Korean J Physiol Pharmacol.
2021 May;25(3):239-249. 10.4196/kjpp.2021.25.3.239.
Hydrogen sulfide restores cardioprotective effects of remote ischemic preconditioning in aged rats via HIF-1α/Nrf2 signaling pathway
- Affiliations
-
- 1Department of Cardiovascular, First Affiliated Hospital of Xi’an Jiaotong University, Xi’an, Shaanxi 710061, China
- 2Departments of Cardiovascular, Affiliated Zhongshan Hospital of Dalian University, Dalian, Liaoning 116001, China
- 3Departments of Judicial Expertise, Affiliated Zhongshan Hospital of Dalian University, Dalian, Liaoning 116001, China
- KMID: 2514949
- DOI: http://doi.org/10.4196/kjpp.2021.25.3.239
Abstract
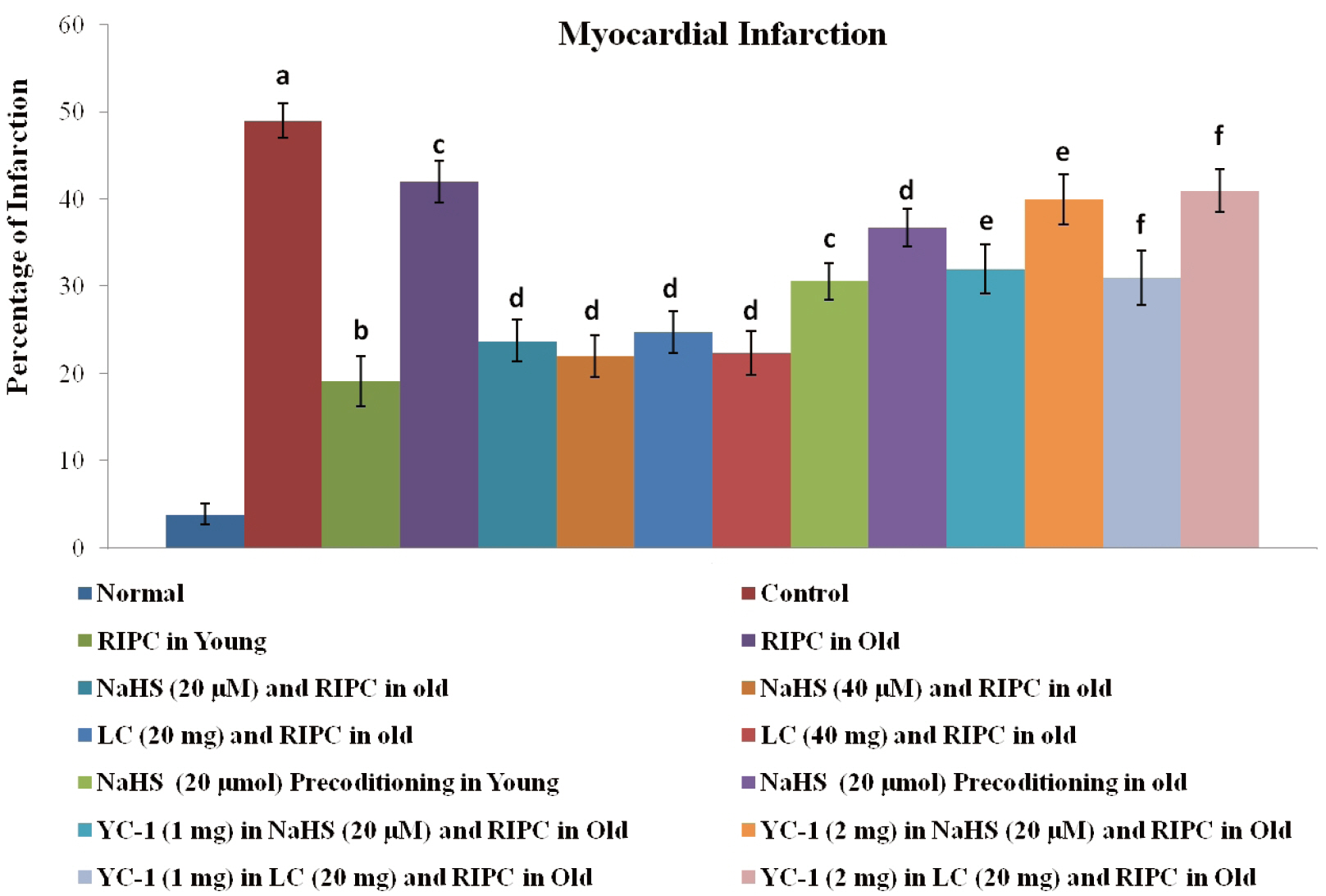
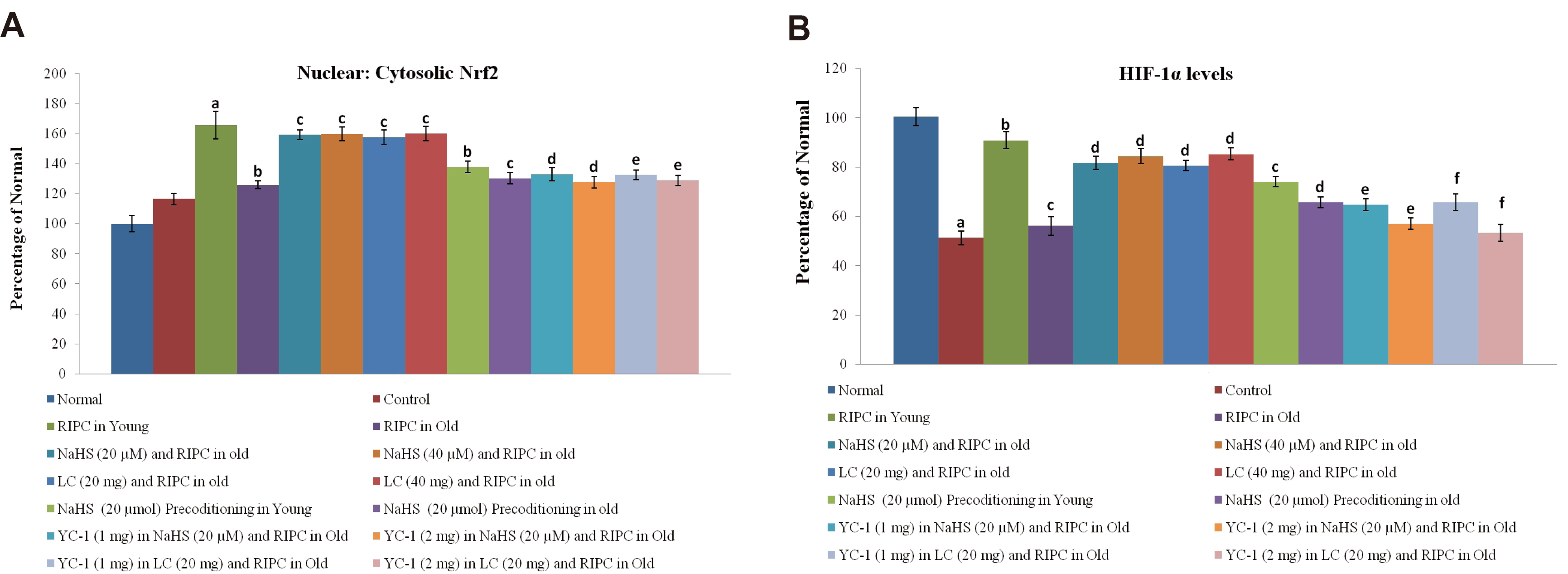
- The present study explored the therapeutic potential of hydrogen sulfide (H2 S) in restoring aging-induced loss of cardioprotective effect of remote ischemic preconditioning (RIPC) along with the involvement of signaling pathways. The left hind limb was subjected to four short cycles of ischemia and reperfusion (IR) in young and aged male rats to induce RIPC. The hearts were subjected to IR injury on the Langendorff apparatus after 24 h of RIPC. The measurement of lactate dehydrogenase, creatine kinase and cardiac troponin served to assess the myocardial injury. The levels of H2S, cystathionine β-synthase (CBS), cystathionine γ-lyase (CSE), nuclear factor erythroid 2-related factor 2 (Nrf2), and hypoxia-inducible factor (HIF-1α) were also measured. There was a decrease