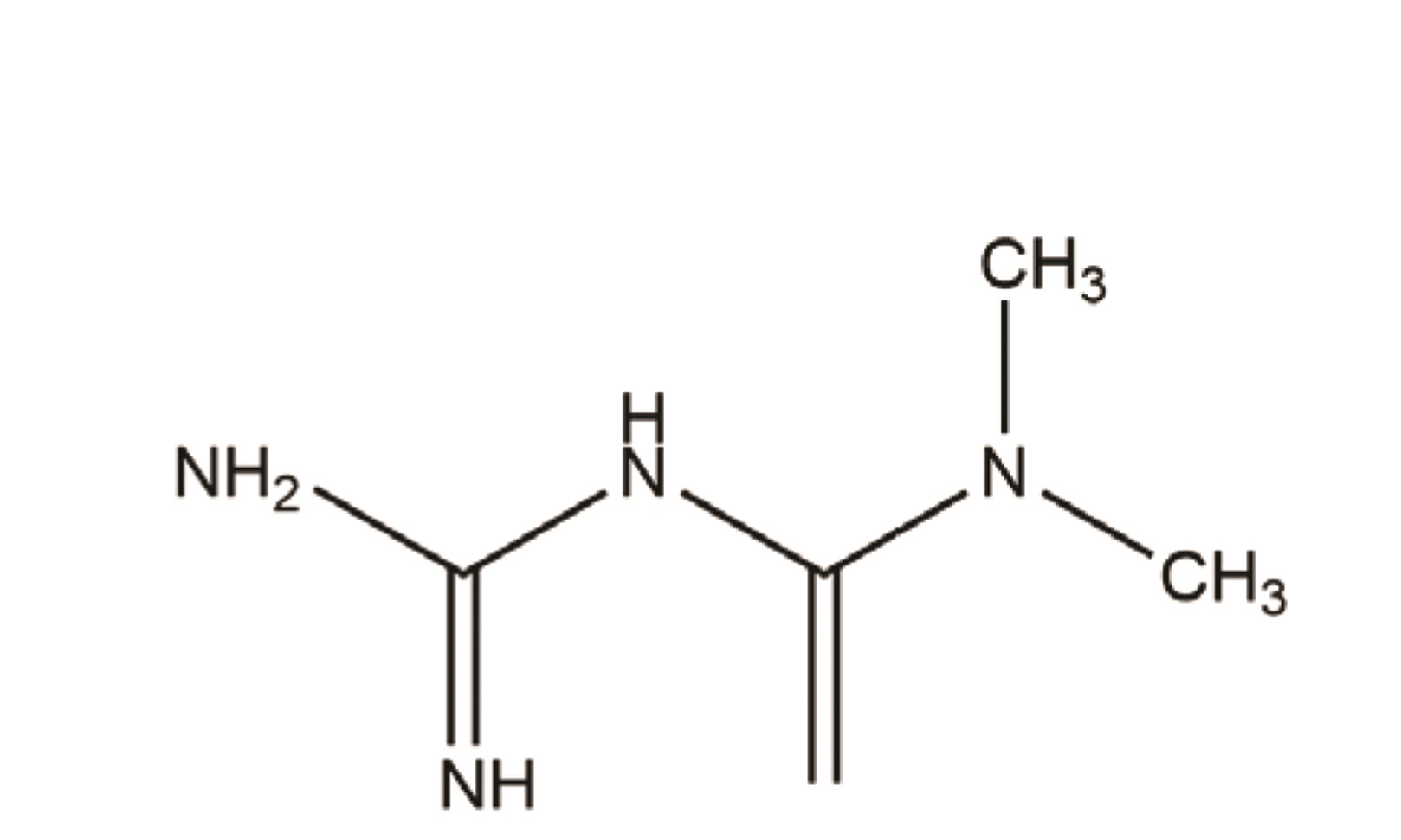
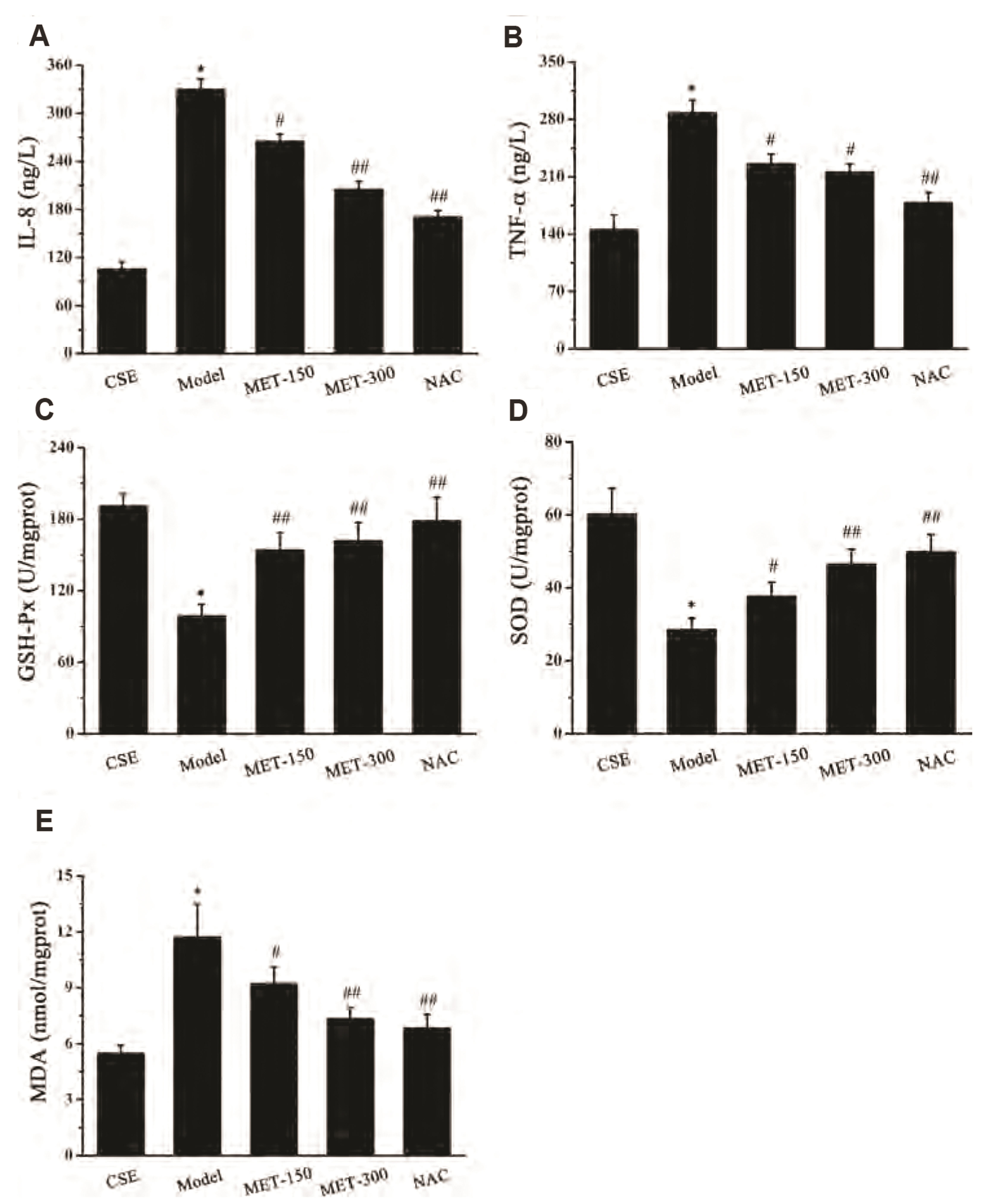
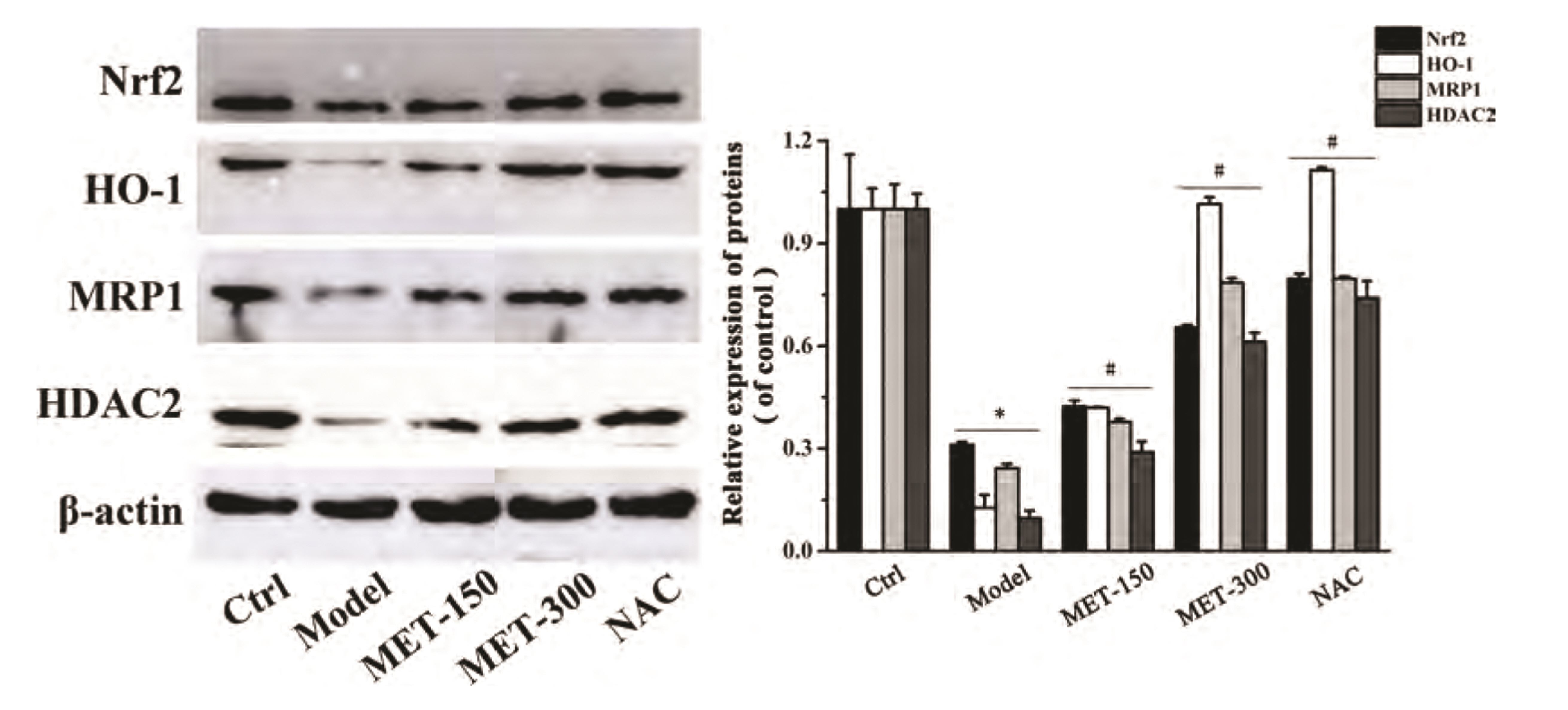
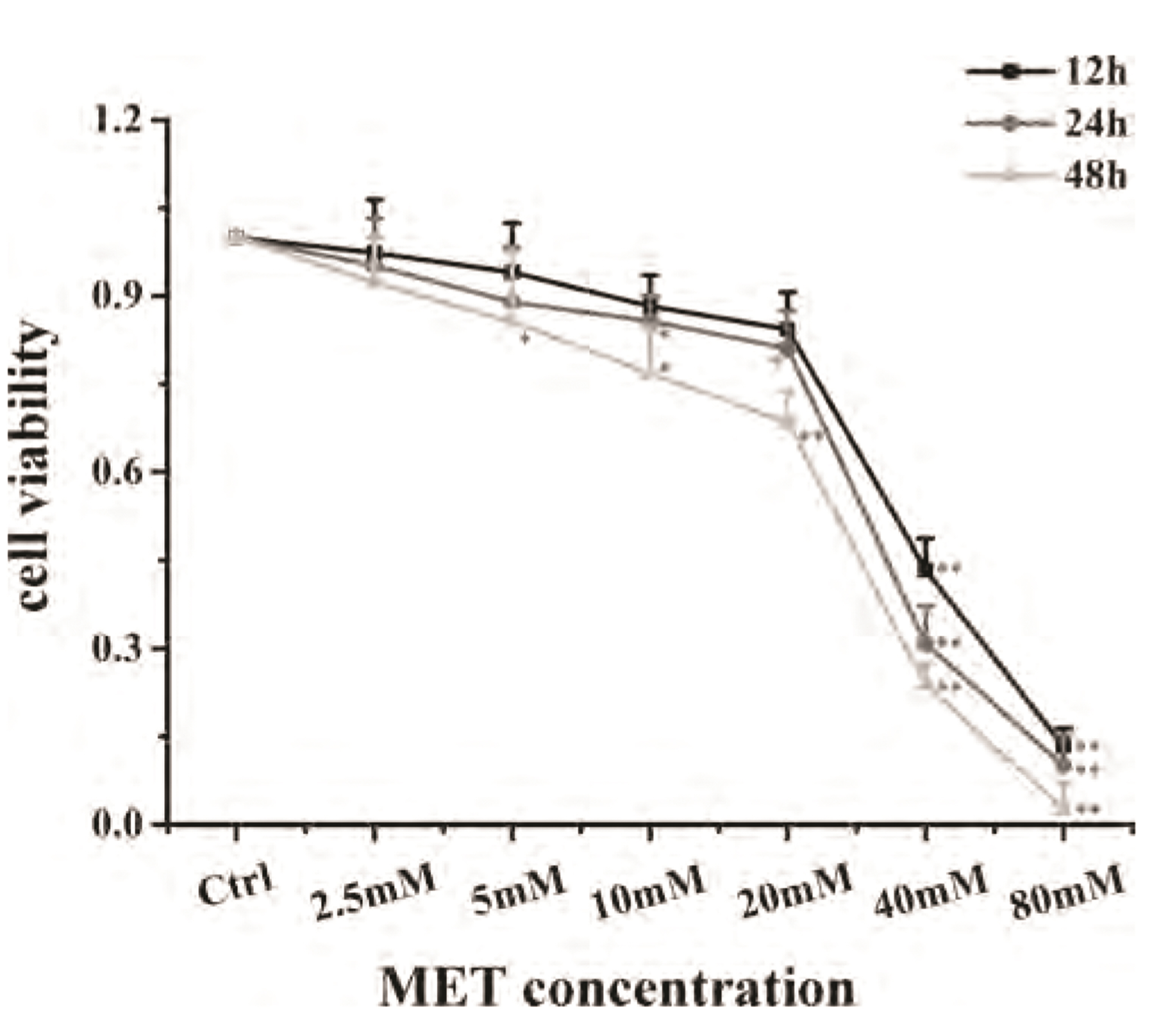
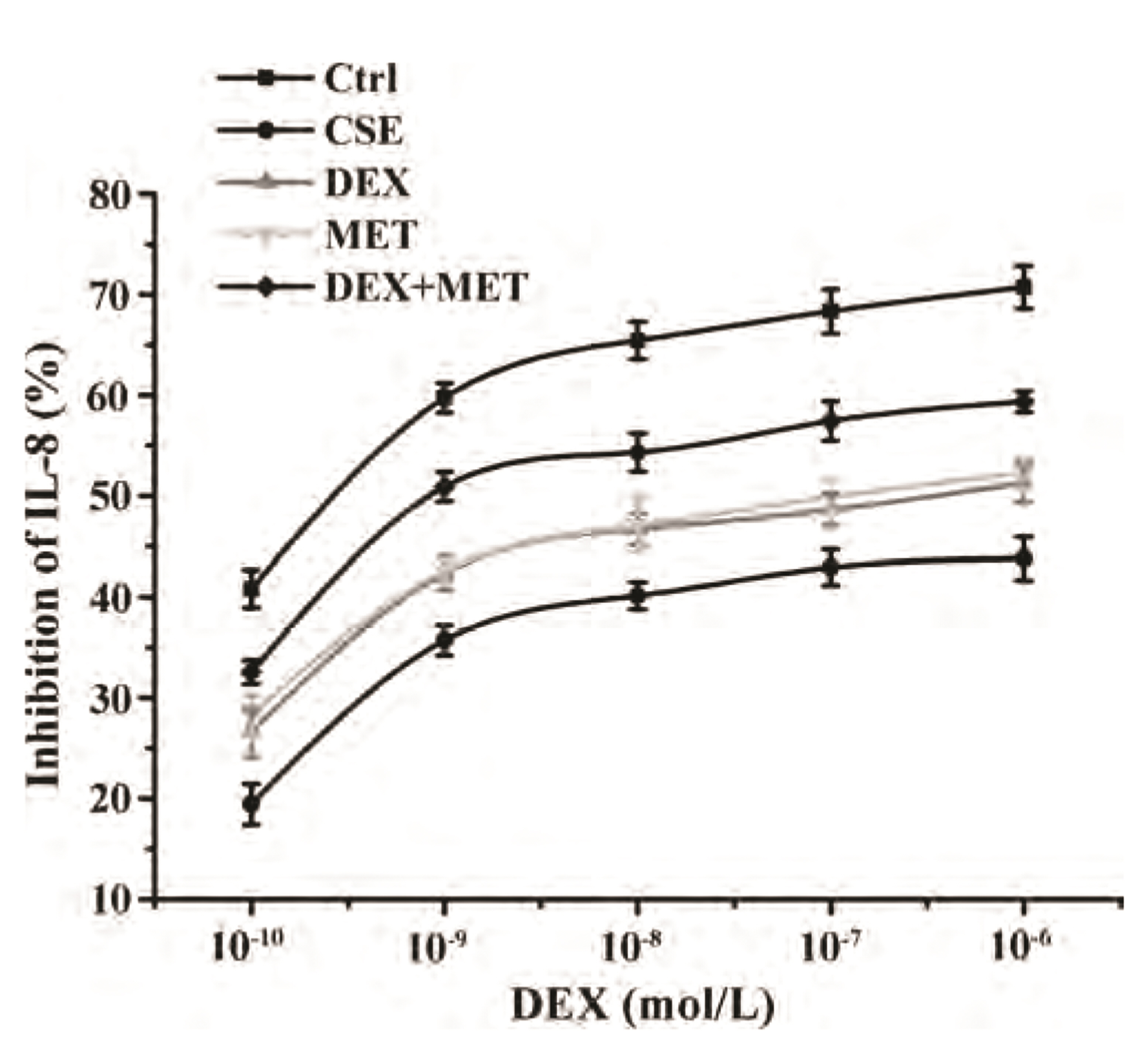
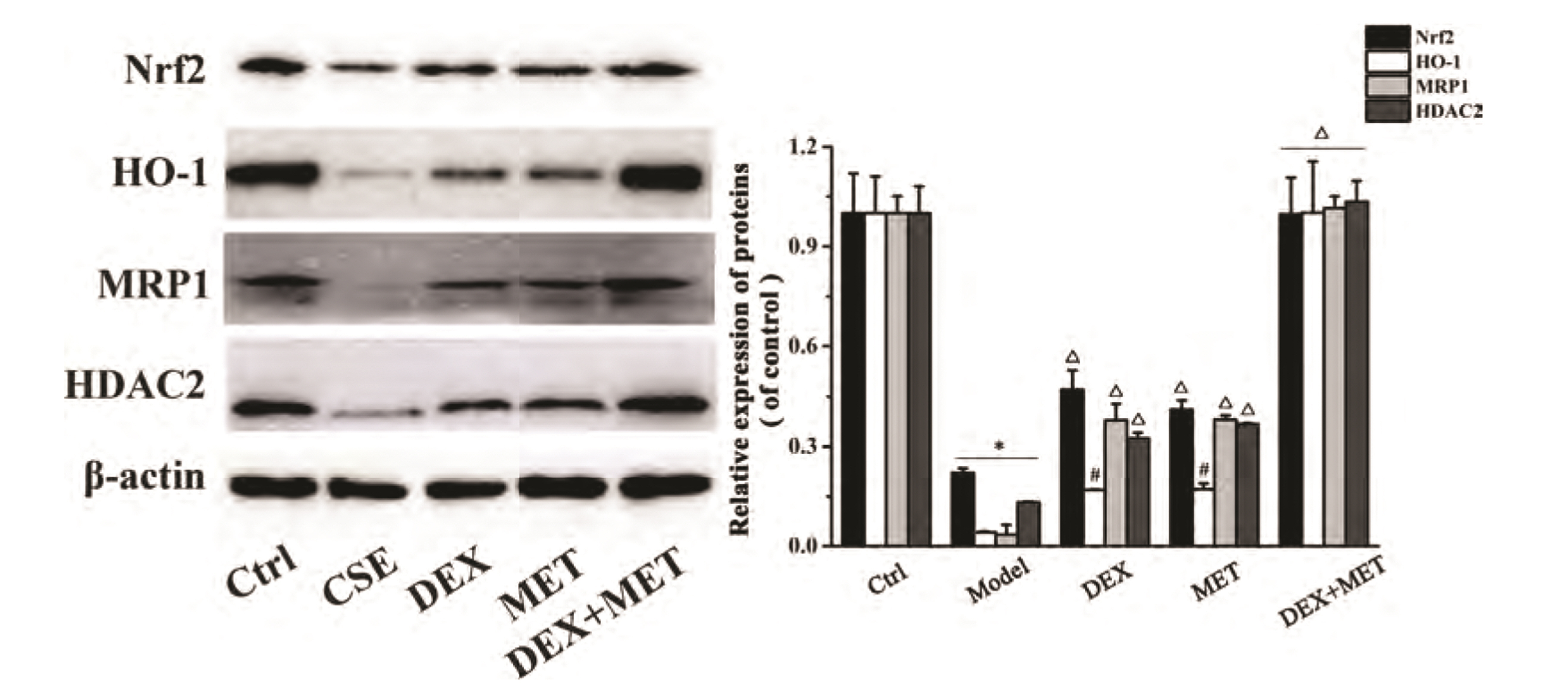
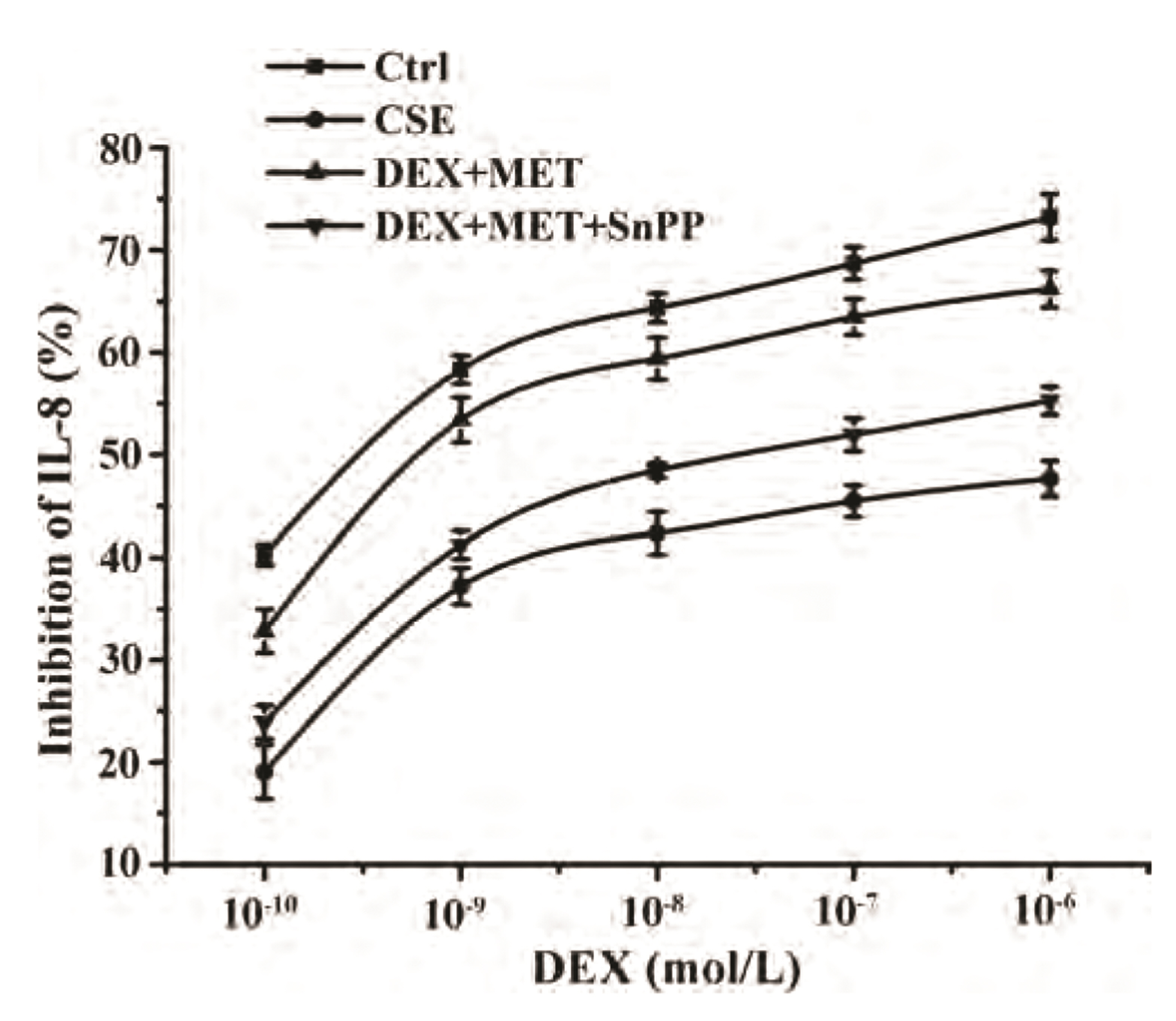
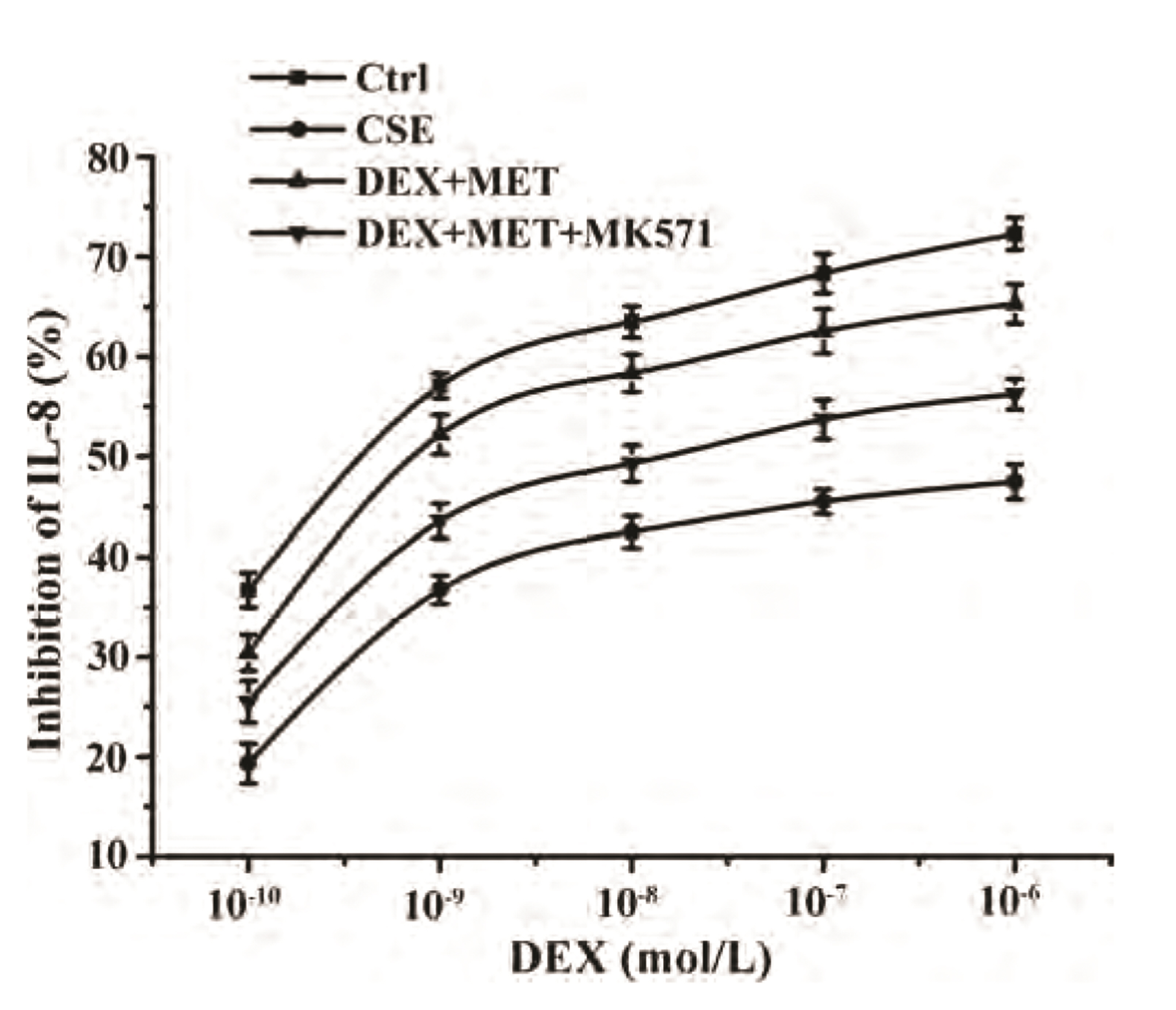
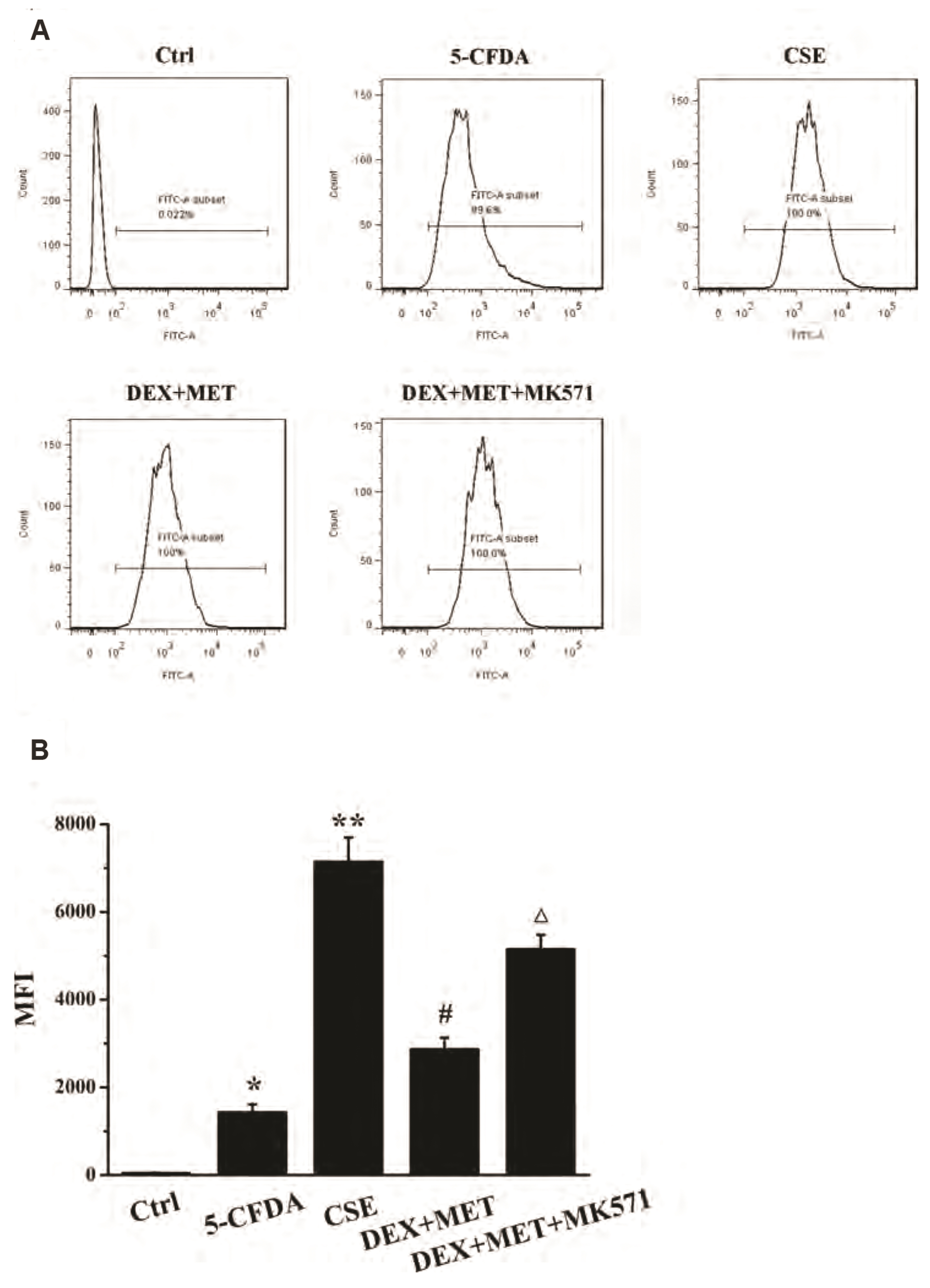
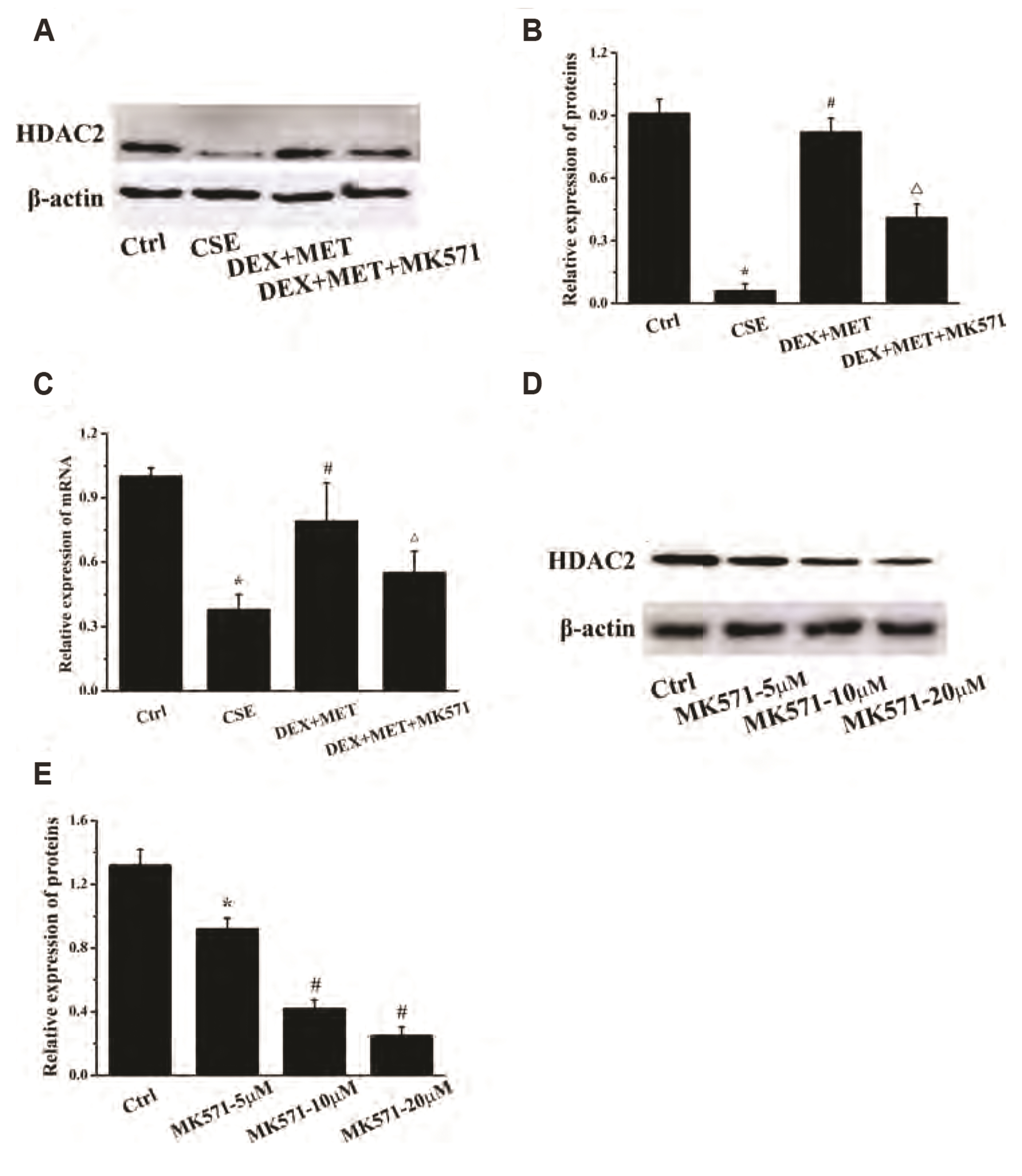
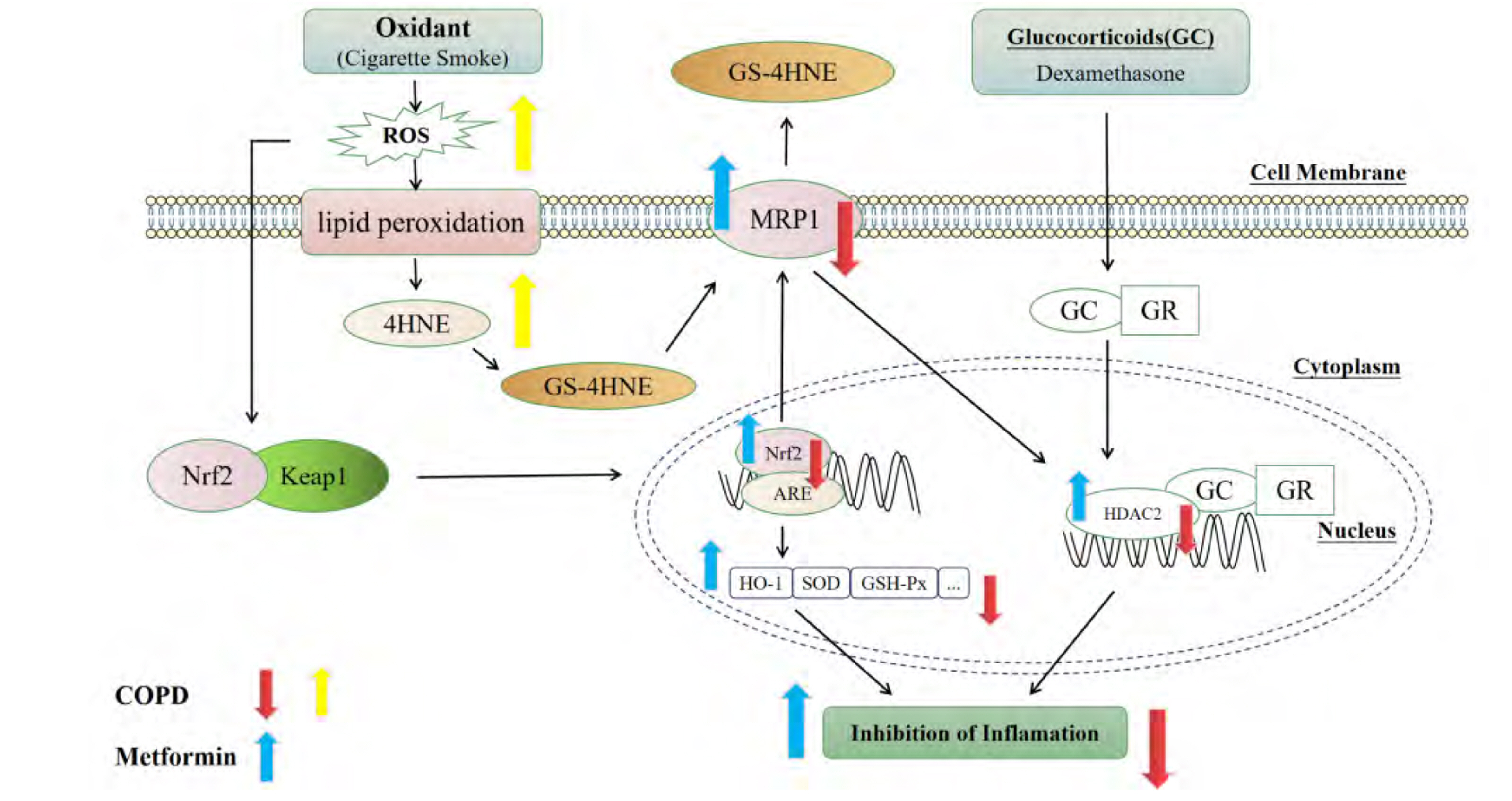
- Chronic obstructive pulmonary disease (COPD) is an important healthcare problem worldwide. Often, glucocorticoid (GC) resistance develops during COPD treatment. As a classic hypoglycemic drug, metformin (MET) can be used as a treatment strategy for COPD due to its anti-inflammatory and antioxidant effects, but