Korean Circ J.
2021 Sep;51(9):733-751. 10.4070/kcj.2021.0153.
Hypertrophic Cardiomyopathy in Infants from the Perspective of Cardiomyocyte Maturation
- Affiliations
-
- 1Biomedical Research Institute, Seoul National University Hospital, Seoul, Korea
- 2Department of Pediatrics, St. Vincent's Hospital, College of Medicine, The Catholic University of Korea, Seoul, Korea
- KMID: 2519834
- DOI: http://doi.org/10.4070/kcj.2021.0153
Abstract
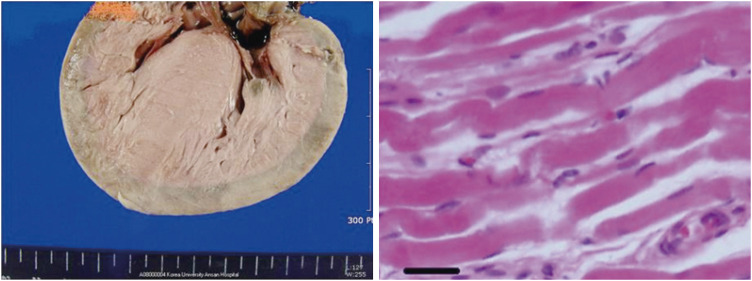
- Hypertrophic cardiomyopathy (HCM) is characterized by ventricular wall hypertrophy with diastolic dysfunction. Pediatric HCM is distinguished from the adult in many aspects. Most children with HCM do not present clinically until the adolescent period, even when they are born with genetic mutations. Some infants with early-onset HCM present with massive progressive myocardial hypertrophy in the first few months of life, which is often fatal. The mortality of pediatric HCM peaks during the infantile and adolescent periods. These periods roughly correlate with children's growth spurt. Non-sarcomeric causes of HCM are more frequent in pediatric HCM, while sarcomeric causes are more common in adults. From the perspective of cardiac development, the fetal heart has immature cardiomyocytes, which are characterized by proliferation and exit their cell cycles with a decreased regenerative property after birth. In the perinatal period, there is a dynamic change in maturation of cardiomyocytes from immature to mature cells. Infants who are treated with steroids or born to mothers with diabetes or hyperthyroidism often show phenotypes of HCM, which gradually resolve. With remarkable advancement of molecular biology, understanding on maturation of cardiomyocytes has increased. Neonates undergo abrupt environmental changes during the transitional circulation, which is affected by oxygen, metabolic and hormonal fluctuations. Derangement in physiological transition to the normal postnatal environment may influence maturation of proliferative immature cardiomyocytes during early infancy. This article reviews updates of infantile HCM and recent molecular studies related to maturation of cardiomyocytes from the clinical point of view of identifying distinct characteristics of infantile HCM.
Keyword
Figure
Reference
-
1. Seo HS, Lee IH, Song YW, et al. A case of congenital hypertrophic cardiomyopathy. Korean Circ J. 2013; 43:54–56. PMID: 23407623.
Article2. Lipshultz SE, Law YM, Asante-Korang A, et al. Cardiomyopathy in children: classification and diagnosis: a scientific statement from the American Heart Association. Circulation. 2019; 140:e9–68. PMID: 31132865.
Article3. Oh JH, Hong YM, Choi JY, et al. Idiopathic cardiomyopathies in Korean children: 9-year Korean multicenter study. Circ J. 2011; 75:2228–2234. PMID: 21747192.4. Ommen SR, Mital S, Burke MA, et al. 2020 AHA/ACC guideline for the diagnosis and treatment of patients with hypertrophic cardiomyopathy: executive summary: a report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice Guidelines. J Am Coll Cardiol. 2020; 76:3022–3055. PMID: 33229115.5. Bamborschke D, Özdemir Ö, Kreutzer M, et al. Ultra-rapid emergency genomic diagnosis of Donahue syndrome in a preterm infant within 17 hours. Am J Med Genet A. 2021; 185:90–96. PMID: 33048476.
Article6. Lafreniere-Roula M, Bolkier Y, Zahavich L, et al. Family screening for hypertrophic cardiomyopathy: is it time to change practice guidelines? Eur Heart J. 2019; 40:3672–3681. PMID: 31170284.
