Cancer Res Treat.
2021 Apr;53(2):576-583. 10.4143/crt.2020.801.
Knockdown of EMMPRIN (OX47) in MRMT-1 Carcinoma Cells Inhibits Tumor Growth and Decreases Cancer-Induced Bone Destruction and Pain
- Affiliations
-
- 1Department of Cell Biology and Genetics and Key Laboratory of Environment and Genes Related to Diseases, Xi’an Jiaotong University, Xi’an, China
- 2Shaanxi Key Laboratory of Brain Disorders and Institute of Basic and Translational Medicine, Xi’an Medical University, Xi’an, China
- 3Department of Obstetrics, The First Affiliated Hospital, Xi’an Jiaotong University, Xi’an, China
- KMID: 2514938
- DOI: http://doi.org/10.4143/crt.2020.801
Abstract
- Purpose
Bone destruction and pain caused by cancer is one of the most devastating complications of cancer patients with bone metastases, and it seriously affects the quality of patients’ life. Extracellular matrix metalloproteinase inducer (EMMPRIN) is a cell adhesion molecule with increased expression in a variety of tumors. This study focused to clarify the specific function of EMMPRIN in bone metastasis of breast cancer.
Materials and Methods
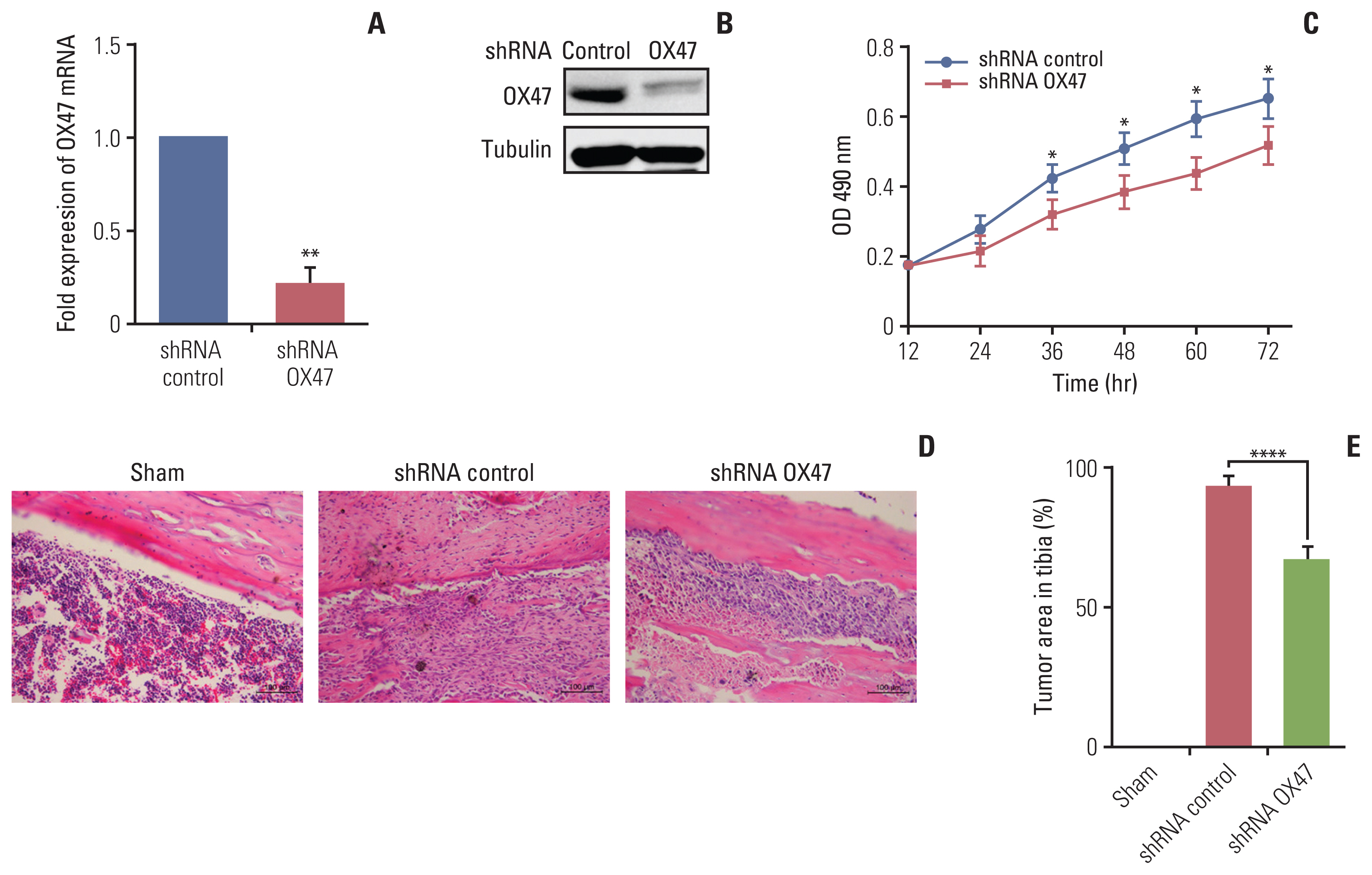
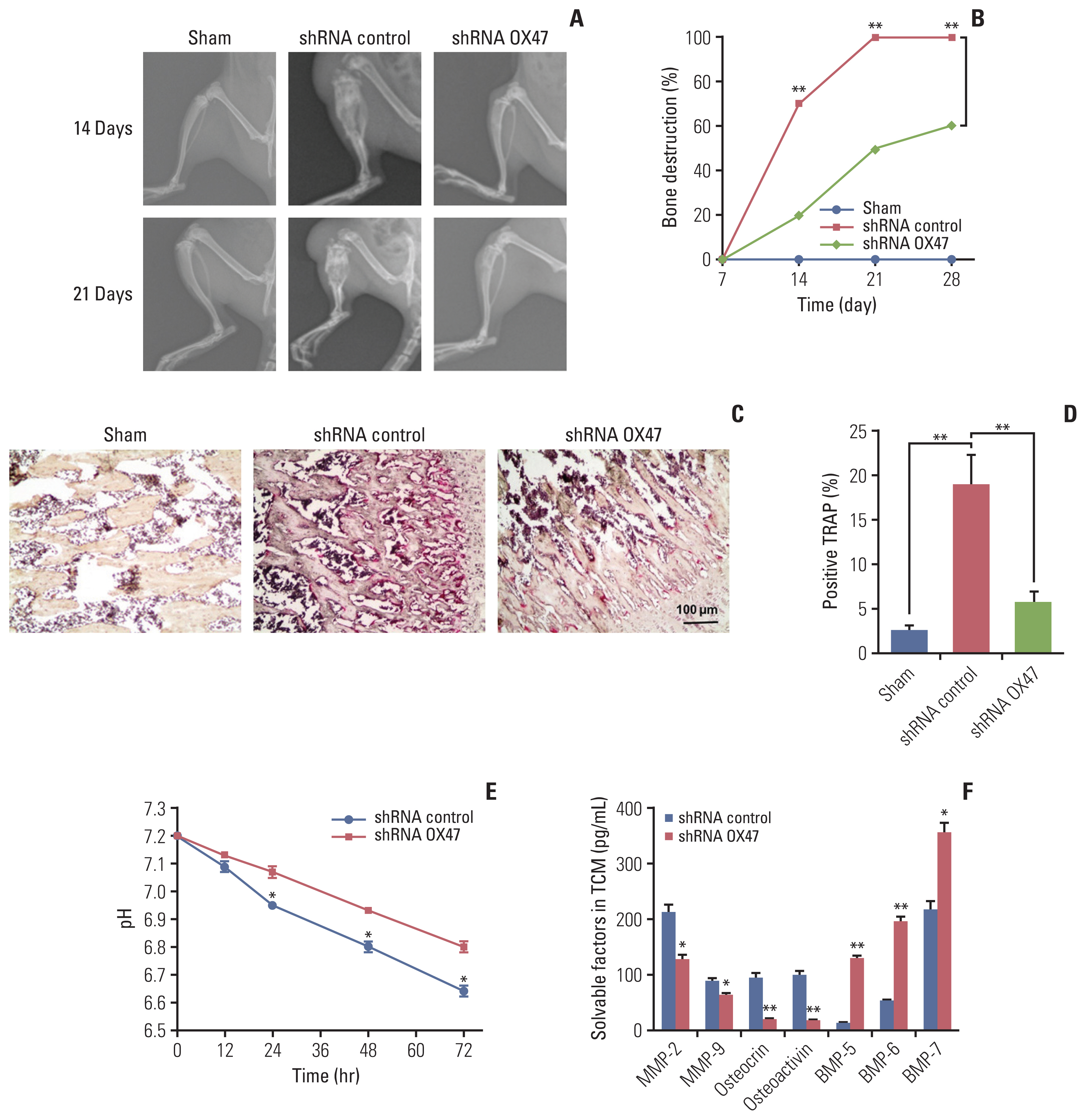
