Ann Pediatr Endocrinol Metab.
2021 Dec;26(4):242-251. 10.6065/apem.2040258.129.
Association of fibroblast growth factor 21 with metabolic syndrome and endothelial function in children: a prospective cross-sectional study on novel biomarkers
- Affiliations
-
- 1Child Health Department, Faculty of Medicine, School of Health Sciences, University of Ioannina, Ioannina, Greece
- 2Second Department of Cardiology, Faculty of Medicine, School of Health Sciences, University of Ioannina, Ioannina, Greece
- 3Department of Endocrinology, Faculty of Medicine, School of Health Sciences, University of Ioannina, Ioannina, Greece
- KMID: 2523830
- DOI: http://doi.org/10.6065/apem.2040258.129
Abstract
- Purpose
Metabolic and cardiovascular disease prevention starting in childhood is critical for reducing morbidity later in life. In the present study, the association of novel biomarkers with metabolic syndrome (MS) and vascular function/structure indices of early atherosclerosis in children was investigated.
Methods
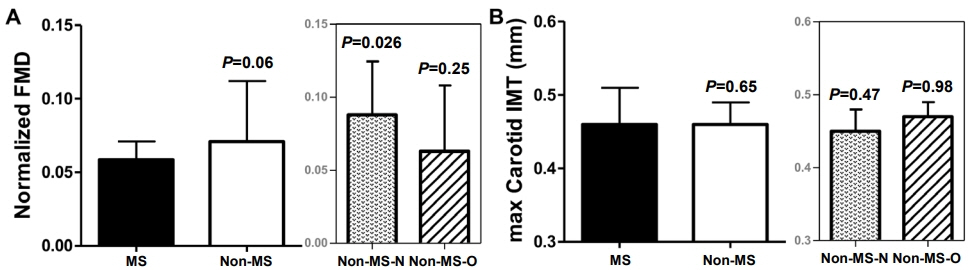
This was a prospective study of 78 children (8–16 years of age) grouped based on the presence or absence of MS. The serum biomarkers investigated included fibroblast growth factor 21 (FGF21), leptin, adiponectin, and insulinlike growth factor binding protein-1 (IGFBP1). Endothelial function and carotid atherosclerosis were assessed based on brachial artery flow-mediated dilation (FMD) and carotid intima-media thickness, respectively.
Results
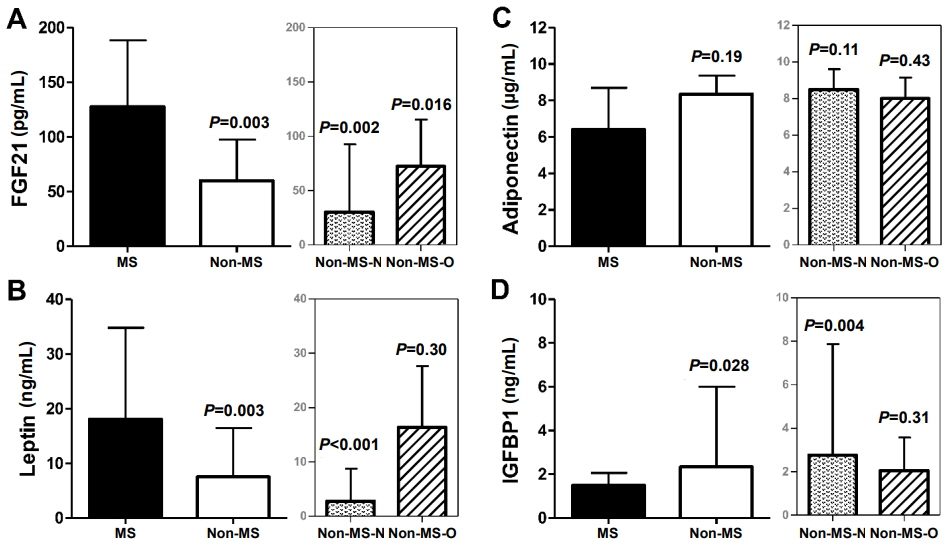
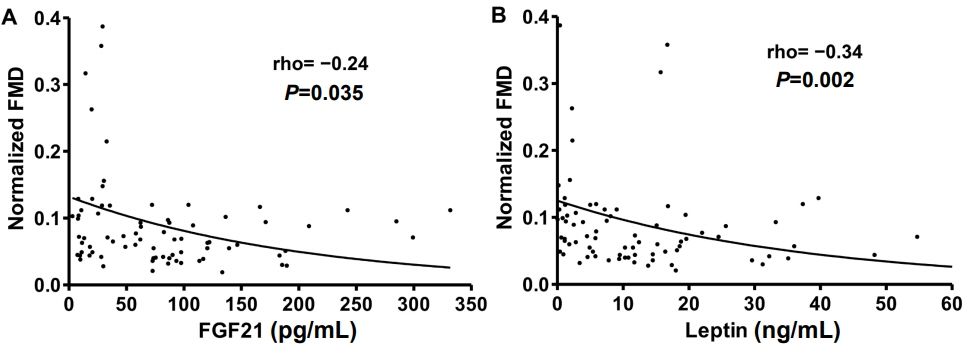
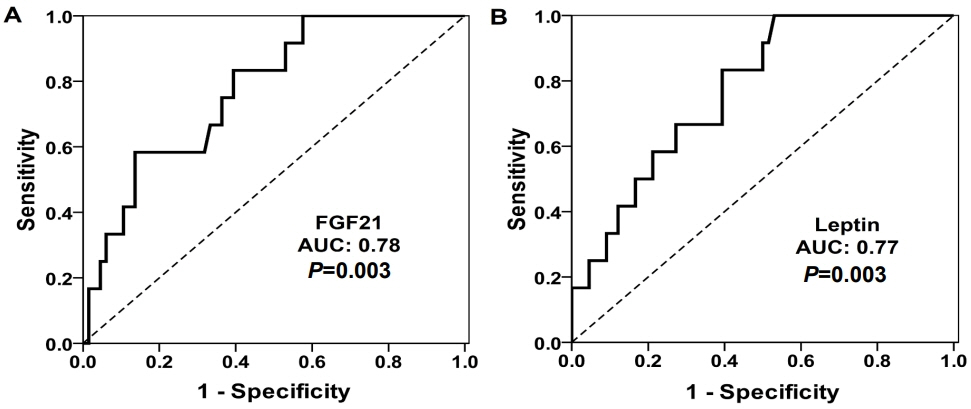
Children with MS (n=12) had higher levels of FGF21 (median [interquartile range]: 128 [76–189] pg/mL vs. 60 [20–98] pg/mL, P=0.003) and leptin (18.1 [11–34.8] pg/mL vs. 7.5 [1.9–16.5] ng/mL, P=0.003), and lower levels of IGFBP1 (1.5 [1.2–2.1] ng/mL vs. 2.3 [1.5–6] ng/mL, P=0.028) compared with children without MS. FMD inversely correlated with FGF21 (Spearman rho= -0.24, P=0.035) and leptin (rho= -0.24, P=0.002) in all children. The best cutoff value of FGF21 levels for MS diagnosis was above 121.3 pg/mL (sensitivity/specificity, 58/86%). Only FGF21 was significantly associated with the presence of MS after adjustment for body mass index, age, and sex (odds ratio per 10 pg/mL increase: 1.10 [95% confidence interval, 1.01–1.22]; P=0.043).
Conclusion
Increased FGF21 levels were associated with the presence of MS and worse endothelial function in children. Larger studies are needed to evaluate the potential value of FGF21 as a biomarker that could predict future metabolic/cardiovascular disease at an early stage.
Keyword
Figure
Cited by 1 articles
-
Relationship Between the Serum FGF21 Level and Growth in Children of Short Stature
Yun Jeong Lee, So Yoon Jung, Young Ah Lee, Jaehyun Kim, Seong Yong Lee, Choong Ho Shin
J Korean Med Sci. 2023;38(7):e63. doi: 10.3346/jkms.2023.38.e63.
Reference
-
References
1. Fon Tacer K, Bookout AL, Ding X, Kurosu H, John GB, Wang L, et al. Research resource: comprehensive expression atlas of the fibroblast growth factor system in adult mouse. Mol Endocrinol. 2010; 24:2050–64.
Article2. Markan KR, Naber MC, Ameka MK, Anderegg MD, Mangelsdorf DJ, Kliewer SA, et al. Circulating FGF21 is liver derived and enhances glucose uptake during refeeding and overfeeding. Diabetes. 2014; 63:4057–63.
Article3. Ogawa Y, Kurosu H, Yamamoto M, Nandi A, Rosenblatt KP, Goetz R, et al. BetaKlotho is required for metabolic activity of fibroblast growth factor 21. Proc Natl Acad Sci U S A. 2007; 104:7432–7.4. Kurosu H, Choi M, Ogawa Y, Dickson AS, Goetz R, Eliseenkova AV, et al. Tissue-specific expression of betaKlotho and fibroblast growth factor (FGF) receptor isoforms determines metabolic activity of FGF19 and FGF21. J Biol Chem. 2007; 282:26687–95.5. Domouzoglou EM, Naka KK, Vlahos AP, Papafaklis MI, Michalis LK, Tsatsoulis A, et al. Fibroblast growth factors in cardiovascular disease: The emerging role of FGF21. Am J Physiol Heart Circ Physiol. 2015; 309:H1029–38.