its specific mechanism of action is not known. We aimed to clarify the role of MET on COPD and cigarette smoke extract (CSE)-induced GC resistance. Through establishment of a COPD model in rats, we found that MET could improve lung function, reduce pathological injury, as well as reduce the level of inflammation and oxidative stress in COPD, and upregulate expression of nuclear factor E2-related factor 2 (Nrf2), heme oxygenase-1 (HO-1), multidrug resistance protein 1 (MRP1), and histone deacetylase 2 (HDAC2). By establishing a model of GC resistance in human bronchial epithelial cells stimulated by CSE, we found that MET reduced secretion of interleukin-8, and could upregulate expression of Nrf2, HO-1, MRP1, and HDAC2. MET could also increase the inhibition of MRP1 efflux by MK571 significantly, and increase expression of HDAC2 mRNA and protein. In conclusion, MET may upregulate MRP1 expression by activating the Nrf2/HO-1 signaling pathway, and then regulate expression of HDAC2 protein to reduce GC resistance.
Keyword
Figure
Reference
-
1. Lareau SC, Fahy B, Meek P, Wang A. 2019; Chronic obstructive pulmonary disease (COPD). Am J Respir Crit Care Med. 199:P1–P2. DOI: 10.1164/rccm.1991P1. PMID: 30592446.
Article2. Rabe KF, Watz H. 2017; Chronic obstructive pulmonary disease. Lancet. 389:1931–1940. DOI: 10.1016/S0140-6736(17)31222-9. PMID: 22314182. PMCID: PMC7172377.
Article3. Guan R, Wang J, Li Z, Ding M, Li D, Xu G, Wang T, Chen Y, Yang Q, Long Z, Cai Z, Zhang C, Liang X, Dong L, Zhao L, Zhang