Korean J Gastroenterol.
2020 Sep;76(3):150-158. 10.4166/kjg.2020.76.3.150.
Arthrospira (Spirulina) platensis Attenuates Dextran Sulfate Sodium-induced Colitis in Mice by Suppressing Key Pro-inflammatory Cytokines
- Affiliations
-
- 1Faculty of Pharmaceutical Sciences, The University of British Columbia (UBC), Vancouver, British Columbia, Canada
- 2Departamento de Microbiologia, Imunologia e Parasitologia, Universidade Federal do Triangulo Mineiro (UFTM), Minas Gerais, Brazil
- 3Departamento de Farmacologia, Universidade Federal do Ceará (UFC), Fortaleza, CE, Brazil
- 4Departamento de Fisiologia, Faculdade de Medicina Estacio de Juazeiro do Norte (ESTACIO), Juazeiro do Norte, CE, Brazil
- 5Instituto de Patologia Tropical e Saúde Pública, Universidade Federal de Goiás, Goiânia, GO, Brazil
- KMID: 2506671
- DOI: http://doi.org/10.4166/kjg.2020.76.3.150
Abstract
- Background/Aims
Therapies aimed at modulating cytokines have been used to treat inflammatory illnesses, such as inflammatory bowel disease. On the other hand, patients may become