Article6. Fisher FM, Chui PC, Antonellis PJ, Bina HA, Kharitonenkov A, Flier JS, et al. Obesity is a fibroblast growth factor 21 (FGF21)-resistant state. Diabetes. 2010; 59:2781–9.
Article7. Diez JJ, Iglesias P. The role of the novel adipocyte-derived hormone adiponectin in human disease. Eur J Endocrinol. 2003; 148:293–300.
Article8. El-Haschimi K, Pierroz DD, Hileman SM, Bjorbaek C, Flier JS. Two defects contribute to hypothalamic leptin resistance in mice with diet-induced obesity. J Clin Invest. 2000; 105:1827–32.
Article9. Lewitt MS, Hilding A, Ostenson CG, Efendic S, Brismar K, Hall K. Insulin-like growth factor-binding protein-1 in the prediction and development of type 2 diabetes in middle-aged Swedish men. Diabetologia. 2008; 51:1135–45.
Article10. Zimmet P, Alberti KG, Kaufman F, Tajima N, Silink M, Arslanian S, et al. The metabolic syndrome in children and adolescents - an IDF consensus report. Pediatr Diabetes. 2007; 8:299–306.
Article11. Kelly AS, Steinberger J, Jacobs DR, Hong CP, Moran A, Sinaiko AR. Predicting cardiovascular risk in young adulthood from the metabolic syndrome, its component risk factors, and a cluster score in childhood. Int J Pediatr Obes. 2011; 6:e283–9.
Article12. Matthews DR, Hosker JP, Rudenski AS, Naylor BA, Treacher DF, Turner RC. Homeostasis model assessment: insulin resistance and beta-cell function from fasting plasma glucose and insulin concentrations in man. Diabetologia. 1985; 28:412–9.
Article13. Vlahos AP, Theocharis P, Bechlioulis A, Naka KK, Vakalis K, Papamichael ND, et al. Changes in vascular function and structure in juvenile idiopathic arthritis. Arthritis Care Res (Hoboken). 2011; 63:1736–44.
Article14. Urbina EM, Williams RV, Alpert BS, Collins RT, Daniels SR, Hayman L, et al. Noninvasive assessment of subclinical atherosclerosis in children and adolescents: recommendations for standard assessment for clinical research: a scientific statement from the American Heart Association. Hypertension. 2009; 54:919–50.
Article15. P yke KE, Tschakovsky ME. Peak vs. total reactive hyperemia: which determines the magnitude of flow-mediated dilation? J Appl Physiol (1985). 2007; 102:1510–9.
Article16. Goodman E, Daniels SR, Morrison JA, Huang B, Dolan LM. Contrasting prevalence of and demographic disparities in the World Health Organization and National Cholesterol Education Program Adult Treatment Panel III definitions of metabolic syndrome among adolescents. J Pediatr. 2004; 145:445–51.
Article17. Papoutsakis C, Yannakoulia M, Ntalla I, Dedoussis GV. Metabolic syndrome in a Mediterranean pediatric cohort: prevalence using International Diabetes Federation-derived criteria and associations with adiponectin and leptin. Metabolism. 2012; 61:140–5.
Article18. Kim HR, Han MA. Association between serum liver enzymes and metabolic syndrome in Korean adults. Int J Environ Res Public Health. 2018; 15:1658.
Article19. Lee S, Bacha F, Gungor N, Arslanian SA. Waist circumference is an independent predictor of insulin resistance in black and white youths. J Pediatr. 2006; 148:188–94.
Article20. Bremer AA, Mietus-Snyder M, Lustig RH. Toward a unifying hypothesis of metabolic syndrome. Pediatrics. 2012; 129:557–70.
Article21. Alemzadeh R, Kichler J. Comparison of apolipoprotein (ApoB/ApoA-1) and lipoprotein (total cholesterol/HDL) ratios in obese adolescents. Metab Syndr Relat Disord. 2018; 16:40–5.
Article22. Friedman JM, Halaas JL. Leptin and the regulation of body weight in mammals. Nature. 1998; 395:763–70.
Article23. Badman MK, Pissios P, Kennedy AR, Koukos G, Flier JS, Maratos-Flier E. Hepatic fibroblast growth factor 21 is regulated by PPARalpha and is a key mediator of hepatic lipid metabolism in ketotic states. Cell Metab. 2007; 5:426–37.24. Inagaki T, Dutchak P, Zhao G, Ding X, Gautron L, Parameswara V, et al. Endocrine regulation of the fasting response by PPARalpha-mediated induction of fibroblast growth factor 21. Cell Metab. 2007; 5:415–25.25. Zhang X, Yeung DC, Karpisek M, Stejskal D, Zhou ZG, Liu F, et al. Serum FGF21 levels are increased in obesity and are independently associated with the metabolic syndrome in humans. Diabetes. 2008; 57:1246–53.