H, Sun D, Lu W. 2018; Sodium tanshinone IIA sulfonate decreases cigarette smoke-induced inflammation and oxidative stress via blocking the activation of MAPK/HIF-1α signaling pathway. Front Pharmacol. 9:263. DOI: 10.3389/fphar.2018.00263. PMID: 29765317. PMCID: PMC5938387.
Article4. Gong J, Zhao H, Liu T, Li L, Cheng E, Zhi S, Kong L, Yao HW, Li J. 2019; Cigarette smoke reduces fatty acid catabolism, leading to apoptosis in lung endothelial cells: implication for pathogenesis of COPD. Front Pharmacol. 10:941. DOI: 10.3389/fphar.2019.00941. PMID: 31555131. PMCID: PMC6727183.
Article5. Shi K, Chen X, Xie B, Yang SS, Liu D, Dai G, Chen Q. 2018; Celastrol alleviates chronic obstructive pulmonary disease by inhibiting cellular inflammation induced by cigarette smoke via the Ednrb/Kng1 signaling pathway. Front Pharmacol. 9:1276. DOI: 10.3389/fphar.2018.01276. PMID: 30498444. PMCID: PMC6249343.
Article6. Anzalone G, Arcoleo G, Bucchieri F, Montalbano AM, Marchese R, Albano GD, Di Sano C, Moscato M, Gagliardo R, Ricciardolo FLM, Profita M. 2019; Cigarette smoke affects the onco-suppressor DAB2IP expression in bronchial epithelial cells of COPD patients. Sci Rep. 9:15682. DOI: 10.1038/s41598-019-52179-5. PMID: 31666665. PMCID: PMC6821751.
Article7. van der Deen M, de Vries EG, Visserman H, Zandbergen W, Postma DS, Timens W, Timmer-Bosscha H. 2007; Cigarette smoke extract affects functional activity of MRP1 in bronchial epithelial cells. J Biochem Mol Toxicol. 21:243–251. DOI: 10.1002/jbt.20187. PMID: 17912704.