Article7. Helms AS, Tang VT, O'Leary TS, et al. Effects of MYBPC3 loss-of-function mutations preceding hypertrophic cardiomyopathy. JCI Insight. 2020; 5:e133782.
Article8. Kolokotronis K, Kühnisch J, Klopocki E, et al. Biallelic mutation in MYH7 and MYBPC3 leads to severe cardiomyopathy with left ventricular noncompaction phenotype. Hum Mutat. 2019; 40:1101–1114. PMID: 30924982.9. Xin B, Puffenberger E, Tumbush J, Bockoven JR, Wang H. Homozygosity for a novel splice site mutation in the cardiac myosin-binding protein C gene causes severe neonatal hypertrophic cardiomyopathy. Am J Med Genet A. 2007; 143A:2662–2667. PMID: 17937428.
Article10. Bergmann O, Zdunek S, Felker A, et al. Dynamics of cell generation and turnover in the human heart. Cell. 2015; 161:1566–1575. PMID: 26073943.
Article11. Zhu W, Zhang E, Zhao M, et al. Regenerative potential of neonatal porcine hearts. Circulation. 2018; 138:2809–2816. PMID: 30030418.
Article12. Galdos FX, Guo Y, Paige SL, VanDusen NJ, Wu SM, Pu WT. Cardiac regeneration: lessons from development. Circ Res. 2017; 120:941–959. PMID: 28302741.13. Bergmann O, Bhardwaj RD, Bernard S, et al. Evidence for cardiomyocyte renewal in humans. Science. 2009; 324:98–102. PMID: 19342590.
Article14. Porrello ER, Mahmoud AI, Simpson E, et al. Transient regenerative potential of the neonatal mouse heart. Science. 2011; 331:1078–1080. PMID: 21350179.
Article15. Mollova M, Bersell K, Walsh S, et al. Cardiomyocyte proliferation contributes to heart growth in young humans. Proc Natl Acad Sci U S A. 2013; 110:1446–1451. PMID: 23302686.
Article16. Garbern JC, Lee RT. Mitochondria and metabolic transitions in cardiomyocytes: lessons from development for stem cell-derived cardiomyocytes. Stem Cell Res Ther. 2021; 12:177. PMID: 33712058.
Article17. Soonpaa MH, Kim KK, Pajak L, Franklin M, Field LJ. Cardiomyocyte DNA synthesis and binucleation during murine development. Am J Physiol. 1996; 271:H2183–9. PMID: 8945939.
Article18. Naqvi N, Li M, Calvert JW, et al. A proliferative burst during preadolescence establishes the final cardiomyocyte number. Cell. 2014; 157:795–807. PMID: 24813607.
Article19. Senyo SE, Lee RT, Kühn B. Cardiac regeneration based on mechanisms of cardiomyocyte proliferation and differentiation. Stem Cell Res (Amst). 2014; 13:532–541.
Article20. Bishop SP, Zhou Y, Nakada Y, Zhang J. Changes in cardiomyocyte cell cycle and hypertrophic growth during fetal to adult in mammals. J Am Heart Assoc. 2021; 10:e017839. PMID: 33399005.
Article21. Guo Y, Pu WT. Cardiomyocyte maturation: new phase in development. Circ Res. 2020; 126:1086–1106. PMID: 32271675.22. Guo Y, Cao Y, Jardin BD, et al. Sarcomeres regulate murine cardiomyocyte maturation through MRTF-SRF signaling. Proc Natl Acad Sci U S A. 2021; 118:e2008861118. PMID: 33361330.
Article23. Seok H, Deng R, Cowan DB, Wang DZ. Application of CRISPR-Cas9 gene editing for congenital heart disease. Clin Exp Pediatr. 2021; 64:269–279. PMID: 33677855.
Article24. Guo Y, Jardin BD, Zhou P, et al. Hierarchical and stage-specific regulation of murine cardiomyocyte maturation by serum response factor. Nat Commun. 2018; 9:3837. PMID: 30242271.
Article25. Guo Y, VanDusen NJ, Zhang L, et al. Analysis of cardiac myocyte maturation using CASAAV, a platform for rapid dissection of cardiac myocyte gene function in vivo. Circ Res. 2017; 120:1874–1888. PMID: 28356340.