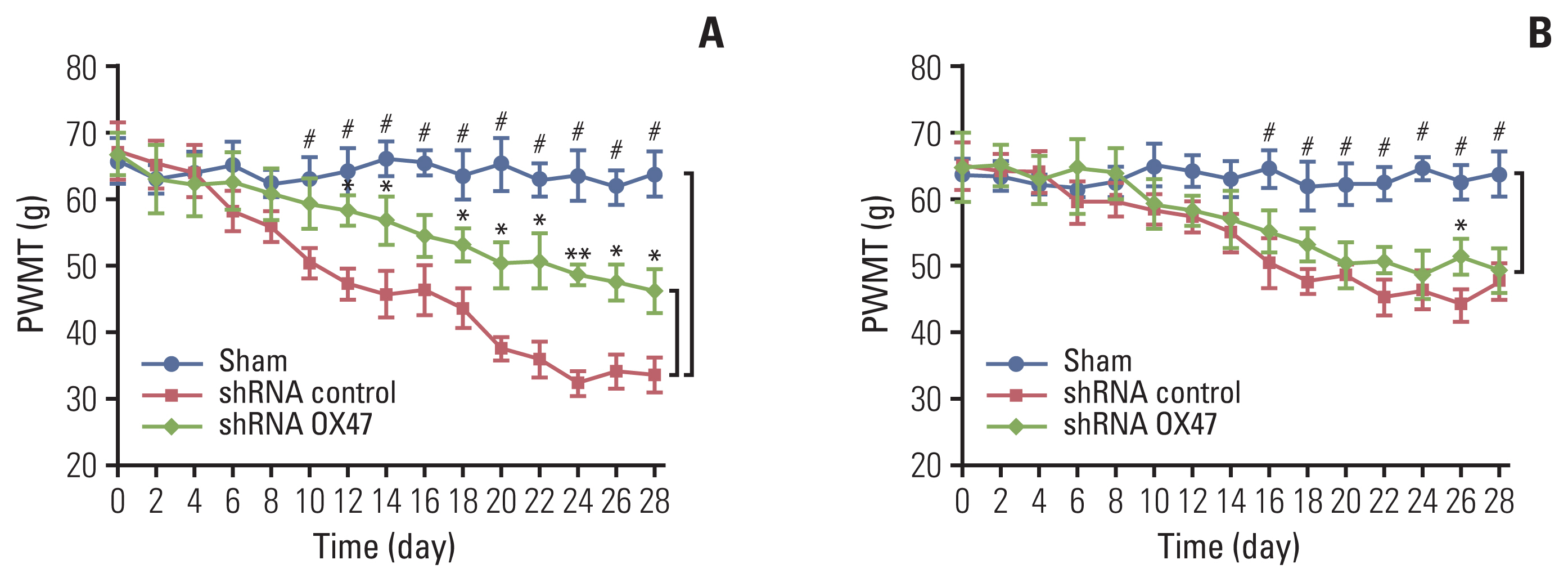
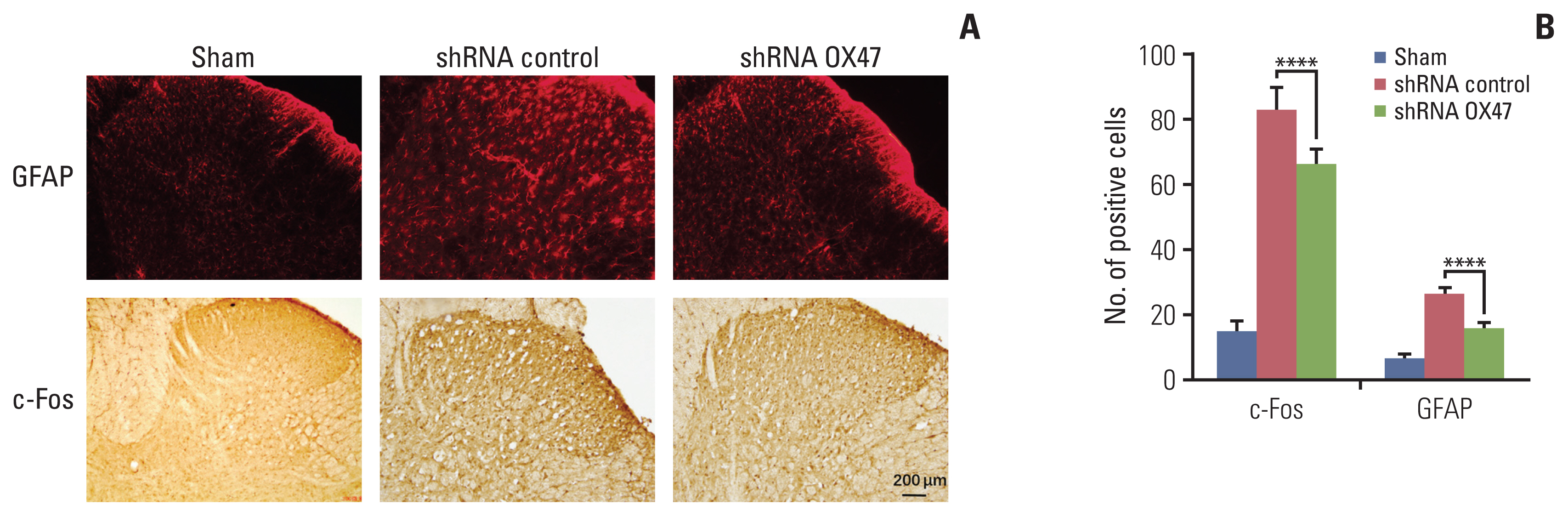
Adenovirus with shRNA-EMMPRIN was transfected into MRMT-1 rat breast carcinoma cells, and the MRMT-1 cells with different expression levels of EMMPRIN were implanted into the bone marrow cavity of rat tibia. Next, the effect of down-regulation of EMMPRIN was evaluated as follows: bone damage was detected by X-ray radiological and tartrate-resistant acid phosphatase staining; the tumor burden was evaluated by hematoxylin and eosin staining; the test of pain-related behaviors was assessed used the bilateral paw withdrawal mechanical threshold; and the levels of secretory factors in tumor conditioned medium were determined by using enzyme-linked immunosorbent assay.
Results
We found that down-regulation of EMMPRIN in tumor cells can simultaneously reduce tumor burden, relieve cancer-induced bone destruction and pain.
Conclusion
Materials and Methods
EMMPRIN is expected to be a therapeutic target for relieving bone metastasis of breast cancer and alleviating cancerinduced bone destruction and pain. The method of targeting EMMPRIN may be a promising strategy for the treatment of cancer in the future.