in cardioprotection in RIPC-subjected old rats in comparison to young rats along with a reduction in the myocardial levels of 2, CBS, CSE, HIF-1α, and nuclear: cytoplasmic Nrf2 ratio. Supplementation with sodium hydrogen sulfide (NaHS, an H2S donor) and l-cysteine ( H 2S precursor) restored the cardioprotective actions of RIPC in old hearts. It increased the levels of H2S, HIF-1α, and Nrf2 ratio without affecting CBS and CSE. YC-1 (HIF-1α antagonist) abolished the effects of NaHS and l-cysteine in RIPC-subjected old rats by decreasing the Nrf2 ratio and HIF-1α levels, without altering 2.The late phase of cardioprotection of RIPC involves an increase in the activity of H2S biosynthetic enzymes, which increases the levels of H2S to upregulate HIF-1α and Nrf2. H2S has the potential to restore aging-induced loss of cardioprotective effects of RIPC by upregulating HIF-1α/Nrf2 signaling.
Keyword
Figure
Reference
-
1. Donato M, Evelson P, Gelpi RJ. 2017; Protecting the heart from ischemia/reperfusion injury: an update on remote ischemic preconditioning and postconditioning. Curr Opin Cardiol. 32:784–790. DOI: 10.1097/HCO.0000000000000447. PMID: 28902715.2. Singh A, Randhawa PK, Bali A, Singh N, Jaggi AS. 2017; Exploring the role of TRPV and CGRP in adenosine preconditioning and remote hind limb preconditioning-induced cardioprotection in rats. Cardiovasc Drugs Ther. 31:133–143. DOI: 10.1007/s10557-017-6716-3. PMID: 28194544.