Article26. Pierpont ME, Brueckner M, Chung WK, et al. Genetic basis for congenital heart disease: revisited: a scientific statement from the American Heart Association. Circulation. 2018; 138:e653–711. PMID: 30571578.
Article27. Akerberg BN, Pu WT. Genetic and epigenetic control of heart development. Cold Spring Harb Perspect Biol. 2020; 12:a036756. PMID: 31818853.
Article28. Cutie S, Huang GN. Vertebrate cardiac regeneration: evolutionary and developmental perspectives. Cell Regen (Lond). 2021; 10:6.
Article29. Taber LA. Biomechanics of cardiovascular development. Annu Rev Biomed Eng. 2001; 3:1–25. PMID: 11447055.
Article30. Fukuda R, Gunawan F, Ramadass R, et al. Mechanical forces regulate cardiomyocyte myofilament maturation via the VCL-SSH1-CFL axis. Dev Cell. 2019; 51:62–77.e5. PMID: 31495694.
Article31. Hines MH. Neonatal cardiovascular physiology. Semin Pediatr Surg. 2013; 22:174–178. PMID: 24331090.
Article32. Bubb KJ, Cock ML, Black MJ, et al. Intrauterine growth restriction delays cardiomyocyte maturation and alters coronary artery function in the fetal sheep. J Physiol. 2007; 578:871–881. PMID: 17124269.
Article33. Helms AS, Alvarado FJ, Yob J, et al. Genotype-dependent and -independent calcium signaling dysregulation in human hypertrophic cardiomyopathy. Circulation. 2016; 134:1738–1748. PMID: 27688314.
Article34. Hamaguchi S, Kawakami Y, Honda Y, et al. Developmental changes in excitation-contraction mechanisms of the mouse ventricular myocardium as revealed by functional and confocal imaging analyses. J Pharmacol Sci. 2013; 123:167–175. PMID: 24096881.
Article35. Piquereau J, Novotova M, Fortin D, et al. Postnatal development of mouse heart: formation of energetic microdomains. J Physiol. 2010; 588:2443–2454. PMID: 20478976.
Article36. Atkison P, Joubert G, Barron A, et al. Hypertrophic cardiomyopathy associated with tacrolimus in paediatric transplant patients. Lancet. 1995; 345:894–896. PMID: 7535875.
Article37. Turska-Kmieć A, Jankowska I, Pawłowska J, et al. Reversal of tacrolimus-related hypertrophic cardiomyopathy after conversion to rapamycin in a pediatric liver transplant recipient. Pediatr Transplant. 2007; 11:319–323. PMID: 17430490.
Article38. Davis J, Davis LC, Correll RN, et al. A tension-based model distinguishes hypertrophic versus dilated cardiomyopathy. Cell. 2016; 165:1147–1159. PMID: 27114035.
Article39. Robinson P, Liu X, Sparrow A, et al. Hypertrophic cardiomyopathy mutations increase myofilament Ca2+ buffering, alter intracellular Ca2+ handling, and stimulate Ca2+-dependent signaling. J Biol Chem. 2018; 293:10487–10499. PMID: 29760186.40. Bensley JG, Moore L, De Matteo R, Harding R, Black MJ. Impact of preterm birth on the developing myocardium of the neonate. Pediatr Res. 2018; 83:880–888. PMID: 29278645.
Article41. Aye CY, Lewandowski AJ, Lamata P, et al. Disproportionate cardiac hypertrophy during early postnatal development in infants born preterm. Pediatr Res. 2017; 82:36–46. PMID: 28399117.
Article42. Kannan S, Kwon C. Regulation of cardiomyocyte maturation during critical perinatal window. J Physiol. 2020; 598:2941–2956. PMID: 30571853.
Article43. Aiyengar A, Howarth C, Pereira S. Hypertrophic cardiomyopathy in an extremely preterm infant. BMJ Case Rep. 2021; 14:14.
Article44. Doyle LW, Cheong JL, Ehrenkranz RA, Halliday HL. Early (< 8 days) systemic postnatal corticosteroids for prevention of bronchopulmonary dysplasia in preterm infants. Cochrane Database Syst Rev. 2017; 10:CD001146. PMID: 29063585.45. Halliday HL. Update on postnatal steroids. Neonatology. 2017; 111:415–422. PMID: 28538237.
