Cancer Res Treat.
2021 Oct;53(4):1104-1112. 10.4143/crt.2020.1213.
Pulmonary Metastasectomy in Colorectal Cancer: A Population-Based Retrospective Cohort Study Using the Korean National Health Insurance Database
- Affiliations
-
- 1Department of Thoracic and Cardiovascular Surgery, Ajou University School of Medicine, Suwon, Korea
- 2Department of Thoracic and Cardiovascular Surgery, National Health Insurance Service Ilsan Hospital, Goyang, Korea
- 3Institute of Health Insurance and Clinical Research, National Health Insurance Service Ilsan Hospital, Goyang, Korea
- 4Department of General Surgery, National Health Insurance Service Ilsan Hospital, Goyang, Korea
- 5Department of Thoracic and Cardiovascular Surgery, Seoul National University Hospital, Seoul National University College of Medicine, Seoul, Korea
- KMID: 2521585
- DOI: http://doi.org/10.4143/crt.2020.1213
Abstract
- Purpose
The study aimed to investigate the current status and prognostic factors for overall survival in patients who had undergone pulmonary metastasectomy for colorectal cancer.
Materials and Methods
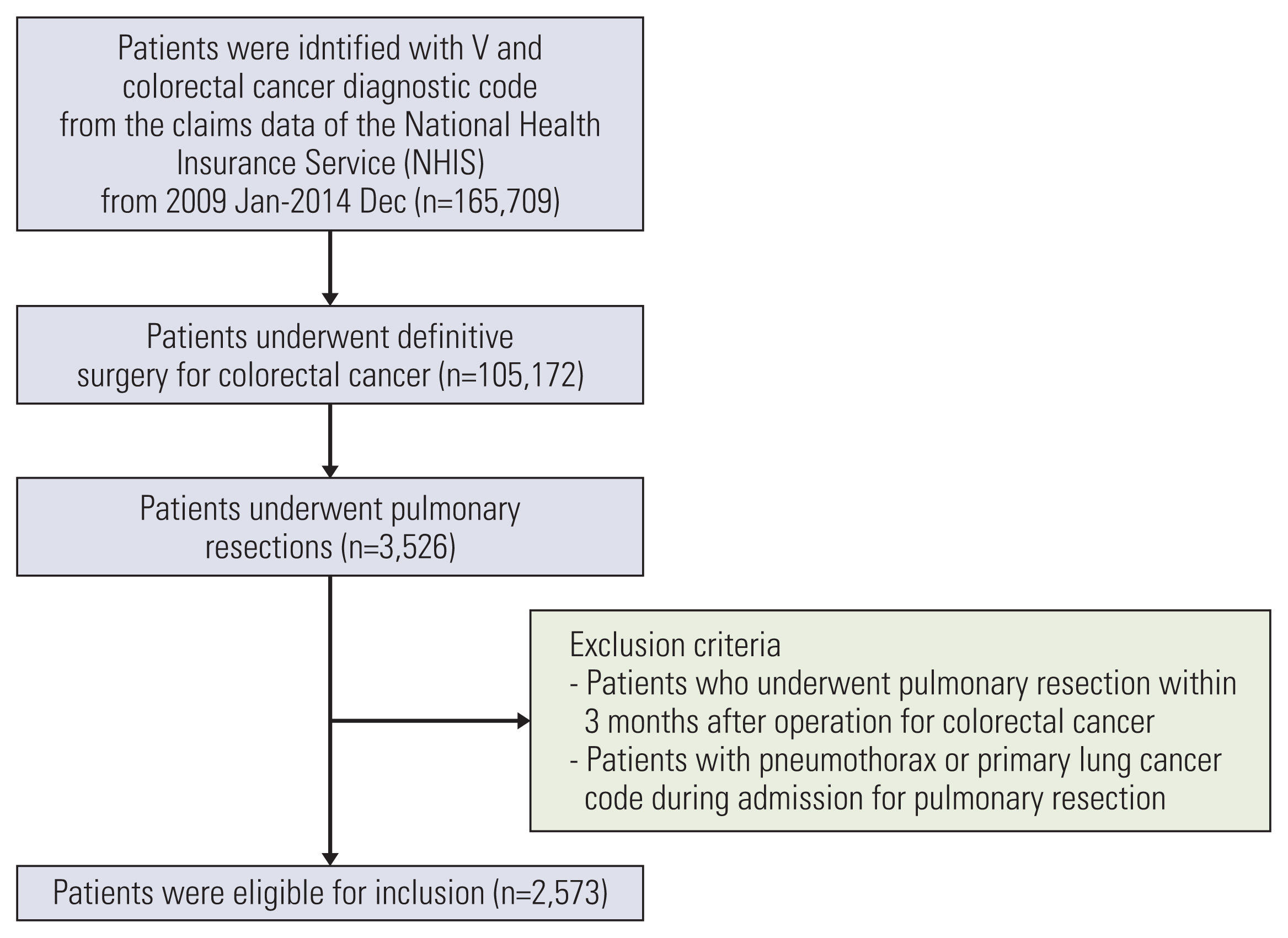
The data of 2,573 patients who had undergone pulmonary metastasectomy after surgery for colorectal cancer between January 2009 and December 2014 were extracted from the Korean National Health Insurance Service claims database. Patient-, colorectal cancer–, pulmonary metastasis–, and hospital-related factors were analyzed using the Kaplan-Meier method, log-rank test, and Cox proportional hazards analysis to identify prognostic factors for overall survival after pulmonary metastasectomy.