Article3. Loukogeorgakis SP, Panagiotidou AT, Broadhead MW, Donald A, Deanfield JE, MacAllister RJ. 2005; Remote ischemic preconditioning provides early and late protection against endothelial ischemia-reperfusion injury in humans: role of the autonomic nervous system. J Am Coll Cardiol. 46:450–456. DOI: 10.1016/j.jacc.2005.04.044. PMID: 16053957.4. Singh H, Kumar M, Singh N, Jaggi AS. 2019; Late phases of cardioprotection during remote ischemic preconditioning and adenosine preconditioning involve activation of neurogenic pathway. J Cardiovasc Pharmacol. 73:63–69. DOI: 10.1097/FJC.0000000000000634. PMID: 30422893.
Article5. Ravingerova T, Farkasova V, Griecsova L, Carnicka S, Murarikova M, Barlaka E, Kolar F, Bartekova M, Lonek L, Slezak J, Lazou A. 2016; Remote preconditioning as a novel "conditioning" approach to repair the broken heart: potential mechanisms and clinical applications. Physiol Res. 65 Suppl 1:S55–S64. DOI: 10.33549/physiolres.933392. PMID: 27643940.
Article6. Lau JK, Roy P, Javadzadegan A, Moshfegh A, Fearon WF, Ng M, Lowe H, Brieger D, Kritharides L, Yong AS. 2018; Remote ischemic preconditioning acutely improves coronary microcirculatory function. J Am Heart Assoc. 7:e009058. DOI: 10.1161/JAHA.118.009058. PMID: 30371329. PMCID: PMC6404904.
Article7. Moscarelli M, Fiorentino F, Suleiman MS, Emanueli C, Reeves BC, Punjabi PP, Angelini GD. 2019; Remote ischaemic preconditioning in isolated aortic valve and coronary artery bypass surgery: a randomized trial†. Eur J Cardiothorac Surg. 55:905–912. DOI: 10.1093/ejcts/ezy404. PMID: 30544237. PMCID: PMC6477640.