Article8. Sun X, Feng X, Zheng D, Li A, Li C, Li S, Zhao Z. 2019; Ergosterol attenuates cigarette smoke extract-induced COPD by modulating inflammation, oxidative stress and apoptosis in vitro and in vivo. Clin Sci (Lond). 133:1523–1536. DOI: 10.1042/CS20190331. PMID: 31270147.
Article9. Fang X, Wang X, Bai C. 2011; COPD in China: the burden and importance of proper management. Chest. 139:920–929. DOI: 10.1378/chest.10-1393. PMID: 21467059. PMCID: PMC7125604.10. Li N, Nel AE. 2006; Role of the Nrf2-mediated signaling pathway as a negative regulator of inflammation: implications for the impact of particulate pollutants on asthma. Antioxid Redox Signal. 8:88–98. DOI: 10.1089/ars.2006.8.88. PMID: 16487041.
Article11. Lee W, Thomas PS. 2009; Oxidative stress in COPD and its measurement through exhaled breath condensate. Clin Transl Sci. 2:150–155. DOI: 10.1111/j.1752-8062.2009.00093.x. PMID: 20443881. PMCID: PMC5350676.
Article12. López-Campos JL, Soler-Cataluña JJ, Miravitlles M. 2020; Global strategy for the diagnosis, management, and prevention of chronic obstructive lung disease 2019 report: future challenges. Arch Bronconeumol (Engl Ed). 56:65–67. DOI: 10.1016/j.arbres.2019.06.001. PMID: 31320191.
Article13. Barnes PJ. 2017; Glucocorticosteroids. Handb Exp Pharmacol. 237:93–115. DOI: 10.1007/164_2016_98. PMID: 27864677.
Article14. Yang IA, Clarke MS, Sim EH, Fong KM. 2012; Inhaled corticosteroids for stable chronic obstructive pulmonary disease. Cochrane Database Syst Rev. (7):CD002991. DOI: 10.1002/14651858.CD002991.pub3. PMID: 22786484.
Article15. Cosío BG, Jahn A, Iglesias A, Shafiek H, Busquets X, Agustí A. 2015; Haemophilus influenzae induces steroid-resistant inflammatory responses in COPD. BMC Pulm Med. 15:157. DOI: 10.1186/s12890-015-0155-3. PMID: 26642881. PMCID: PMC4672509.
Article16. Matera MG, Calzetta L, Gritti G, Gallo L, Perfetto B, Donnarumma G, Cazzola M, Rogliani P, Donniacuo M, Rinaldi B. 2018; Role of statins and mevalonate pathway on impaired HDAC2 activity induced by oxidative stress in human airway epithelial cells. Eur J Pharmacol. 832:114–119. DOI: 10.1016/j.ejphar.2018.05.023. PMID: 29782855.
Article17. Marwick JA, Ito K, Adcock IM, Kirkham PA. 2007; Oxidative stress and steroid resistance in asthma and COPD: pharmacological manipulation of HDAC-2 as a therapeutic strategy. Expert Opin Ther Targets. 11:745–755. DOI: 10.1517/14728222.11.6.745. PMID: 17504013.
Article18. Biswal S, Thimmulappa RK, Harvey CJ. 2012; Experimental therapeutics of Nrf2 as a target for prevention of bacterial exacerbations in COPD. Proc Am Thorac Soc. 9:47–51. DOI: 10.1513/pats.201201-009MS. PMID: 22550241. PMCID: PMC3359107.
Article19. Pace E, Ferraro M, Di Vincenzo S, Cipollina C, Gerbino S, Cigna D, Caputo V, Balsamo R, Lanata L, Gjomarkaj M. 2013; Comparative cytoprotective effects of carbocysteine and fluticasone propionate in cigarette smoke extract-stimulated bronchial epithelial cells. Cell Stress Chaperones. 18:733–743. DOI: 10.1007/s12192-013-0424-0. PMID: 23580157. PMCID: PMC3789875.
Article20. Huang D, Ma Z, He Y, Xiao Y, Luo H, Liang Q, Zhong X, Bai J, He Z. 2017; Long-term cigarette smoke exposure inhibits histone deacetylase 2 expression and enhances the nuclear factor-κB activation in skeletal muscle of mice. Oncotarget. 8:56726–56736. DOI: 10.18632/oncotarget.18089. PMID: 28915625. PMCID: PMC5593596.
Article21. Meja KK, Rajendrasozhan S, Adenuga D, Biswas SK, Sundar IK, Spooner G, Marwick JA, Chakravarty P, Fletcher D, Whittaker P, Megson IL, Kirkham PA, Rahman I. 2008; Curcumin restores corticosteroid function in monocytes exposed to oxidants by maintaining HDAC2. Am J Respir Cell Mol Biol. 39:312–323. DOI: 10.1165/rcmb.2008-0012OC. PMID: 18421014. PMCID: PMC2542449.
Article22. Yang Y, Sharma R, Sharma A, Awasthi S, Awasthi YC. 2003; Lipid peroxidation and cell cycle signaling: 4-hydroxynonenal, a key molecule in stress mediated signaling. Acta Biochim Pol. 50:319–336. DOI: 10.18388/abp.2003_3689. PMID: 12833161.
Article23. Rahman I, van Schadewijk AA, Crowther AJ, Hiemstra PS, Stolk J, MacNee W, De Boer WI. 2002; 4-Hydroxy-2-nonenal, a specific lipid peroxidation product, is elevated in lungs of patients with chronic obstructive pulmonary disease. Am J Respir Crit Care Med. 166:490–495. DOI: 10.1164/rccm.2110101. PMID: 12186826.
Article24. Rahman I, Marwick J, Kirkham P. 2004; Redox modulation of chromatin remodeling: impact on histone acetylation and deacetylation, NF-kappaB and pro-inflammatory gene expression. Biochem Pharmacol. 68:1255–1267. DOI: 10.1016/j.bcp.2004.05.042. PMID: 15313424.
Article25. Moodie FM, Marwick JA, Anderson CS, Szulakowski P, Biswas SK, Bauter MR, Kilty I, Rahman I. 2004; Oxidative stress and cigarette smoke alter chromatin remodeling but differentially regulate NF-kappaB activation and proinflammatory cytokine release in alveolar epithelial cells. FASEB J. 18:1897–1899. DOI: 10.1096/fj.04-1506fje. PMID: 15456740.
Article26. Li M, Zhong X, He Z, Wen M, Li J, Peng X, Liu G, Deng J, Zhang J, Bai J. 2012; Effect of erythromycin on cigarette-induced histone deacetylase protein expression and nuclear factor-κB activity in human macrophages in vitro. Int Immunopharmacol. 12:643–650. DOI: 10.1016/j.intimp.2011.12.022. PMID: 22265969.
Article27. Marwick JA, Kirkham PA, Stevenson CS, Danahay H, Giddings J, Butler K, Donaldson K, Macnee W, Rahman I. 2004; Cigarette smoke alters chromatin remodeling and induces proinflammatory genes in rat lungs. Am J Respir Cell Mol Biol. 31:633–642. DOI: 10.1165/rcmb.2004-0006OC. PMID: 15333327.
Article28. Fang WF, Chen YM, Lin CY, Huang HL, Yeh H, Chang YT, Huang KT, Lin MC. 2018; Histone deacetylase 2 (HDAC2) attenuates lipopolysaccharide (LPS)-induced inflammation by regulating PAI-1 expression. J Inflamm (Lond). 15:3. DOI: 10.1186/s12950-018-0179-6. PMID: 29344006. PMCID: PMC5763578.
Article29. Malhotra D, Thimmulappa RK, Mercado N, Ito K, Kombairaju P, Kumar S, Ma J, Feller-Kopman D, Wise R, Barnes P, Biswal S. 2011; Denitrosylation of HDAC2 by targeting Nrf2 restores glucocorticosteroid sensitivity in macrophages from COPD patients. J Clin Invest. 121:4289–4302. DOI: 10.1172/JCI45144. PMID: 22005302. PMCID: PMC3204828.
Article30. Ma Q. 2013; Role of nrf2 in oxidative stress and toxicity. Annu Rev Pharmacol Toxicol. 53:401–426. DOI: 10.1146/annurev-pharmtox-011112-140320. PMID: 23294312. PMCID: PMC4680839.
Article31. Yu D, Liu X, Zhang G, Ming Z, Wang T. 2018; Isoliquiritigenin inhibits cigarette smoke-induced COPD by attenuating inflammation and oxidative stress via the regulation of the Nrf2 and NF-κB signaling pathways. Front Pharmacol. 9:1001. DOI: 10.3389/fphar.2018.01001. PMID: 30294270. PMCID: PMC6159750.
Article32. Kundu JK, Surh YJ. 2010; Nrf2-Keap1 signaling as a potential target for chemoprevention of inflammation-associated carcinogenesis. Pharm Res. 27:999–1013. DOI: 10.1007/s11095-010-0096-8. PMID: 20354764.