intolerant, refractory, or present with several side effects. Arthrospira (Spirulina) platensis (SPI) is a blue-green microalga with bioactive molecules that have been evaluated to treat inflammatory diseases. On the other hand, few studies have examined their effects on the production of specific cytokines and the intestinal architecture in dextran sulfate sodium (DSS)-induced colitis. Therefore, this study examined the effects of a treatment using SPI in a murine model of intestinal inflammation.
Methods
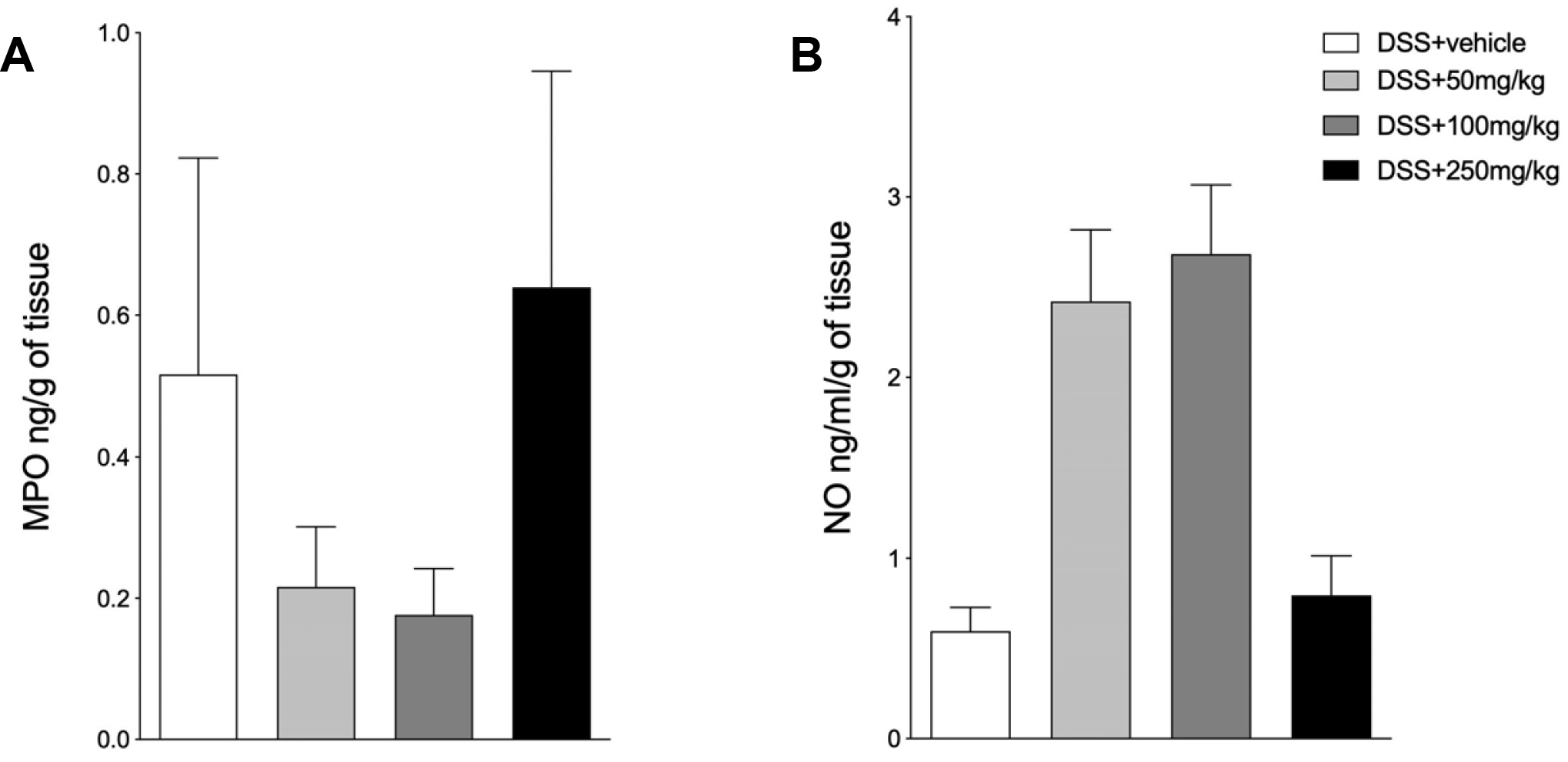
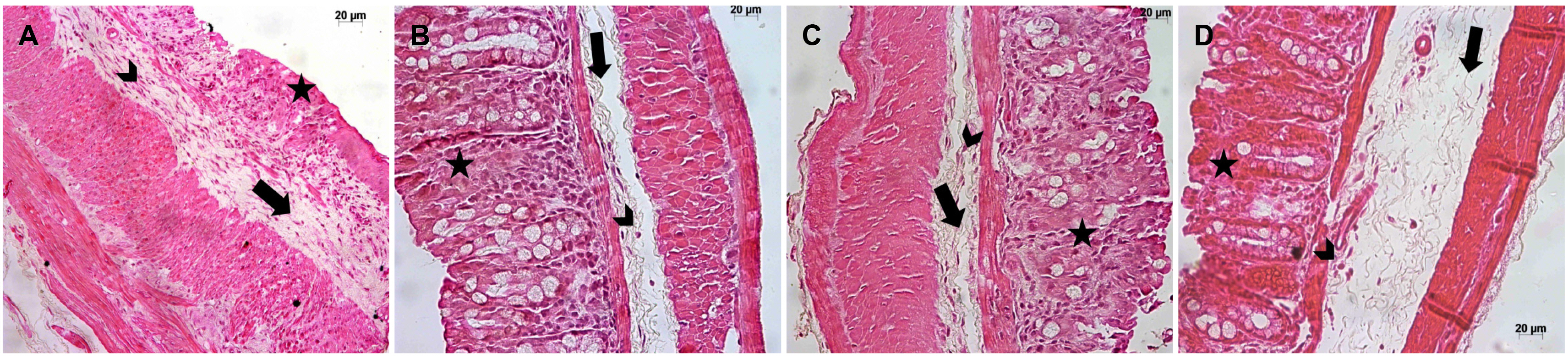
All mice (C57BL/6 male) were evaluated daily for their food and water intake, bodyweight variations, and clinical signs of disease. Colon inflammation was induced by exposure to DSS for 6 consecutive days. SPI was given orally at 50, 100, and 250 mg/kg/day. ELISA was performed to assess the production of cytokines. Myeloperoxidase and nitric oxide were also investigated. The level of microscopic damage was assessed by staining colon sections with hematoxylin and eosin.