Results
The mean age of the patients was 60.9±10.5 years; 66.2% and 79.1% of the participants were male and had distally located colorectal cancer, respectively. Wedge resection (71.7%) was the most frequent extent of pulmonary resection; 21.8% of the patients underwent repeated pulmonary metastasectomies; 73% of pulmonary metastasectomy cases were performed in tertiary hospitals; 53.9% of patients were treated in Seoul area; 82% of patients received chemotherapy in conjunction with pulmonary metastasectomy. The median survival duration was 51.8 months. The 3- and 5-year overall survival rates were 67.7% and 39.4%, respectively. In multivariate analysis, female sex, distally located colorectal cancer, pulmonary metastasectomy-only treatment, and high hospital volume (> 10 pulmonary metastasectomy cases/yr) were positive prognostic factors for survival.
Conclusion
Pulmonary metastasectomy seemed to provide long-term survival of patients with colorectal cancer. The female sex, presence of distally located colorectal cancer, and performance of pulmonary metastasectomy in high-volume centers were positive prognostic factors for survival.
Keyword
Figure
Cited by 1 articles
-
The Clinical Efficacy of Colorectal Cancer Patients with Pulmonary Oligometastases by Sterotactic Body Ablative Radiotherapy: A Meta-Analysis
Jae-Uk Jeong, Chai Hong Rim, Gyu Sang Yoo, Won Kyung Cho, Eui Kyu Chie, Yong Chan Ahn, Jong Hoon Lee
Cancer Res Treat. 2024;56(3):809-824. doi: 10.4143/crt.2023.920.
Reference
-
References
1. Hong S, Won YJ, Park YR, Jung KW, Kong HJ, Lee ES, et al. Cancer statistics in Korea: incidence, mortality, survival, and prevalence in 2017. Cancer Res Treat. 2020; 52:335–50.
Article2. Mitry E, Guiu B, Cosconea S, Jooste V, Faivre J, Bouvier AM. Epidemiology, management and prognosis of colorectal cancer with lung metastases: a 30-year population-based study. Gut. 2010; 59:1383–8.
Article3. Pfannschmidt J, Dienemann H, Hoffmann H. Surgical resection of pulmonary metastases from colorectal cancer: a systematic review of published series. Ann Thorac Surg. 2007; 84:324–38.
Article4. Booth CM, Nanji S, Wei X, Mackillop WJ. Outcomes of resected colorectal cancer lung metastases in routine clinical practice: a population-based study. Ann Surg Oncol. 2016; 23:1057–63.
Article5. Iida T, Nomori H, Shiba M, Nakajima J, Okumura S, Horio H, et al. Prognostic factors after pulmonary metastasectomy for colorectal cancer and rationale for determining surgical indications: a retrospective analysis. Ann Surg. 2013; 257:1059–64.6. Kim JY, Park IJ, Kim HR, Kim DK, Lee JL, Yoon YS, et al. Post-pulmonary metastasectomy prognosis after curative resection for colorectal cancer. Oncotarget. 2017; 8:36566–77.
Article7. Suzuki H, Kiyoshima M, Kitahara M, Asato Y, Amemiya R. Long-term outcomes after surgical resection of pulmonary metastases from colorectal cancer. Ann Thorac Surg. 2015; 99:435–40.
Article8. Kwon S. Thirty years of national health insurance in South Korea: lessons for achieving universal health care coverage. Health Policy Plan. 2009; 24:63–71.
Article9. Pastorino U, Buyse M, Friedel G, Ginsberg RJ, Girard P, Goldstraw P, et al. Long-term results of lung metastasectomy: prognostic analyses based on 5206 cases. J Thorac Cardiovasc Surg. 1997; 113:37–49.
Article10. Bartlett EK, Simmons KD, Wachtel H, Roses RE, Fraker DL, Kelz RR, et al. The rise in metastasectomy across cancer types over the past decade. Cancer. 2015; 121:747–57.
Article11. National Comprehensive Cancer Network. NCCN guidelines for colon/rectal cancer, version 6.2020 [Internet]. Plymouth Meeting, PA: National Comprehensive Cancer Network;2020. [cited 2020 Dec 30]. Available from: https://www.nccn.org/professionals/physician_dls .12. Handy JR, Bremner RM, Crocenzi TS, Detterbeck FC, Fernando HC, Fidias PM, et al. Expert consensus document on pulmonary metastasectomy. Ann Thorac Surg. 2019; 107:631–49.