Article46. Jiang J, Zhang J, Kang M, Yang J. Transient hypertrophic cardiomyopathy and hypertension associated with hydrocortisone in preterm infant: a case report. Medicine (Baltimore). 2019; 98:e16838. PMID: 31415406.47. Ramaswamy VV, Bandyopadhyay T, Nanda D, et al. Assessment of postnatal corticosteroids for the prevention of bronchopulmonary dysplasia in preterm neonates: a systematic review and network meta-analysis. JAMA Pediatr. 2021; 175:e206826. PMID: 33720274.48. Vimala J, Prabhu A, Pavithran S, Kumar RN. Hydrocortisone induced hypertrophic cardiomyopathy. Int J Cardiol. 2011; 150:e94–5. PMID: 20045574.
Article49. Kale Y, Aydemir O, Ceylan O, Bas AY, Demirel N. Hypertrophic cardiomyopathy after a single dose of dexamethasone in a preterm infant. Pediatr Neonatol. 2015; 56:268–270. PMID: 23639746.50. Phad NS, de Waal K, Holder C, Oldmeadow C. Dilated hypertrophy: a distinct pattern of cardiac remodeling in preterm infants. Pediatr Res. 2020; 87:146–152. PMID: 31493773.
Article51. Puente BN, Kimura W, Muralidhar SA, et al. The oxygen-rich postnatal environment induces cardiomyocyte cell-cycle arrest through DNA damage response. Cell. 2014; 157:565–579. PMID: 24766806.
Article52. Burgoyne JR, Mongue-Din H, Eaton P, Shah AM. Redox signaling in cardiac physiology and pathology. Circ Res. 2012; 111:1091–1106. PMID: 23023511.
Article53. Seok H, Lee H, Lee S, et al. Position-specific oxidation of miR-1 encodes cardiac hypertrophy. Nature. 2020; 584:279–285. PMID: 32760005.
Article54. de Salvi Guimarães F, de Moraes WM, Bozi LH, et al. Dexamethasone-induced cardiac deterioration is associated with both calcium handling abnormalities and calcineurin signaling pathway activation. Mol Cell Biochem. 2017; 424:87–98. PMID: 27761848.
Article55. Sakurai K, Osada Y, Takeba Y, et al. Exposure of immature rat heart to antenatal glucocorticoid results in cardiac proliferation. Pediatr Int. 2019; 61:31–42. PMID: 30387893.
Article56. Li F, Wang X, Capasso JM, Gerdes AM. Rapid transition of cardiac myocytes from hyperplasia to hypertrophy during postnatal development. J Mol Cell Cardiol. 1996; 28:1737–1746. PMID: 8877783.
Article57. Velayutham N, Alfieri CM, Agnew EJ, et al. Cardiomyocyte cell cycling, maturation, and growth by multinucleation in postnatal swine. J Mol Cell Cardiol. 2020; 146:95–108. PMID: 32710980.
Article58. Paech C, Wolf N, Thome UH, Knüpfer M. Hypertrophic intraventricular flow obstruction after very-low-dose dexamethasone (Minidex) in preterm infants: case presentation and review of the literature. J Perinatol. 2014; 34:244–246. PMID: 24573211.
Article59. Wong IH, Digby AM, Warren AE, Pepelassis D, Vincer M, Chen RP. Dexamethasone given to premature infants and cardiac diastolic function in early childhood. J Pediatr. 2011; 159:227–231. PMID: 21397911.
Article60. Sperl W, Sengers RC, Trijbels JM, et al. Enzyme activities of the mitochondrial energy generating system in skeletal muscle tissue of preterm and fullterm neonates. Ann Clin Biochem. 1992; 29:638–645. PMID: 1489160.
Article61. Lai L, Leone TC, Zechner C, et al. Transcriptional coactivators PGC-1alpha and PGC-lbeta control overlapping programs required for perinatal maturation of the heart. Genes Dev. 2008; 22:1948–1961. PMID: 18628400.62. Dorn GW 2nd, Vega RB, Kelly DP. Mitochondrial biogenesis and dynamics in the developing and diseased heart. Genes Dev. 2015; 29:1981–1991. PMID: 26443844.