Keyword
Figure
Reference
-
References
1. Mercadante S. Malignant bone pain: pathophysiology and treatment. Pain. 1997; 69:1–18.
Article2. Mao-Ying QL, Zhao J, Dong ZQ, Wang J, Yu J, Yan MF, et al. A rat model of bone cancer pain induced by intra-tibia inoculation of Walker 256 mammary gland carcinoma cells. Biochem Biophys Res Commun. 2006; 345:1292–8.
Article3. Yoneda T, Hata K, Nakanishi M, Nagae M, Nagayama T, Wakabayashi H, et al. Involvement of acidic microenvironment in the pathophysiology of cancer-associated bone pain. Bone. 2011; 48:100–5.
Article4. Deryugina EI, Quigley JP. Tumor angiogenesis: MMP-mediated induction of intravasation- and metastasis-sustaining neovasculature. Matrix Biol. 2015; 44–46:94–112.
Article5. Sun J, Hemler ME. Regulation of MMP-1 and MMP-2 production through CD147/extracellular matrix metalloproteinase inducer interactions. Cancer Res. 2001; 61:2276–81.6. Caudroy S, Polette M, Nawrocki-Raby B, Cao J, Toole BP, Zucker S, et al. EMMPRIN-mediated MMP regulation in tumor and endothelial cells. Clin Exp Metastasis. 2002; 19:697–702.7. Riethdorf S, Reimers N, Assmann V, Kornfeld JW, Terracciano L, Sauter G, et al. High incidence of EMMPRIN expression in human tumors. Int J Cancer. 2006; 119:1800–10.
Article8. Ueland T, Laugsand LE, Vatten LJ, Janszky I, Platou C, Michelsen AE, et al. Extracellular matrix markers and risk of myocardial infarction: the HUNT Study in Norway. Eur J Prev Cardiol. 2017; 24:1161–7.
Article9. Cui HY, Wang SJ, Miao JY, Fu ZG, Feng F, Wu J, et al. CD147 regulates cancer migration via direct interaction with Annexin A2 and DOCK3-beta-catenin-WAVE2 signaling. Oncotarget. 2016; 7:5613–29.10. Chen Y, Gou X, Kong DK, Wang X, Wang J, Chen Z, et al. EMMPRIN regulates tumor growth and metastasis by recruiting bone marrow-derived cells through paracrine signaling of SDF-1 and VEGF. Oncotarget. 2015; 6:32575–85.
Article11. Kaito T, Morimoto T, Mori Y, Kanayama S, Makino T, Takenaka S, et al. BMP-2/7 heterodimer strongly induces bone regeneration in the absence of increased soft tissue inflammation. Spine J. 2018; 18:139–46.
Article12. Ishimaru Y, Bando T, Ohuchi H, Noji S, Mito T. Bone morphogenetic protein signaling in distal patterning and intercalation during leg regeneration of the cricket, Gryllus bimaculatus. Dev Growth Differ. 2018; 60:377–86.
Article13. Thomas G, Moffatt P, Salois P, Gaumond MH, Gingras R, Godin E, et al. Osteocrin, a novel bone-specific secreted protein that modulates the osteoblast phenotype. J Biol Chem. 2003; 278:50563–71.
Article14. Singh M, Del Carpio-Cano F, Belcher JY, Crawford K, Frara N, Owen TA, et al. Functional roles of osteoactivin in normal and disease processes. Crit Rev Eukaryot Gene Expr. 2010; 20:341–57.
Article15. Wang Q, Sun Y, Ren Y, Gao Y, Tian L, Liu Y, et al. Upregulation of EMMPRIN (OX47) in rat dorsal root ganglion contributes to the development of mechanical allodynia after nerve injury. Neural Plast. 2015; 2015:249756.