Article8. Antonowicz SS, Cavallaro D, Jacques N, Brown A, Wiggins T, Haddow JB, Kapila A, Coull D, Walden A. 2018; Remote ischemic preconditioning for cardioprotection in elective inpatient abdominal surgery - a randomized controlled trial. BMC Anesthesiol. 18:76. DOI: 10.1186/s12871-018-0524-6. PMID: 29945555. PMCID: PMC6020340.
Article9. Cho YJ, Nam K, Kim TK, Choi SW, Kim SJ, Hausenloy DJ, Jeon Y. 2019; Sevoflurane, propofol and carvedilol block myocardial protection by limb remote ischemic preconditioning. Int J Mol Sci. 20:269. DOI: 10.3390/ijms20020269. PMID: 30641885. PMCID: PMC6359553.
Article10. Bunte S, Behmenburg F, Eckelskemper F, Mohr F, Stroethoff M, Raupach A, Heinen A, Hollmann MW, Huhn R. 2019; Cardioprotection by humoral factors released after remote ischemic preconditioning depends on anesthetic regimen. Crit Care Med. 47:e250–e255. DOI: 10.1097/CCM.0000000000003629. PMID: 30608281.
Article11. Lou B, Gao H, Zhou C. 2017; Myocardial protection by remote ischemic preconditioning in elective PCI: effect of ageing. Int J Cardiol. 243:105. DOI: 10.1016/j.ijcard.2017.03.142. PMID: 28747017.
Article12. Randhawa PK, Bali A, Virdi JK, Jaggi AS. 2018; Conditioning-induced cardioprotection: aging as a confounding factor. Korean J Physiol Pharmacol. 22:467–479. DOI: 10.4196/kjpp.2018.22.5.467. PMID: 30181694. PMCID: PMC6115349.
Article13. Kabil O, Banerjee R. 2014; Enzymology of H2S biogenesis, decay and signaling. Antioxid Redox Signal. 20:770–782. DOI: 10.1089/ars.2013.5339. PMID: 23600844. PMCID: PMC3910450.
Article14. Wu D, Wang J, Li H, Xue M, Ji A, Li Y. 2015; Role of hydrogen sulfide in ischemia-reperfusion injury. Oxid Med Cell Longev. 2015:186908. DOI: 10.1155/2015/186908. PMID: 26064416. PMCID: PMC4443900.
Article15. Li C, Hu M, Wang Y, Lu H, Deng J, Yan X. 2015; Hydrogen sulfide preconditioning protects against myocardial ischemia/reperfusion injury in rats through inhibition of endo/sarcoplasmic reticulum stress. Int J Clin Exp Pathol. 8:7740–7751. PMID: 26339339. PMCID: PMC4555667.16. Zhou C, Li H, Yao Y, Li L. 2014; Delayed remote ischemic preconditioning produces an additive cardioprotection to sevoflurane postconditioning through an enhanced heme oxygenase 1 level partly via nuclear factor erythroid 2-related factor 2 nuclear translocation. J Cardiovasc Pharmacol Ther. 19:558–566. DOI: 10.1177/1074248414524479. PMID: 24651515.
Article17. Kalakech H, Tamareille S, Pons S, Godin-Ribuot D, Carmeliet P, Furber A, Martin V, Berdeaux A, Ghaleh B, Prunier F. 2013; Role of hypoxia inducible factor-1α in remote limb ischemic preconditioning. J Mol Cell Cardiol. 65:98–104. DOI: 10.1016/j.yjmcc.2013.10.001. PMID: 24140799.
Article18. Ling K, Xu A, Chen Y, Chen X, Li Y, Wang W. 2019; Protective effect of a hydrogen sulfide donor on balloon injury-induced restenosis via the Nrf2/HIF-1α signaling pathway. Int J Mol Med. 43:1299–1310. DOI: 10.3892/ijmm.2019.4076. PMID: 30747216. PMCID: PMC6365080.