Article33. Ji L, Li H, Gao P, Shang G, Zhang DD, Zhang N, Jiang T. 2013; Nrf2 pathway regulates multidrug-resistance-associated protein 1 in small cell lung cancer. PLoS One. 8:e63404. DOI: 10.1371/journal.pone.0063404. PMID: 23667609. PMCID: PMC3646742.
Article34. Chen HH, Chen YT, Huang YW, Tsai HJ, Kuo CC. 2012; 4-Ketopinoresinol, a novel naturally occurring ARE activator, induces the Nrf2/HO-1 axis and protects against oxidative stress-induced cell injury via activation of PI3K/AKT signaling. Free Radic Biol Med. 52:1054–1066. DOI: 10.1016/j.freeradbiomed.2011.12.012. PMID: 22245092.
Article35. Burk RF, Hill KE, Nakayama A, Mostert V, Levander XA, Motley AK, Johnson DA, Johnson JA, Freeman ML, Austin LM. 2008; Selenium deficiency activates mouse liver Nrf2-ARE but vitamin E deficiency does not. Free Radic Biol Med. 44:1617–1623. DOI: 10.1016/j.freeradbiomed.2008.01.016. PMID: 18279678. PMCID: PMC2346531.
Article36. Uddin MJ, Kim EH, Hannan MA, Ha H. 2021; Pharmacotherapy against oxidative stress in chronic kidney disease: promising small molecule natural products targeting Nrf2-HO-1 signaling. Antioxidants (Basel). 10:258. DOI: 10.3390/antiox10020258. PMID: 33562389. PMCID: PMC7915495. PMID: 052381c26def48a284a2ce985fa297a5.
Article37. Xu X, Li H, Hou X, Li D, He S, Wan C, Yin P, Liu M, Liu F, Xu J. 2015; Punicalagin induces Nrf2/HO-1 expression via upregulation of PI3K/AKT pathway and inhibits LPS-induced oxidative stress in RAW264.7 macrophages. Mediators Inflamm. 2015:380218. DOI: 10.1155/2015/380218. PMID: 25969626. PMCID: PMC4417599.
Article38. Verbon A, Leemans JC, Weijer S, Florquin S, Van Der Poll T. 2002; Mice lacking the multidrug resistance protein 1 have a transiently impaired immune response during tuberculosis. Clin Exp Immunol. 130:32–36. DOI: 10.1046/j.1365-2249.2002.01952.x. PMID: 12296850. PMCID: PMC1906484.
Article39. Wen X, Iwata K, Ikuta K, Zhang X, Zhu K, Ibi M, Matsumoto M, Asaoka N, Liu J, Katsuyama M, Yabe-Nishimura C. 2019; NOX1/NADPH oxidase regulates the expression of multidrug resistance-associated protein 1 and maintains intracellular glutathione levels. FEBS J. 286:678–687. DOI: 10.1111/febs.14753. PMID: 30653821.
Article40. Jungsuwadee P, Cole MP, Sultana R, Joshi G, Tangpong J, Butterfield DA, St Clair DK, Vore M. 2006; Increase in Mrp1 expression and 4-hydroxy-2-nonenal adduction in heart tissue of Adriamycin-treated C57BL/6 mice. Mol Cancer Ther. 5:2851–2860. DOI: 10.1158/1535-7163.MCT-06-0297. PMID: 17121932.
Article41. Yoshioka M, Sagara H, Takahashi F, Harada N, Nishio K, Mori A, Ushio H, Shimizu K, Okada T, Ota M, Ito YM, Nagashima O, Atsuta R, Suzuki T, Fukuda T, Fukuchi Y, Takahashi K. 2009; Role of multidrug resistance-associated protein 1 in the pathogenesis of allergic airway inflammation. Am J Physiol Lung Cell Mol Physiol. 296:L30–L36. DOI: 10.1152/ajplung.00026.2008. PMID: 18931056.
Article42. Wang DL, Wang CY, Cao Y, Zhang X, Tao XH, Yang LL, Chen JP, Wang SS, Li ZG. 2014; Allyl isothiocyanate increases MRP1 function and expression in a human bronchial epithelial cell line. Oxid Med Cell Longev. 2014:547379. DOI: 10.1155/2014/547379. PMID: 24672635. PMCID: PMC3942196.
Article43. Zhou Y, Xu X, Wu J, Xu L, Zhang M, Li Z, Wang D. 2020; Allyl isothiocyanate treatment alleviates chronic obstructive pulmonary disease through the Nrf2-Notch1 signaling and upregulation of MRP1. Life Sci. 243:117291. DOI: 10.1016/j.lfs.2020.117291. PMID: 31927049.
Article44. Li M, Hu X, Xu Y, Hu X, Zhang C, Pang S. 2019; A possible mechanism of metformin in improving insulin resistance in diabetic rat models. Int J Endocrinol. 2019:3248527. DOI: 10.1155/2019/3248527. PMID: 31737069. PMCID: PMC6815615.
Article45. Madka V, Kumar G, Pathuri G, Zhang Y, Lightfoot S, Asch AS, Mohammed A, Steele VE, Rao CV. 2020; Bisphosphonates zometa and fosamax synergize with metformin to prevent AOM-induced colon cancer in F344 rat model. Cancer Prev Res (Phila). 13:185–194. DOI: 10.1158/1940-6207.CAPR-19-0265. PMID: 31699708. PMCID: PMC7007371.
Article46. Han Y, Yuan F, Deng C, He F, Zhang Y, Shen H, Chen Z, Qian L. 2019; Metformin decreases LPS-induced inflammatory response in rabbit annulus fibrosus stem/progenitor cells by blocking HMGB1 release. Aging (Albany NY). 11:10252–10265. DOI: 10.18632/aging.102453. PMID: 31772144. PMCID: PMC6914423.
Article47. Wu MC, Ye WR, Zheng YJ, Zhang SS. 2017; Oxamate enhances the anti-inflammatory and insulin-sensitizing effects of metformin in diabetic mice. Pharmacology. 100:218–228. DOI: 10.1159/000478909. PMID: 28728159.
Article48. Koh SJ, Kim JM, Kim IK, Ko SH, Kim JS. 2014; Anti-inflammatory mechanism of metformin and its effects in intestinal inflammation and colitis-associated colon cancer. J Gastroenterol Hepatol. 29:502–510. DOI: 10.1111/jgh.12435. PMID: 24716225.
Article49. Soberanes S, Misharin AV, Jairaman A, Morales-Nebreda L, McQuattie-Pimentel AC, Cho T, Hamanaka RB, Meliton AY, Reyfman PA, Walter JM, Chen CI, Chi M, Chiu S, Gonzalez-Gonzalez FJ, Antalek M, Abdala-Valencia H, Chiarella SE, Sun KA, Woods PS, Ghio AJ, et al. 2019; Metformin targets mitochondrial electron transport to reduce air-pollution-induced thrombosis. Cell Metab. 29:335–347.e5. DOI: 10.1016/j.cmet.2018.09.019. PMID: 30318339. PMCID: PMC6365216.
Article50. Vangaveti S, Das P, Kumar VL. 2021; Metformin and silymarin afford protection in cyclosporine A induced hepatorenal toxicity in rat by modulating redox status and inflammation. J Biochem Mol Toxicol. 35:e22614. DOI: 10.1002/jbt.22614. PMID: 32886845.
Article51. Quaile MP, Melich DH, Jordan HL, Nold JB, Chism JP, Polli JW, Smith GA, Rhodes MC. 2010; Toxicity and toxicokinetics of metformin in rats. Toxicol Appl Pharmacol. 243:340–347. DOI: 10.1016/j.taap.2009.11.026. PMID: 20004680.
Article52. Pandey A, Verma S, Kumar VL. 2017; Metformin maintains mucosal integrity in experimental model of colitis by inhibiting oxidative stress and pro-inflammatory signaling. Biomed Pharmacother. 94:1121–1128. DOI: 10.1016/j.biopha.2017.08.020. PMID: 28821163.
Article53. Sanguinetti CM. 2016; N-acetylcysteine in COPD: why, how, and when? Multidiscip Respir Med. 11:8. DOI: 10.4081/mrm.2016.263. PMID: 26855777. PMCID: PMC4744393.
Article54. Sadowska AM, Manuel-Y-Keenoy B, De Backer WA. 2007; Antioxidant and anti-inflammatory efficacy of NAC in the treatment of COPD: discordant in vitro and in vivo dose-effects: a review. Pulm Pharmacol Ther. 20:9–22. DOI: 10.1016/j.pupt.2005.12.007. PMID: 16458553.
Article55. Cai S, Chen P, Zhang C, Chen JB, Wu J. 2009; Oral N-acetylcysteine attenuates pulmonary emphysema and alveolar septal cell apoptosis in smoking-induced COPD in rats. Respirology. 14:354–359. DOI: 10.1111/j.1440-1843.2009.01511.x. PMID: 19341424.
Article56. Xu L, Wu J, Li N, Jiang C, Guo Y, Cao P, Wang D. 2020; AITC induces MRP1 expression by protecting against CS/CSE-mediated DJ-1 protein degradation via activation of the DJ-1/Nrf2 axis. Korean J Physiol Pharmacol. 24:481–492. DOI: 10.4196/kjpp.2020.24.6.481. PMID: 33093270. PMCID: PMC7585591.