Results
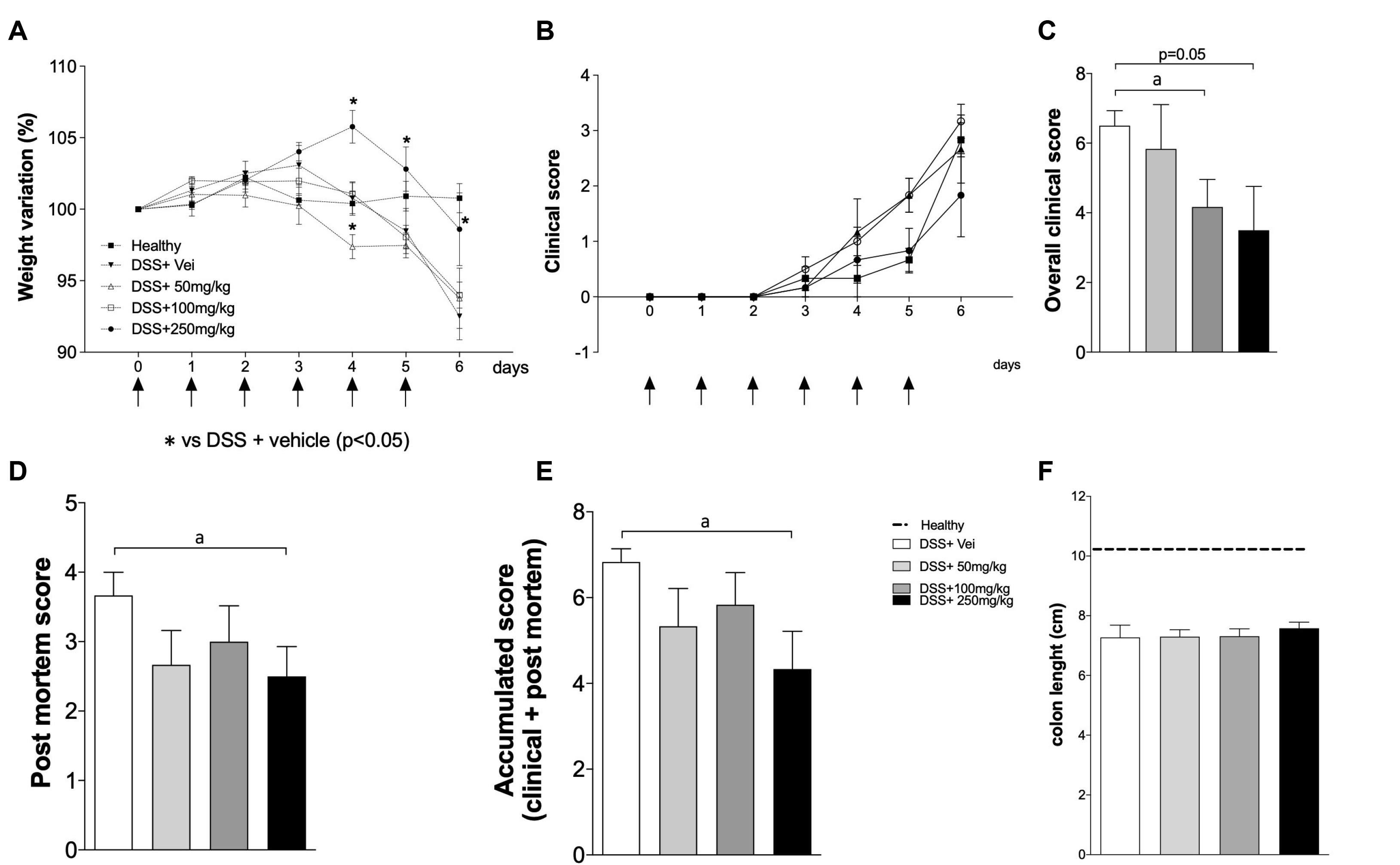
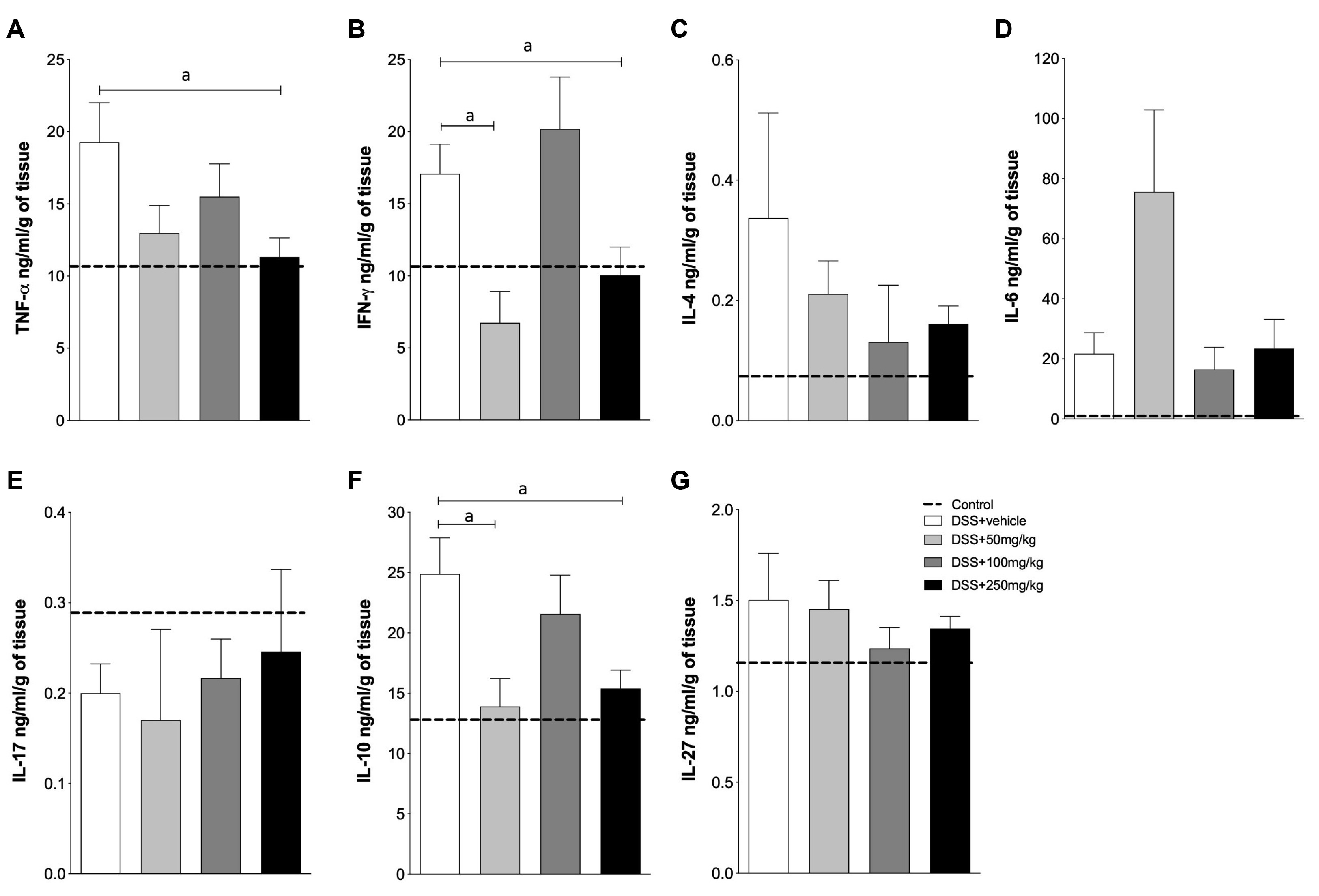
SPI attenuated the DSS-induced inflammation, with improvements in the clinical signs and a decrease in the production of inflammatory cytokines, such as tumor necrosis factor-α and interferon-γ. In addition, particularly at 250 mg/kg, SPI attenuated the severity of colitis by modulating the level of mucosal and submucosal cell infiltration, which preserved the epithelial barrier.
Conclusions
SPI may be an alternative source of bioactive molecules with immunomodulatory properties, and has great potential to be used in the treatment of inflammatory diseases.
Figure
Cited by 1 articles
-
The Usefulness of
Arthrospira (Spirulina) platensis in Inflammatory Bowel Disease
Ji Hyun Kim, Sung Chul Park
Korean J Gastroenterol. 2020;76(3):99-101. doi: 10.4166/kjg.2020.76.3.99.
Reference
-
1. Basso PJ, Fonseca MT, Bonfá G, et al. 2014; Association among genetic predisposition, gut microbiota, and host immune response in the etiopathogenesis of inflammatory bowel disease. Braz J Med Biol Res. 47:727–737. DOI: 10.1590/1414-431X20143932. PMID: 25075576. PMCID: PMC4143199.