Article19. Mesbahzadeh B, Salarjavan H, Samarghandian S, Farkhondeh T. 2021; Chlorpyrifos with age-dependent effects in cardiac tissue of male rats. Curr Mol Pharmacol. doi: 10.2174/1874467214666210111105321. [Epub ahead of print]. DOI: 10.2174/1874467214666210111105321. PMID: 33430739.
Article20. Wen J, Chen Z, Wang S, Zhao M, Wang S, Zhao S, Zhang X. 2021; Age-related reductions in the excitability of phasic dorsal root ganglion neurons innervating the urinary bladder in female rats. Brain Res. 1752:147251. DOI: 10.1016/j.brainres.2020.147251. PMID: 33421375.
Article21. Kim KW, Cho HJ, Khaliq SA, Son KH, Yoon MS. 2020; Comparative analyses of mTOR/Akt and muscle atrophy-related signaling in aged respiratory and gastrocnemius muscles. Int J Mol Sci. 21:2862. DOI: 10.3390/ijms21082862. PMID: 32326050. PMCID: PMC7215274.
Article22. Gao C, Xu DQ, Gao CJ, Ding Q, Yao LN, Li ZC, Chai W. 2012; An exogenous hydrogen sulphide donor, NaHS, inhibits the nuclear factor κB inhibitor kinase/nuclear factor κb inhibitor/nuclear factor-κB signaling pathway and exerts cardioprotective effects in a rat hemorrhagic shock model. Biol Pharm Bull. 35:1029–1034. DOI: 10.1248/bpb.b110679. PMID: 22791148.
Article23. Zhang XJ, Meng XY, Huang XL, Dai HY, Wei P, Ling YL. 2011; [Administration of hydrogen sulfide intraperitoneally reverses the hyporesponsiveness of rat pulmonary artery induced by lipopolysaccharide and its relationship with carbon monoxide]. Zhongguo Wei Zhong Bing Ji Jiu Yi Xue. 23:200–203. Chinese. PMID: 21473819.24. Yang LJ, Wan R, Shen JQ, Shen J, Wang XP. 2013; Effect of L-cysteine on remote organ injury in rats with severe acute pancreatitis induced by bile-pancreatic duct obstruction. Hepatobiliary Pancreat Dis Int. 12:428–435. DOI: 10.1016/S1499-3872(13)60067-3. PMID: 23924502.
Article25. Yan J, Zhou B, Taheri S, Shi H. 2011; Differential effects of HIF-1 inhibition by YC-1 on the overall outcome and blood-brain barrier damage in a rat model of ischemic stroke. PLoS One. 6:e27798. DOI: 10.1371/journal.pone.0027798. PMID: 22110762. PMCID: PMC3218033.
Article26. Komsuoglu Celikyurt I, Utkan T, Ozer C, Gacar N, Aricioglu F. 2014; Effects of YC-1 on learning and memory functions of aged rats. Med Sci Monit Basic Res. 20:130–137. DOI: 10.12659/MSMBR.891064. PMID: 25144469. PMCID: PMC4148360.
Article27. Dong L, Fan Y, Shao X, Chen Z. 2011; Vitexin protects against myocardial ischemia/reperfusion injury in Langendorff-perfused rat hearts by attenuating inflammatory response and apoptosis. Food Chem Toxicol. 49:3211–3216. DOI: 10.1016/j.fct.2011.09.040. PMID: 22001368.
Article28. Diwan V, Kant R, Jaggi AS, Singh N, Singh D. 2008; Signal mechanism activated by erythropoietin preconditioning and remote renal preconditioning-induced cardioprotection. Mol Cell Biochem. 315:195–201. DOI: 10.1007/s11010-008-9808-3. PMID: 18528635.
Article29. Kakimoto Y, Tsuruyama T, Miyao M, Abiru H, Sumiyoshi S, Kotani H, Haga H, Tamaki K. 2013; The effectiveness and limitations of triphenyltetrazolium chloride to detect acute myocardial infarction at forensic autopsy. Am J Forensic Med Pathol. 34:242–247. DOI: 10.1097/PAF.0b013e31828879cd. PMID: 23949140.