Article57. Gueders MM, Bertholet P, Perin F, Rocks N, Maree R, Botta V, Louis R, Foidart JM, Noel A, Evrard B, Cataldo DD. 2008; A novel formulation of inhaled doxycycline reduces allergen-induced inflammation, hyperresponsiveness and remodeling by matrix metalloproteinases and cytokines modulation in a mouse model of asthma. Biochem Pharmacol. 75:514–526. DOI: 10.1016/j.bcp.2007.09.012. PMID: 17950252.
Article58. Chillappagari S, Venkatesan S, Garapati V, Mahavadi P, Munder A, Seubert A, Sarode G, Guenther A, Schmeck BT, Tümmler B, Henke MO. 2014; Impaired TLR4 and HIF expression in cystic fibrosis bronchial epithelial cells downregulates hemeoxygenase-1 and alters iron homeostasis in vitro. Am J Physiol Lung Cell Mol Physiol. 307:L791–L799. DOI: 10.1152/ajplung.00167.2014. PMID: 25239913.
Article59. Xu Y, Liu H, Song L. 2020; Novel drug delivery systems targeting oxidative stress in chronic obstructive pulmonary disease: a review. J Nanobiotechnology. 18:145. DOI: 10.1186/s12951-020-00703-5. PMID: 33076918. PMCID: PMC7570055. PMID: 991ca13e1cf94dfc8513666fc43d95cf.
Article60. Agustí A, Hogg JC. 2019; Update on the pathogenesis of chronic obstructive pulmonary disease. N Engl J Med. 381:1248–1256. DOI: 10.1056/NEJMra1900475. PMID: 31553836.
Article61. Liu L, Shang Y, Li M, Han X, Wang J, Wang J. 2015; Curcumin ameliorates asthmatic airway inflammation by activating nuclear factor-E2-related factor 2/haem oxygenase (HO)-1 signalling pathway. Clin Exp Pharmacol Physiol. 42:520–529. DOI: 10.1111/1440-1681.12384. PMID: 25739561.
Article62. Ruiz S, Pergola PE, Zager RA, Vaziri ND. 2013; Targeting the transcription factor Nrf2 to ameliorate oxidative stress and inflammation in chronic kidney disease. Kidney Int. 83:1029–1041. DOI: 10.1038/ki.2012.439. PMID: 23325084. PMCID: PMC3633725.
Article63. Cheng L, Li F, Ma R, Hu X. 2015; Forsythiaside inhibits cigarette smoke-induced lung inflammation by activation of Nrf2 and inhibition of NF-κB. Int Immunopharmacol. 28:494–499. DOI: 10.1016/j.intimp.2015.07.011. PMID: 26209933.
Article64. Shanmugam T, Selvaraj M, Poomalai S. 2016; Epigallocatechin gallate potentially abrogates fluoride induced lung oxidative stress, inflammation via Nrf2/Keap1 signaling pathway in rats: an in-vivo and in-silico study. Int Immunopharmacol. 39:128–139. DOI: 10.1016/j.intimp.2016.07.022. PMID: 27472294.
Article65. Singla E, Puri G, Dharwal V, Naura AS. 2021; Gallic acid ameliorates COPD-associated exacerbation in mice. Mol Cell Biochem. 476:293–302. DOI: 10.1007/s11010-020-03905-5. PMID: 32965595.
Article66. Cui W, Zhang Z, Zhang P, Qu J, Zheng C, Mo X, Zhou W, Xu L, Yao H, Gao J. 2018; Nrf2 attenuates inflammatory response in COPD/emphysema: crosstalk with Wnt3a/β-catenin and AMPK pathways. J Cell Mol Med. 22:3514–3525. DOI: 10.1111/jcmm.13628. PMID: 29659176. PMCID: PMC6010849.
Article67. Marwick JA, Chung KF. 2010; Glucocorticoid insensitivity as a future target of therapy for chronic obstructive pulmonary disease. Int J Chron Obstruct Pulmon Dis. 5:297–309. DOI: 10.2147/COPD.S7390. PMID: 20856829. PMCID: PMC2939685.
Article68. Chung KF, Marwick JA. 2010; Molecular mechanisms of oxidative stress in airways and lungs with reference to asthma and chronic obstructive pulmonary disease. Ann N Y Acad Sci. 1203:85–91. DOI: 10.1111/j.1749-6632.2010.05600.x. PMID: 20716288.
Article69. Fujita Y, Inagaki N. 2017; Metformin: new preparations and nonglycemic benefits. Curr Diab Rep. 17:5. DOI: 10.1007/s11892-017-0829-8. PMID: 28116648.
Article70. Park CS, Bang BR, Kwon HS, Moon KA, Kim TB, Lee KY, Moon HB, Cho YS. 2012; Metformin reduces airway inflammation and remodeling via activation of AMP-activated protein kinase. Biochem Pharmacol. 84:1660–1670. DOI: 10.1016/j.bcp.2012.09.025. PMID: 23041647.
Article71. Gao J, Yuan J, Wang Q, Lei T, Shen X, Cui B, Zhang F, Ding W, Lu Z. 2020; Metformin protects against PM2.5-induced lung injury and cardiac dysfunction independent of AMP-activated protein kinase α2. Redox Biol. 28:101345. DOI: 10.1016/j.redox.2019.101345. PMID: 31669973. PMCID: PMC6838896.72. Chen CZ, Hsu CH, Li CY, Hsiue TR. 2017; Insulin use increases risk of asthma but metformin use reduces the risk in patients with diabetes in a Taiwanese population cohort. J Asthma. 54:1019–1025. DOI: 10.1080/02770903.2017.1283698. PMID: 28135899.
Article73. Forno E. 2016; Asthma and diabetes: does treatment with metformin improve asthma? Respirology. 21:1144–1145. DOI: 10.1111/resp.12869. PMID: 27533627. PMCID: PMC5021626.
Article74. Al Faraj A, Sultana Shaik A, Pureza MA, Alnafea M, Halwani R. 2014; Preferential macrophage recruitment and polarization in LPS-induced animal model for COPD: noninvasive tracking using MRI. PLoS One. 9:e90829. DOI: 10.1371/journal.pone.0090829. PMID: 24598763. PMCID: PMC3945006.
Article75. Song L, Guan XJ, Chen X, Cui ZL, Han FF, Guo XJ, Xu WG. 2014; Mesenchymal stem cells reduce cigarette smoke-induced inflammation and airflow obstruction in rats via TGF-β1 signaling. COPD. 11:582–590. DOI: 10.3109/15412555.2014.898032. PMID: 24766333.
Article76. Zeng YY, Hu WP, Zuo YH, Wang XR, Zhang J. 2019; Altered serum levels of type I collagen turnover indicators accompanied by IL-6 and IL-8 release in stable COPD. Int J Chron Obstruct Pulmon Dis. 14:163–168. DOI: 10.2147/COPD.S188139. PMID: 30655663. PMCID: PMC6322508.
Article77. Huang AX, Lu LW, Liu WJ, Huang M. 2016; Plasma inflammatory cytokine IL-4, IL-8, IL-10, and TNF-α levels correlate with pulmonary function in patients with asthma-chronic obstructive pulmonary disease (COPD) overlap syndrome. Med Sci Monit. 22:2800–2808. DOI: 10.12659/MSM.896458. PMID: 27501772. PMCID: PMC4982526.
Article78. Luo JF, Shen XY, Lio CK, Dai Y, Cheng CS, Liu JX, Yao YD, Yu Y, Xie Y, Luo P, Yao XS, Liu ZQ, Zhou H. 2018; Activation of Nrf2/HO-1 pathway by nardochinoid C inhibits inflammation and oxidative stress in lipopolysaccharide-stimulated macrophages. Front Pharmacol. 9:911. DOI: 10.3389/fphar.2018.00911. PMID: 30233360. PMCID: PMC6131578.
Article79. Chen X, Su W, Wan T, Yu J, Zhu W, Tang F, Liu G, Olsen N, Liang D, Zheng SG. 2017; Sodium butyrate regulates Th17/Treg cell balance to ameliorate uveitis via the Nrf2/HO-1 pathway. Biochem Pharmacol. 142:111–119. DOI: 10.1016/j.bcp.2017.06.136. PMID: 28684304.
Article80. Adenuga D, Caito S, Yao H, Sundar IK, Hwang JW, Chung S, Rahman I. 2010; Nrf2 deficiency influences susceptibility to steroid resistance via HDAC2 reduction. Biochem Biophys Res Commun. 403:452–456. DOI: 10.1016/j.bbrc.2010.11.054. PMID: 21094147. PMCID: PMC3031165.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Heme Oxygenase-1: Its Therapeutic Roles in Inflammatory Diseases
- Downregulation of Reactive Oxygen Species in Apoptosis
- Four active monomers from Moutan Cortex exert inhibitoryeffects against oxidative stress by activating Nrf2/Keap1signaling pathway
- Role of Nuclear Factor Erythroid 2-Related Factor 2 in Chronic Obstructive Pulmonary Disease
- SiO2 Nanoparticles Induced Cytotoxicity by Oxidative Stress in Human Bronchial Epithelial Cell, Beas-2B