Article26. Bobbert T, Schwarz F, Fischer-Rosinsky A, Pfeiffer AF, Mohlig M, Mai K, et al. Fibroblast growth factor 21 predicts the metabolic syndrome and type 2 diabetes in Caucasians. Diabetes Care. 2013; 36:145–9.
Article27. Fazeli PK, Lun M, Kim SM, Bredella MA, Wright S, Zhang Y, et al. FGF21 and the late adaptive response to starvation in humans. J Clin Invest. 2015; 125:4601–11.
Article28. Ko BJ, Kim SM, Park KH, Park HS, Mantzoros CS. Levels of circulating selenoprotein P, fibroblast growth factor (FGF) 21 and FGF23 in relation to the metabolic syndrome in young children. Int J Obes (Lond). 2014; 38:1497–502.
Article29. Reinehr T, Woelfle J, Wunsch R, Roth CL. Fibroblast growth factor 21 (FGF-21) and its relation to obesity, metabolic syndrome, and nonalcoholic fatty liver in children: a longitudinal analysis. J Clin Endocrinol Metab. 2012; 97:2143–50.
Article30. Chan JL, Heist K, DePaoli AM, Veldhuis JD, Mantzoros CS. The role of falling leptin levels in the neuroendocrine and metabolic adaptation to short-term starvation in healthy men. J Clin Invest. 2003; 111:1409–21.
Article31. Madeira I, Bordallo MA, Rodrigues NC, Carvalho C, Gazolla F, Collett-Solberg P, et al. Leptin as a predictor of metabolic syndrome in prepubertal children. Arch Endocrinol Metab. 2017; 61:7–13.
Article32. Nappo A, Gonzalez-Gil EM, Ahrens W, Bammann K, Michels N, Moreno LA, et al. Analysis of the association of leptin and adiponectin concentrations with metabolic syndrome in children: results from the IDEFICS study. Nutr Metab Cardiovasc Dis. 2017; 27:543–51.
Article33. Fantuzzi G, Mazzone T. Adipose tissue and atherosclerosis: exploring the connection. Arterioscler Thromb Vasc Biol. 2007; 27:996–1003.34. Satoh N, Naruse M, Usui T, Tagami T, Suganami T, Yamada K, et al. Leptin-to-adiponectin ratio as a potential atherogenic index in obese type 2 diabetic patients. Diabetes Care. 2004; 27:2488–90.
Article35. Sandhu MS, Heald AH, Gibson JM, Cruickshank JK, Dunger DB, Wareham NJ. Circulating concentrations of insulin-like growth factor-I and development of glucose intolerance: a prospective observational study. Lancet. 2002; 359:1740–5.
Article36. Lago R, Gomez R, Lago F, Gomez-Reino J, Gualillo O. Leptin beyond body weight regulation--current concepts concerning its role in immune function and inflammation. Cell Immunol. 2008; 252:139–45.
Article37. Planavila A, Redondo-Angulo I, Ribas F, Garrabou G, Casademont J, Giralt M, et al. Fibroblast growth factor 21 protects the heart from oxidative stress. Cardiovasc Res. 2015; 106:19–31.
Article38. Planavila A, Redondo I, Hondares E, Vinciguerra M, Munts C, Iglesias R, et al. Fibroblast growth factor 21 protects against cardiac hypertrophy in mice. Nat Commun. 2013; 4:2019.
Article39. Chow WS, Xu A, Woo YC, Tso AW, Cheung SC, Fong CH, et al. Serum fibroblast growth factor-21 levels are associated with carotid atherosclerosis independent of established cardiovascular risk factors. Arterioscler Thromb Vasc Biol. 2013; 33:2454–9.
Article40. Lin Z, Wu Z, Yin X, Liu Y, Yan X, Lin S, et al. Serum levels of FGF-21 are increased in coronary heart disease patients and are independently associated with adverse lipid profile. PLoS One. 2010; 5:e15534.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Transcriptional Regulation of Fibroblast Growth Factor 21 Expression
- Serum Fibroblast Growth Factor 21 and New-Onset Metabolic Syndrome: KoGES-ARIRANG Study
- Zerumbone, Sesquiterpene Photochemical from Ginger, Inhibits Angiogenesis
- Fibroblast Growth Factor 21: A Novel Metabolic Regulator
- Hepatokines as a Link between Obesity and Cardiovascular Diseases