Lab Med Online.
2020 Apr;10(2):137-143. 10.3343/lmo.2020.10.2.137.
Evaluation of Automated Platelet Aggregation Test Using a Sysmex CS-5100 Analyzer
- Affiliations
-
- 1Department of Laboratory Medicine, Dong-A University College of Medicine, Busan, Korea
- 2Department of Laboratory Medicine, Ulsan Medical Center, Ulsan, Korea
- 3Seegene Medical Foundation, Busan, Korea
- KMID: 2512244
- DOI: http://doi.org/10.3343/lmo.2020.10.2.137
Abstract
- Background
The platelet aggregation test is widely used to measure antiplatelet therapy response and to detect platelet function disorders. CS-5100 (Sysmex Co., Japan) is a recently introduced coagulation analyzer that can also measure platelet aggregation. We evaluated the performance of CS-5100 in the platelet aggregation test for use in clinical laboratories.
Methods
We investigated the precision, stability, dilution test, and correlation of CS-5100 with a traditional aggregometer. Precision was tested using normal and patient samples. Stability was assessed over 5 different time points for 8 hours. The dilution test was performed with normal samples using ADP agonists. We tested correlations between the results of Chrono-log aggregometer (Chrono-Log Co., USA) and CS-5100 using 10 samples with 5 agonists each. We also calculated the reference range of 5 agonists using 22-30 normal samples.
Results
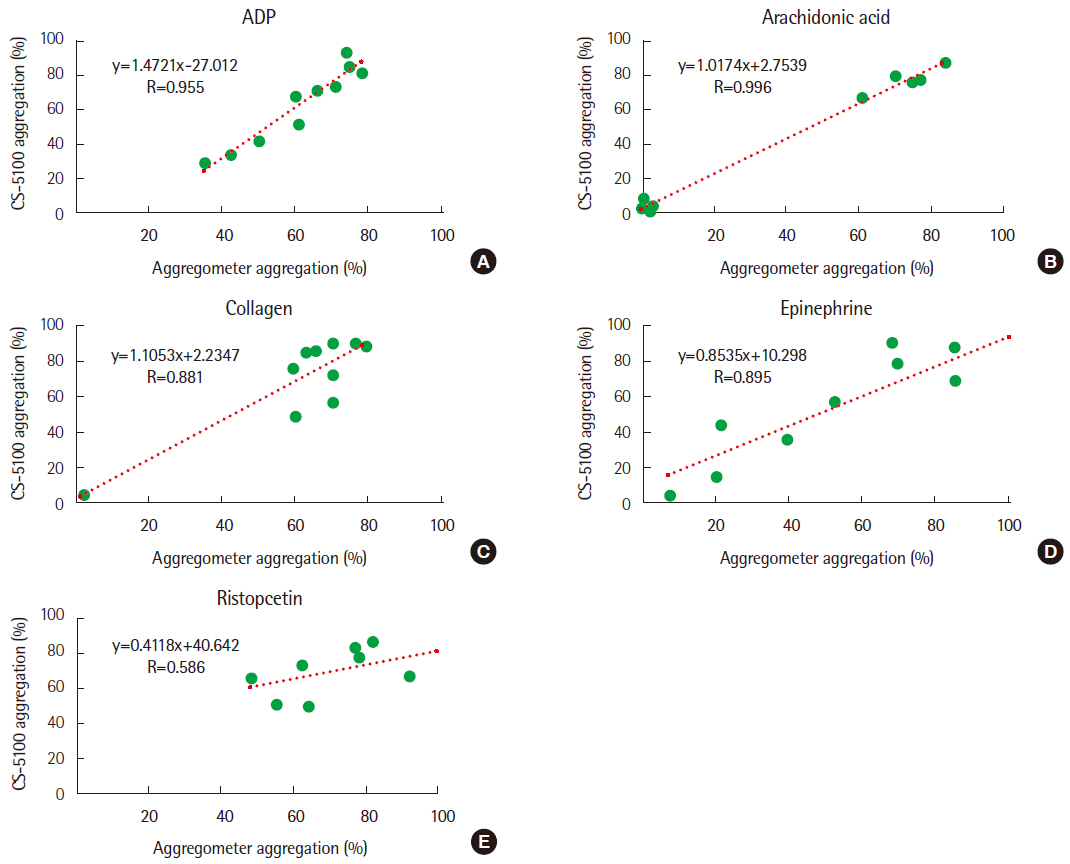
The coefficients of variation using normal samples were 7.45% and 3.27% for ADP and arachidonic acid, respectively. Stability was maintained for up to 2 hours in most samples. Dilution tests showed similar results until reaching a dilution factor of 2. Strong correlations of the results between Chrono-log and CS-5100 were found, except for ristocetin. The reference ranges of 5 reagents in CS-5100 were similar to those obtained with the Chrono-log aggregometer.
Conclusions
The performance of CS-5100 in platelet aggregation tests showed reliable results compared to a traditional aggregometer. CS-5100 can perform coagulation test and platelet aggregation test, simultaneously. Thus, CS-5100 can enable cost saving and reduce turn-around-time by reducing the inspection time.