Article16. Medhurst SJ, Walker K, Bowes M, Kidd BL, Glatt M, Muller M, et al. A rat model of bone cancer pain. Pain. 2002; 96:129–40.
Article17. You HJ, Lei J, Sui MY, Huang L, Tan YX, Tjolsen A, et al. Endogenous descending modulation: spatiotemporal effect of dynamic imbalance between descending facilitation and inhibition of nociception. J Physiol. 2010; 588:4177–88.
Article18. Chen YK, Lei J, Jin L, Tan YX, You HJ. Dynamic variations of c-Fos expression in the spinal cord exposed to intramuscular hypertonic saline-induced muscle nociception. Eur J Pain. 2013; 17:336–46.
Article19. Xiong L, Edwards CK 3rd, Zhou L. The biological function and clinical utilization of CD147 in human diseases: a review of the current scientific literature. Int J Mol Sci. 2014; 15:17411–41.
Article20. Sun J, Hemler ME. Regulation of MMP-1 and MMP-2 production through CD147/extracellular matrix metalloproteinase inducer interactions. Cancer Res. 2001; 61:2276–81.21. Zhou J, Zhu P, Jiang JL, Zhang Q, Wu ZB, Yao XY, et al. Involvement of CD147 in overexpression of MMP-2 and MMP-9 and enhancement of invasive potential of PMA-differentiated THP-1. BMC Cell Biol. 2005; 6:25.22. Tang Y, Nakada MT, Rafferty P, Laraio J, McCabe FL, Millar H, et al. Regulation of vascular endothelial growth factor expression by EMMPRIN via the PI3K-Akt signaling pathway. Mol Cancer Res. 2006; 4:371–7.
Article23. Chen Y, Gou X, Ke X, Cui H, Chen Z. Human tumor cells induce angiogenesis through positive feedback between CD147 and insulin-like growth factor-I. PLoS One. 2012; 7:e40965.
Article24. Lakhan SE, Avramut M. Matrix metalloproteinases in neuropathic pain and migraine: friends, enemies, and therapeutic targets. Pain Res Treat. 2012; 2012:952906.
Article25. Moffatt P, Thomas G, Sellin K, Bessette MC, Lafreniere F, Akhouayri O, et al. Osteocrin is a specific ligand of the natriuretic Peptide clearance receptor that modulates bone growth. J Biol Chem. 2007; 282:36454–62.
Article26. Sheng MH, Wergedal JE, Mohan S, Lau KH. Osteoactivin is a novel osteoclastic protein and plays a key role in osteoclast differentiation and activity. FEBS Lett. 2008; 582:1451–8.
Article27. Rose AA, Pepin F, Russo C, Abou Khalil JE, Hallett M, Siegel PM. Osteoactivin promotes breast cancer metastasis to bone. Mol Cancer Res. 2007; 5:1001–14.
Article28. Zhou LT, Liu FY, Li Y, Peng YM, Liu YH, Li J. Gpnmb/osteoactivin, an attractive target in cancer immunotherapy. Neoplasma. 2012; 59:1–5.
Article29. Majidinia M, Sadeghpour A, Yousefi B. The roles of signaling pathways in bone repair and regeneration. J Cell Physiol. 2018; 233:2937–48.
Article30. Sabino MA, Mantyh PW. Pathophysiology of bone cancer pain. J Support Oncol. 2005; 3:15–24.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- NDRG2 Expression Decreases Tumor-Induced Osteoclast Differentiation by Down-regulating ICAM1 in Breast Cancer Cells
- Dissecting Tumor-Stromal Interactions in Breast Cancer Bone Metastasis
- Increased Expression of EMMPRIN and VEGF in the Rat Brain after Gamma Irradiation
- Knockdown of 14-3-3zeta enhances radiosensitivity and radio-induced apoptosis in CD133+ liver cancer stem cells
- Effects of the knockdown of hypoxia inducible factor-1alpha expression by adenovirus-mediated shRNA on angiogenesis and tumor growth in hepatocellular carcinoma cell lines