Article30. Xu Z, Prathapasinghe G, Wu N, Hwang SY, Siow YL, O K. 2009; Ischemia-reperfusion reduces cystathionine-beta-synthase-mediated hydrogen sulfide generation in the kidney. Am J Physiol Renal Physiol. 297:F27–F35. DOI: 10.1152/ajprenal.00096.2009. PMID: 19439522.31. Siegel LM. 1965; A direct microdetermination for sulfide. Anal Biochem. 11:126–132. DOI: 10.1016/0003-2697(65)90051-5. PMID: 14328633.
Article32. Stipanuk MH, Beck PW. 1982; Characterization of the enzymic capacity for cysteine desulphhydration in liver and kidney of the rat. Biochem J. 206:267–277. DOI: 10.1042/bj2060267. PMID: 7150244. PMCID: PMC1158582.
Article33. Sadoh WE, Eregie CO, Nwaneri DU, Sadoh AE. 2014; The diagnostic value of both troponin T and creatinine kinase isoenzyme (CK-MB) in detecting combined renal and myocardial injuries in asphyxiated infants. PLoS One. 9:e91338. DOI: 10.1371/journal.pone.0091338. PMID: 24625749. PMCID: PMC3953387.
Article34. Amani M, Jeddi S, Ahmadiasl N, Usefzade N, Zaman J. 2013; Effect of HEMADO on level of CK-MB and LDH enzymes after ischemia/reperfusion injury in isolated rat heart. Bioimpacts. 3:101–104. DOI: 10.5681/bi.2013.003. PMID: 23878794. PMCID: PMC3713869.35. Tapuria N, Junnarkar S, Abu-Amara M, Fuller B, Seifalian AM, Davidson BR. 2012; Modulation of microcirculatory changes in the late phase of hepatic ischaemia-reperfusion injury by remote ischaemic preconditioning. HPB (Oxford). 14:87–97. DOI: 10.1111/j.1477-2574.2011.00407.x. PMID: 22221569. PMCID: PMC3277050.
Article36. Song Y, Ye YJ, Li PW, Zhao YL, Miao Q, Hou DY, Ren XP. 2016; The cardioprotective effects of late-phase remote preconditioning of trauma depends on neurogenic pathways and the activation of PKC and NF-κB (but not iNOS) in mice. J Cardiovasc Pharmacol Ther. 21:310–319. DOI: 10.1177/1074248415609435. PMID: 26450997.
Article37. Ferdinandy P, Hausenloy DJ, Heusch G, Baxter GF, Schulz R. 2014; Interaction of risk factors, comorbidities, and comedications with ischemia/reperfusion injury and cardioprotection by preconditioning, postconditioning, and remote conditioning. Pharmacol Rev. 66:1142–1174. DOI: 10.1124/pr.113.008300. PMID: 25261534.
Article38. Przyklenk K. 2011; Efficacy of cardioprotective 'conditioning' strategies in aging and diabetic cohorts: the co-morbidity conundrum. Drugs Aging. 28:331–343. DOI: 10.2165/11587190-000000000-00000. PMID: 21542657.39. Behmenburg F, Heinen A, Bruch LV, Hollmann MW, Huhn R. 2017; Cardioprotection by remote ischemic preconditioning is blocked in the aged rat heart in vivo. J Cardiothorac Vasc Anesth. 31:1223–1226. DOI: 10.1053/j.jvca.2016.07.005. PMID: 27793521.
Article40. Heinen A, Behmenburg F, Aytulun A, Dierkes M, Zerbin L, Kaisers W, Schaefer M, Meyer-Treschan T, Feit S, Bauer I, Hollmann MW, Huhn R. 2018; The release of cardioprotective humoral factors after remote ischemic preconditioning in humans is age- and sex-dependent. J Transl Med. 16:112. DOI: 10.1186/s12967-018-1480-0. PMID: 29703217. PMCID: PMC5921545.