Article63. Gibb AA, Hill BG. Metabolic coordination of physiological and pathological cardiac remodeling. Circ Res. 2018; 123:107–128. PMID: 29929976.
Article64. Haack TB, Danhauser K, Haberberger B, et al. Exome sequencing identifies ACAD9 mutations as a cause of complex I deficiency. Nat Genet. 2010; 42:1131–1134. PMID: 21057504.65. Dewulf JP, Barrea C, Vincent MF, et al. Evidence of a wide spectrum of cardiac involvement due to ACAD9 mutations: report on nine patients. Mol Genet Metab. 2016; 118:185–189. PMID: 27233227.66. Wang G, McCain ML, Yang L, et al. Modeling the mitochondrial cardiomyopathy of Barth syndrome with induced pluripotent stem cell and heart-on-chip technologies. Nat Med. 2014; 20:616–623. PMID: 24813252.
Article67. Ullmo S, Vial Y, Di Bernardo S, et al. Pathologic ventricular hypertrophy in the offspring of diabetic mothers: a retrospective study. Eur Heart J. 2007; 28:1319–1325. PMID: 17158827.
Article68. Huang T, Kelly A, Becker SA, Cohen MS, Stanley CA. Hypertrophic cardiomyopathy in neonates with congenital hyperinsulinism. Arch Dis Child Fetal Neonatal Ed. 2013; 98:F351–4. PMID: 23377780.
Article69. Paauw ND, Stegeman R, de Vroede MA, Termote JU, Freund MW, Breur JM. Neonatal cardiac hypertrophy: the role of hyperinsulinism-a review of literature. Eur J Pediatr. 2020; 179:39–50. PMID: 31840185.
Article70. Bartelds B, Gratama JW, Knoester H, et al. Perinatal changes in myocardial supply and flux of fatty acids, carbohydrates, and ketone bodies in lambs. Am J Physiol. 1998; 274:H1962–9. PMID: 9841523.71. DeBosch BJ, Muslin AJ. Insulin signaling pathways and cardiac growth. J Mol Cell Cardiol. 2008; 44:855–864. PMID: 18423486.
Article72. Perge K, Massoud M, Gauthier-Moulinier H, et al. Intrauterine growth restriction and hypertrophic cardiomyopathy as prenatal ultrasound findings in a case of leprechaunism. Mol Syndromol. 2020; 11:223–227. PMID: 33224016.
Article73. Hayati AR, Cheah FC, Yong JF, Tan AE, Norizah WM. The role of serum insulin-like growth factor I (IGF-I) in neonatal outcome. J Clin Pathol. 2004; 57:1299–1301. PMID: 15563671.
Article74. Gonzalez AB, Young L, Doll JA, Morgan GM, Crawford SE, Plunkett BA. Elevated neonatal insulin-like growth factor I is associated with fetal hypertrophic cardiomyopathy in diabetic women. Am J Obstet Gynecol. 2014; 211:290.e1–290.e7. PMID: 24813596.
Article75. Nakano H, Minami I, Braas D, et al. Glucose inhibits cardiac muscle maturation through nucleotide biosynthesis. eLife. 2017; 6:e29330. PMID: 29231167.
Article76. Gordon EE, Reinking BE, Hu S, et al. Maternal hyperglycemia directly and rapidly induces cardiac septal overgrowth in fetal rats. J Diabetes Res. 2015; 2015:479565. PMID: 26064981.
Article77. Basu M, Garg V. Maternal hyperglycemia and fetal cardiac development: clinical impact and underlying mechanisms. Birth Defects Res. 2018; 110:1504–1516. PMID: 30576094.
Article78. Alemu A, Terefe B, Abebe M, Biadgo B. Thyroid hormone dysfunction during pregnancy: a review. Int J Reprod Biomed. 2016; 14:677–686. PMID: 27981252.
Article79. Männistö T, Mendola P, Reddy U, Laughon SK. Neonatal outcomes and birth weight in pregnancies complicated by maternal thyroid disease. Am J Epidemiol. 2013; 178:731–740. PMID: 23666815.