Article2. Huang Y, Chen Z. 2016; Inflammatory bowel disease related innate immunity and adaptive immunity. Am J Transl Res. 8:2490–2497. DOI: 10.1007/978-981-15-4272-5_26. PMID: 32671761.3. Sales-Campos H, Basso PJ, Alves VB, et al. 2015; Classical and recent advances in the treatment of inflammatory bowel diseases. Braz J Med Biol Res. 48:96–107. DOI: 10.1590/1414-431x20143774. PMID: 25466162. PMCID: PMC4321214.
Article4. Kay RA. 1991; Microalgae as food and supplement. Crit Rev Food Sci Nutr. 30:555–573. DOI: 10.1080/10408399109527556. PMID: 1741951. PMCID: PMC7382111.
Article5. Lupatini AL, Colla LM, Canan C, Colla E. 2017; Potential application of microalga Spirulina platensis as a protein source. J Sci Food Agric. 97:724–732. DOI: 10.1002/jsfa.7987. PMID: 27507218.6. Remirez D, González R, Merino N, Rodriguez S, Ancheta O. 2002; Inhibitory effects of Spirulina in zymosan-induced arthritis in mice. Mediators Inflamm. 11:75–79. DOI: 10.1080/09629350220131917. PMID: 12061427. PMCID: PMC1781650.
Article7. Kumar N, Singh S, Patro N, Patro I. 2009; Evaluation of protective efficacy of Spirulina platensis against collagen-induced arthritis in rats. Inflammopharmacology. 17:181–190. DOI: 10.1007/s10787-009-0004-1. PMID: 19390977.
Article8. Ali EA, Barakat BM, Hassan R. 2015; Antioxidant and angiostatic effect of Spirulina platensis suspension in complete Freund's adjuvant-induced arthritis in rats. PLoS One. 10:e0121523. DOI: 10.1371/journal.pone.0121523. PMID: 25853428. PMCID: PMC4390336.
Article9. Pak W, Takayama F, Mine M, et al. 2012; Anti-oxidative and anti-inflammatory effects of spirulina on rat model of non-alcoholic steatohepatitis. J Clin Biochem Nutr. 51:227–234. DOI: 10.3164/jcbn.12-18. PMID: 23170052. PMCID: PMC3491249.
Article10. Mao TK, Van de Water J, Gershwin ME. 2005; Effects of a Spirulina-based dietary supplement on cytokine production from allergic rhinitis patients. J Med Food. 8:27–30. DOI: 10.1089/jmf.2005.8.27. PMID: 15857205.
Article11. Coskun ZK, Kerem M, Gurbuz N, et al. 2011; The study of biochemical and histopathological effects of spirulina in rats with TNBS-induced colitis. Bratisl Lek Listy. 112:235–243. PMID: 21682075.12. Abdel-Daim MM, Farouk SM, Madkour FF, Azab SS. 2015; Anti-inflammatory and immunomodulatory effects of Spirulina platensis in comparison to Dunaliella salina in acetic acid-induced rat experimental colitis. Immunopharmacol Immunotoxicol. 37:126–139. DOI: 10.3109/08923973.2014.998368. PMID: 25567297.13. Wirtz S, Popp V, Kindermann M, et al. 2017; Chemically induced mouse models of acute and chronic intestinal inflammation. Nat Protoc. 12:1295–1309. DOI: 10.1038/nprot.2017.044. PMID: 28569761.
Article14. Wirtz S, Neufert C, Weigmann B, Neurath MF. 2007; Chemically induced mouse models of intestinal inflammation. Nat Protoc. 2:541–546. DOI: 10.1038/nprot.2007.41. PMID: 17406617.
Article15. Morsy MA, Gupta S, Nair AB, Venugopala KN, Greish K, El-Daly M. 2019; Protective effect of Spirulina platensis extract against dextran-sulfate-sodium-induced ulcerative colitis in rats. Nutrients. 11:2309. DOI: 10.3390/nu11102309. PMID: 31569451. PMCID: PMC6836255.
Article16. Perše M, Cerar A. 2012; Dextran sodium sulphate colitis mouse model: traps and tricks. J Biomed Biotechnol. 2012:718617. DOI: 10.1155/2012/718617. PMID: 22665990. PMCID: PMC3361365.