Article13. Treasure T, Farewell V, Macbeth F, Monson K, Williams NR, Brew-Graves C, et al. Pulmonary Metastasectomy versus Continued Active Monitoring in Colorectal Cancer (PulMiCC): a multicentre randomised clinical trial. Trials. 2019; 20:718.
Article14. Milosevic M, Edwards J, Tsang D, Dunning J, Shackcloth M, Batchelor T, et al. Pulmonary Metastasectomy in Colorectal Cancer: updated analysis of 93 randomized patients: control survival is much better than previously assumed. Colorectal Dis. 2020; 22:1314–24.15. Choi KH, Song JH, Jang HS, Kim SH, Lee JH. Current trends in the quality assessment of colorectal cancer practice and treatment in South Korea during 2012–2017. Cancer Res Treat. 2021; 53:487–96.
Article16. Pugh SA, Shinkins B, Fuller A, Mellor J, Mant D, Primrose JN. Site and stage of colorectal cancer influence the likelihood and distribution of disease recurrence and postrecurrence survival: data from the FACS randomized controlled trial. Ann Surg. 2016; 263:1143–7.17. Tepper JE, O’Connell M, Hollis D, Niedzwiecki D, Cooke E, Mayer RJ, et al. Analysis of surgical salvage after failure of primary therapy in rectal cancer: results from Intergroup Study 0114. J Clin Oncol. 2003; 21:3623–8.
Article18. Missiaglia E, Jacobs B, D’Ario G, Di Narzo AF, Soneson C, Budinska E, et al. Distal and proximal colon cancers differ in terms of molecular, pathological, and clinical features. Ann Oncol. 2014; 25:1995–2001.
Article19. Mukund K, Syulyukina N, Ramamoorthy S, Subramaniam S. Right and left-sided colon cancers: specificity of molecular mechanisms in tumorigenesis and progression. BMC Cancer. 2020; 20:317.
Article20. Park HS, Jung M, Shin SJ, Heo SJ, Kim CG, Lee MG, et al. Benefit of adjuvant chemotherapy after curative resection of lung metastasis in colorectal cancer. Ann Surg Oncol. 2016; 23:928–35.
Article21. Li Y, Qin Y. Peri-operative chemotherapy for resectable colorectal lung metastasis: a systematic review and meta-analysis. J Cancer Res Clin Oncol. 2020; 146:545–53.
Article22. Lemini R, Attwood K, Almerey T, Gunn J, Yeager TE, Elias AW, et al. Is metastasectomy a worthy option?: the role of surgery in metastatic colon cancer to liver and lungs. J Gastrointest Oncol. 2019; 10:1032–48.23. Jung KW, Park S, Shin A, Oh CM, Kong HJ, Jun JK, et al. Do female cancer patients display better survival rates compared with males? Analysis of the Korean National Registry data, 2005–2009. PLoS One. 2012; 7:e52457.
Article24. Radkiewicz C, Johansson ALV, Dickman PW, Lambe M, Edgren G. Sex differences in cancer risk and survival: a Swedish cohort study. Eur J Cancer. 2017; 84:130–40.
Article25. Blackmon SH, Stephens EH, Correa AM, Hofstetter W, Kim MP, Mehran RJ, et al. Predictors of recurrent pulmonary metastases and survival after pulmonary metastasectomy for colorectal cancer. Ann Thorac Surg. 2012; 94:1802–9.
Article26. Hillner BE, Smith TJ, Desch CE. Hospital and physician volume or specialization and outcomes in cancer treatment: importance in quality of cancer care. J Clin Oncol. 2000; 18:2327–40.
Article27. Cole AP, Sun M, Lipsitz SR, Sood A, Kibel AS, Trinh QD. Reassessing the value of high-volume cancer care in the era of precision medicine. Cancer. 2018; 124:1319–21.
Article28. Birkmeyer JD, Sun Y, Wong SL, Stukel TA. Hospital volume and late survival after cancer surgery. Ann Surg. 2007; 245:777–83.
Article29. Park S, Park IK, Kim ER, Hwang Y, Lee HJ, Kang CH, et al. Current trends of lung cancer surgery and demographic and social factors related to changes in the trends of lung cancer surgery: an analysis of the national database from 2010 to 2014. Cancer Res Treat. 2017; 49:330–7.
Article30. Doll KM, Rademaker A, Sosa JA. Practical guide to surgical data sets: Surveillance, Epidemiology, and End Results (SEER) database. JAMA Surg. 2018; 153:588–9.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Risk Factors for Colorectal Cancer in Korea: A Population-Based Retrospective Cohort Study
- Pulmonary metastasectomy in uterine malignancy: outcomes and prognostic factors
- Analysis of Correlation between Thyroid Cancer Incidence and Socioeconomic Status Using 10-year Sample Cohort Database
- Integrated database-based Screening Cohort for Asian Nomadic descendants in China (Scan-China): Insights on prospective ethnicity-focused cancer screening
- Curative Resection for Metachronous Pulmonary Metastases from Colorectal Cancer: Analysis of Survival Rates and Prognostic Factors