Article80. Chattergoon NN. Thyroid hormone signaling and consequences for cardiac development. J Endocrinol. 2019; 242:T145–60. PMID: 31117055.
Article81. Chattergoon NN, Giraud GD, Louey S, Stork P, Fowden AL, Thornburg KL. Thyroid hormone drives fetal cardiomyocyte maturation. FASEB J. 2012; 26:397–408. PMID: 21974928.
Article82. Polk DH. Thyroid hormone metabolism during development. Reprod Fertil Dev. 1995; 7:469–477. PMID: 8606958.
Article83. Williams FL, Simpson J, Delahunty C, et al. Developmental trends in cord and postpartum serum thyroid hormones in preterm infants. J Clin Endocrinol Metab. 2004; 89:5314–5320. PMID: 15531476.
Article84. Rauen KA. The RASopathies. Annu Rev Genomics Hum Genet. 2013; 14:355–369. PMID: 23875798.
Article85. Karnik R, Geiger M. Cardiac manifestations of Noonan syndrome. Pediatr Endocrinol Rev. 2019; 16:471–476. PMID: 31115199.86. Tartaglia M, Mehler EL, Goldberg R, et al. Mutations in PTPN11, encoding the protein tyrosine phosphatase SHP-2, cause Noonan syndrome. Nat Genet. 2001; 29:465–468. PMID: 11704759.87. Wilkinson JD, Lowe AM, Salbert BA, et al. Outcomes in children with Noonan syndrome and hypertrophic cardiomyopathy: a study from the Pediatric Cardiomyopathy Registry. Am Heart J. 2012; 164:442–448. PMID: 22980313.
Article88. Aoki Y, Niihori T, Banjo T, et al. Gain-of-function mutations in RIT1 cause Noonan syndrome, a RAS/MAPK pathway syndrome. Am J Hum Genet. 2013; 93:173–180. PMID: 23791108.89. Jaffré F, Miller CL, Schänzer A, et al. Inducible pluripotent stem cell-derived cardiomyocytes reveal aberrant extracellular regulated kinase 5 and mitogen-activated protein kinase kinase 1/2 signaling concomitantly promote hypertrophic cardiomyopathy in RAF1-associated Noonan syndrome. Circulation. 2019; 140:207–224. PMID: 31163979.90. Koda M, Takemura G, Okada H, et al. Nuclear hypertrophy reflects increased biosynthetic activities in myocytes of human hypertrophic hearts. Circ J. 2006; 70:710–718. PMID: 16723792.
Article91. Haubner BJ, Adamowicz-Brice M, Khadayate S, et al. Complete cardiac regeneration in a mouse model of myocardial infarction. Aging (Albany NY). 2012; 4:966–977. PMID: 23425860.
Article92. D'Uva G, Aharonov A, Lauriola M, et al. ERBB2 triggers mammalian heart regeneration by promoting cardiomyocyte dedifferentiation and proliferation. Nat Cell Biol. 2015; 17:627–638. PMID: 25848746.93. Basso C, Thiene G, Corrado D, Buja G, Melacini P, Nava A. Hypertrophic cardiomyopathy and sudden death in the young: pathologic evidence of myocardial ischemia. Hum Pathol. 2000; 31:988–998. PMID: 10987261.
Article94. Nishikawa T, Uto K, Kanai S, et al. Histopathological aspects of cardiac biopsy in pediatric patients with dilated cardiomyopathy. Pediatr Int. 2011; 53:350–353. PMID: 20854285.
Article95. Hahn A, Lauriol J, Thul J, et al. Rapidly progressive hypertrophic cardiomyopathy in an infant with Noonan syndrome with multiple lentigines: palliative treatment with a rapamycin analog. Am J Med Genet A. 2015; 167A:744–751. PMID: 25708222.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Two Cases of Hypertrophic Cardiomyopathy in the Infants of Diabetic Mother
- An autopssy case of infant of diabetic mother with d-transposition of great arteries and hypertrophic cardiomyopathy
- Non-invasive Assessment of Hypertrophic Cardiomyopathy
- Tips for Successful Septal Myectomy in Patients with Hypertrophic Cardiomyopathy
- A Case of Normalized Hypertrophic Cardiomyopathy after Removal of Pheochromocytoma