Article17. Jirkof P, Leucht K, Cesarovic N, et al. 2013; Burrowing is a sensitive behavioural assay for monitoring general wellbeing during dextran sulfate sodium colitis in laboratory mice. Lab Anim. 47:274–283. DOI: 10.1177/0023677213493409. PMID: 23828853.
Article18. Sales-Campos H, de Souza PR, Basso PJ, et al. 2015; Aedes aegypti salivary gland extract ameliorates experimental inflammatory bowel disease. Int Immunopharmacol. 26:13–22. DOI: 10.1016/j.intimp.2015.03.002. PMID: 25770821.
Article19. Coutinho de Sousa B, Reis Machado J, da Silva MV, et al. 2017; Morinda citrifolia (Noni) fruit juice reduces inflammatory cytokines expression and contributes to the maintenance of intestinal mucosal integrity in DSS experimental colitis. Mediators Inflamm. 2017:6567432. DOI: 10.1155/2017/6567432. PMID: 28194046. PMCID: PMC5282445.20. Green LC, Ruiz de Luzuriaga K, Wagner DA, et al. 1981; Nitrate biosynthesis in man. Proc Natl Acad Sci U S A. 78:7764–7768. DOI: 10.1073/pnas.78.12.7764. PMID: 6950416. PMCID: PMC349351.
Article21. Fonseca-Camarillo G, Yamamoto-Furusho JK. 2015; Immunoregulatory pathways involved in inflammatory bowel disease. Inflamm Bowel Dis. 21:2188–2193. DOI: 10.1097/MIB.0000000000000477. PMID: 26111210. PMCID: PMC7049232.
Article22. Yamamoto-Furusho JK. 2018; Inflammatory bowel disease therapy: blockade of cytokines and cytokine signaling pathways. Curr Opin Gastroenterol. 34:187–193. DOI: 10.1097/MOG.0000000000000444. PMID: 29846261. PMCID: PMC7123802.23. Deng R, Chow TJ. 2010; Hypolipidemic, antioxidant, and antiinflammatory activities of microalgae Spirulina. Cardiovasc Ther. 28:e33–e45. DOI: 10.1111/j.1755-5922.2010.00200.x. PMID: 20633020. PMCID: PMC2907180.
Article24. Ku CS, Pham TX, Park Y, et al. 2013; Edible blue-green algae reduce the production of pro-inflammatory cytokines by inhibiting NF-κB pathway in macrophages and splenocytes. Biochim Biophys Acta. 1830:2981–2988. DOI: 10.1016/j.bbagen.2013.01.018. PMID: 23357040. PMCID: PMC3594481.
Article25. Gutiérrez-Rebolledo GA, Galar-Martínez M, García-Rodríguez RV, Chamorro-Cevallos GA, Hernández-Reyes AG, Martínez-Galero E. 2015; Antioxidant effect of Spirulina (arthrospira) maxima on chronic inflammation induced by Freund's complete adjuvant in rats. J Med Food. 18:865–871. DOI: 10.1089/jmf.2014.0117. PMID: 25599112. PMCID: PMC4523079.26. Ricciotti E, FitzGerald GA. 2011; Prostaglandins and inflammation. Arterioscler Thromb Vasc Biol. 31:986–1000. DOI: 10.1161/ATVBAHA.110.207449. PMID: 21508345. PMCID: PMC3081099.
Article27. Osma-Garcia IC, Punzón C, Fresno M, Díaz-Muñoz MD. 2016; Dose-dependent effects of prostaglandin E2 in macrophage adhesion and migration. Eur J Immunol. 46:677–688. DOI: 10.1002/eji.201545629. PMID: 26631603.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Treatment with Extracellular Vesicles from Giardia lamblia Alleviates Dextran Sulfate Sodium-Induced Colitis in C57BL/6 Mice
- Long-Term Effects of Bone Marrow-Derived Mesenchymal Stem Cells in Dextran Sulfate Sodium-Induced Murine Chronic Colitis
- The Usefulness of Arthrospira (Spirulina) platensis in Inflammatory Bowel Disease
- Alloferon Alleviates Dextran Sulfate Sodium-induced Colitis
- The Therapeutic Efficacy of Tonsil-derived Mesenchymal Stem Cells in Dextran Sulfate Sodium-induced Acute Murine Colitis Model