Figure
Reference
-
1. Gale AJ. 2011; Continuing education course #2: Current understanding of hemostasis. Toxicol Pathol. 39:273–80. DOI: 10.1177/0192623310389474. PMID: 21119054. PMCID: PMC3126677.
Article2. Nurden AT, Nurden P. 2014; Congenital platelet disorders and understanding of platelet function. Br J Haematol. 165:165–78. DOI: 10.1111/bjh.12662. PMID: 24286193.
Article3. Konkle BA. 2011; Acquired disorders of platelet function. Hematology Am Soc Hematol Educ Program. 2011:391–6. DOI: 10.1182/asheducation-2011.1.391. PMID: 22160063.
Article4. Born GV, Cross MJ. 1963; The aggregation of blood platelets. J Physiol. 168:178–95. DOI: 10.1113/jphysiol.1963.sp007185. PMID: 14056485. PMCID: PMC1359417.
Article5. Harrison P. 2005; Platelet function analysis. Blood Rev. 19:111–23. DOI: 10.1016/j.blre.2004.05.002. PMID: 15603914.
Article6. Cattaneo M, Hayward CP, Moffat KA, Pugliano MT, Liu Y, Michelson AD. 2009; Results of a worldwide survey on the assessment of platelet function by light transmission aggregometry: a report from the platelet physiology subcommittee of the SSC of the ISTH. J Thromb Haemost. 7:1029. DOI: 10.1111/j.1538-7836.2009.03458.x. PMID: 19422455.
Article7. Sakayori T, Watanabe Y, Nakajima K, Misawa E, Kobayashi K, Kurono H, et al. 2016; Introduction and evaluation of light transmission platelet aggregation method on the Sysmex CS-series automated coagulation analyzer. Sysmex Journal International. 26:1–10.8. Cattaneo M, Cerletti C, Harrison P, Hayward CP, Kenny D, Nugent D, et al. 2013; Recommendations for the standardization of light transmission aggregometry: a consensus of the working party from the platelet physiology subcommittee of SSC/ISTH. J Thromb Haemost. 11:1183–9. DOI: 10.1111/jth.12231. PMID: 23574625.
Article9. Clinical and Laboratory Standard Institute. 2014; Evaluation of precision of quantitative measurement procedures; Approved guideline-third edition. CLSI document EP05-A3. Clinical and LaboratoryStandard Institute;Wayne, PA:10. Clinical and Laboratory Standard Institute. 2013; Measurement procedure comparison and bias estimation using patient samples; Approved guideline-third edition. CLSI document EP09-A3. Clinical and LaboratoryStandard Institute;Wayne, PA:11. Clinical and Laboratory Standard Institute. 2010; Defining, establishing, and verifying reference intervals in the clinical laboratory; Approved guideline-third edition. CLSI document EP28-A3C. Clinical and LaboratoryStandard Institute;Wayne, PA:12. Taylor R. 1990; Interpretation of the correlation coefficient: a basic review. J Diagn Med Sonogr. 6:35–9. DOI: 10.1177/875647939000600106.
Article13. Mukaka MM. 2012; Statistics corner: A guide to appropriate use of correla-tion coefficient in medical research. Malawi Med J. 24:69–71. PMID: 23638278. PMCID: PMC3576830.14. Hayward CP, Moffat KA, Pai M, Liu Y, Seecharan J, McKay H, et al. 2008; An evaluation of methods for determining reference intervals for light transmission platelet aggregation tests on samples with normal or reduced platelet counts. Thromb Haemost. 100:134–45. DOI: 10.1160/TH08-03-0183.
Article15. Miller CH, Rice AS, Garrett K, Stein SF. 2014; Gender, race and diet affect platelet function tests in normal subjects, contributing to a high rate of abnormal results. Br J Haematol. 165:842–53. DOI: 10.1111/bjh.12827. PMID: 24617520. PMCID: PMC4477706.
Article16. Ling LQ, Liao J, Niu Q, Wang X, Jia J, Zuo CH, et al. 2017; Evaluation of an automated light transmission aggregometry. Platelets. 28:712–9. DOI: 10.1080/09537104.2016.1265923. PMID: 28150526.
Article17. Lawrie AS, Kobayashi K, Lane PJ, Mackie IJ, Machin SJ. 2014; The automation of routine light transmission platelet aggregation. Int J Lab Hema-tol. 36:431–8. DOI: 10.1111/ijlh.12161. PMID: 24237750. PMCID: PMC4298021.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Performance Evaluation of the CS-5100 Coagulation Analyzer for Special Coagulation Parameters
- Analytical Performance of INNOVANCE Free Protein S Antigen on Sysmex CS-5100
- A Case of Spuriously Decreased White Blood Cell Count on an Automated Sysmex XN Hematology Analyzer: The Difference Between the WNR and WDF Channels
- Reliability of the DI-60 Digital Image Analyzer for Detecting Platelet Clumping and Obtaining Accurate Platelet Counts
- Evaluation of the Automated Hematology Analyzer Sysmex XN-2000 and the Accuracy of Differential Leukocyte Counts Using the Low WBC Mode