Article41. Ji K, Xue L, Cheng J, Bai Y. 2016; Preconditioning of H2S inhalation protects against cerebral ischemia/reperfusion injury by induction of HSP70 through PI3K/Akt/Nrf2 pathway. Brain Res Bull. 121:68–74. DOI: 10.1016/j.brainresbull.2015.12.007. PMID: 26772627.42. Pan TT, Chen YQ, Bian JS. 2009; All in the timing: a comparison between the cardioprotection induced by H2S preconditioning and post-infarction treatment. Eur J Pharmacol. 616:160–165. DOI: 10.1016/j.ejphar.2009.05.023. PMID: 19482017.
Article43. Kubben N, Zhang W, Wang L, Voss TC, Yang J, Qu J, Liu GH, Misteli T. 2016; Repression of the antioxidant NRF2 pathway in premature aging. Cell. 165:1361–1374. DOI: 10.1016/j.cell.2016.05.017. PMID: 27259148. PMCID: PMC4893198.
Article44. Rivard A, Berthou-Soulie L, Principe N, Kearney M, Curry C, Branellec D, Semenza GL, Isner JM. 2000; Age-dependent defect in vascular endothelial growth factor expression is associated with reduced hypoxia-inducible factor 1 activity. J Biol Chem. 275:29643–29647. DOI: 10.1074/jbc.M001029200. PMID: 10882714.
Article45. Chang EI, Loh SA, Ceradini DJ, Chang EI, Lin SE, Bastidas N, Aarabi S, Chan DA, Freedman ML, Giaccia AJ, Gurtner GC. 2007; Age decreases endothelial progenitor cell recruitment through decreases in hypoxia-inducible factor 1alpha stabilization during ischemia. Circulation. 116:2818–2829. DOI: 10.1161/CIRCULATIONAHA.107.715847. PMID: 18040029.46. Shimizu Y, Nicholson CK, Lambert JP, Barr LA, Kuek N, Herszenhaut D, Tan L, Murohara T, Hansen JM, Husain A, Naqvi N, Calvert JW. 2016; Sodium sulfide attenuates ischemic-induced heart failure by enhancing proteasomal function in an Nrf2-dependent manner. Circ Heart Fail. 9:e002368. DOI: 10.1161/CIRCHEARTFAILURE.115.002368. PMCID: PMC4826721. PMID: 27056879.
Article47. Lohninger L, Tomasova L, Praschberger M, Hintersteininger M, Erker T, Gmeiner BM, Laggner H. 2015; Hydrogen sulphide induces HIF-1α and Nrf2 in THP-1 macrophages. Biochimie. 112:187–195. DOI: 10.1016/j.biochi.2015.03.009. PMID: 25795259.
Article48. Toth RK, Warfel NA. 2017; Strange bedfellows: nuclear factor, erythroid 2-like 2 (Nrf2) and hypoxia-inducible factor 1 (HIF-1) in tumor hypoxia. Antioxidants (Basel). 6:27. DOI: 10.3390/antiox6020027. PMID: 28383481. PMCID: PMC5488007.
Article49. Johansson K, Cebula M, Rengby O, Dreij K, Carlström KE, Sigmundsson K, Piehl F, Arnér ES. 2017; Cross talk in HEK293 cells between Nrf2, HIF, and NF-κB activities upon challenges with redox therapeutics characterized with single-cell resolution. Antioxid Redox Signal. 26:229–246. DOI: 10.1089/ars.2015.6419. PMID: 26415122. PMCID: PMC5704776.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Neurogenic pathways in remote ischemic preconditioning induced cardioprotection: Evidences and possible mechanisms
- Serial MRI of Hypoxic Brain Damage after Hydrogen Sulfide Exposure
- Mechanisms and Prospects of Ischemic Tolerance Induced by Cerebral Preconditioning
- Isolation of hydrogen sulfide producing escherichia coli
- Protective effect of combination therapy with ischemic preconditioning and rapamycin in fibrotic rat livers