Immune Netw.
2015 Jun;15(3):135-141. 10.4110/in.2015.15.3.135.
Alloferon Alleviates Dextran Sulfate Sodium-induced Colitis
- Affiliations
-
- 1Department of Anatomy, Seoul National University College of Medicine, Seoul 110-799, Korea. genius29@snu.ac.kr
- 2Institute of Allergy and Clinical Immunology, Seoul National University Medical Research Center, Seoul 110-799, Korea.
- 3Department of Internal Medicine and Liver Research Institute, Seoul National University College of Medicine, Seoul 110-799, Korea.
- KMID: 2168039
- DOI: http://doi.org/10.4110/in.2015.15.3.135
Abstract
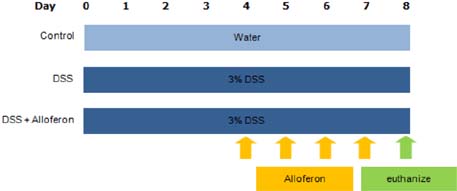
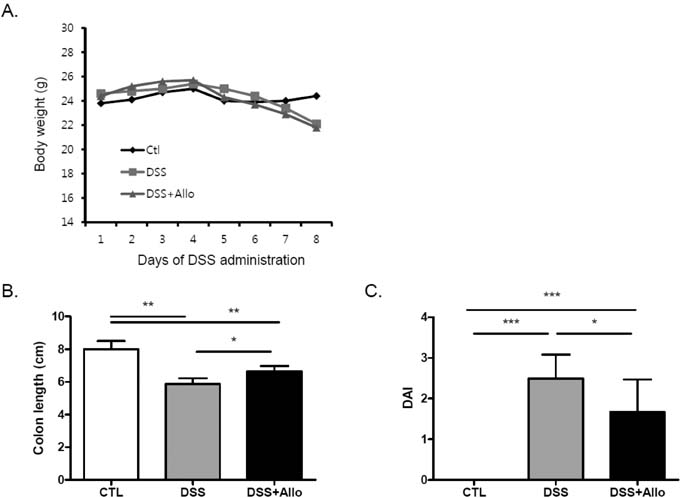
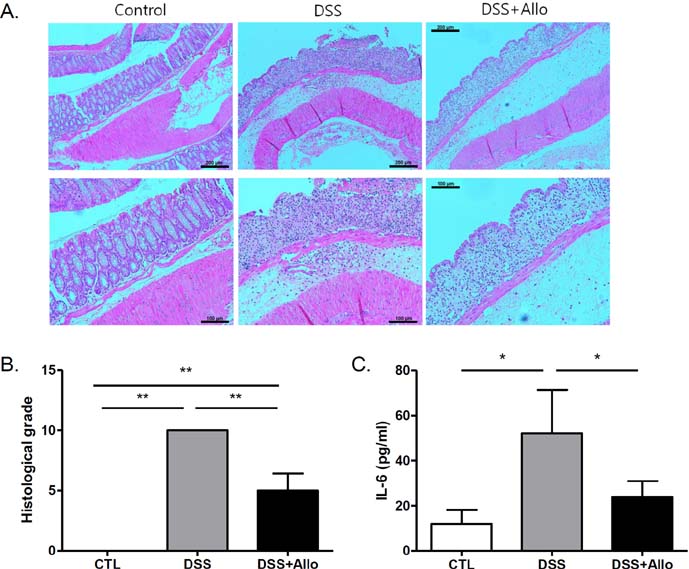
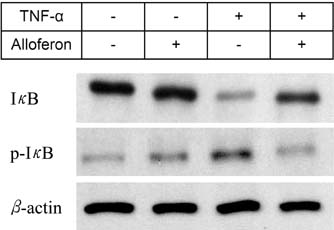
- Dysfunction of gut immune regulation is involved in mucosal damage in inflammatory bowel disease (IBD). However, there is still no efficacious immune-regulator for the treatment of IBD. Alloferon is a novel immune-modulatory peptide that was originally isolated from infected insects. It shows anti-inflammatory effects by the regulation of cytokine production by immune cells and their activities. Therefore, we investigated the effect of alloferon in a mouse model of colitis using dextran sulfate sodium (DSS). Colitis was induced by administration of DSS in drinking water for 7 consecutive days. It was confirmed by the presence of weight loss, diarrhea, hematochezia, and colon contraction. Alloferon was injected 4 days after DSS administration. We found that alloferon improved the pathogenesis of IBD based on the reduced disease activity index (DAI) and colon contraction. Edema, epithelial erosion, and immune cell infiltration were found in mice administered DSS, but the phenomena were reduced following alloferon treatment. The plasma level of IL-6, a classical pro-inflammatory cytokine in colitis, was also decreased by alloferon. Moreover, alloferon inhibited the TNF-alpha-induced degradation and phosphorylation of IkappaB in Colo205 colon cancer cells. Taken together, these results show that alloferon has anti-inflammatory effects and attenuates DSS-induced colitis.
MeSH Terms
Figure
Cited by 1 articles
-
Anti-inflammatory Effect of Alloferon on Ovalbumin-induced Asthma
Jane Jeon, Yejin Kim, Hyemin Kim, Jae Seung Kang, Wang Jae Lee
Immune Netw. 2015;15(6):304-312. doi: 10.4110/in.2015.15.6.304.
Reference
-
1. Xavier RJ, Podolsky DK. Unravelling the pathogenesis of inflammatory bowel disease. Nature. 2007; 448:427–434.
Article2. Baumgart DC, Sandborn WJ. Inflammatory bowel disease: clinical aspects and established and evolving therapies. Lancet. 2007; 369:1641–1657.
Article3. Thia KT, Loftus EV Jr, Sandborn WJ, Yang SK. An update on the epidemiology of inflammatory bowel disease in Asia. Am J Gastroenterol. 2008; 103:3167–3182.
Article4. Yang SK, Yun S, Kim JH, Park JY, Kim HY, Kim YH, Chang DK, Kim JS, Song IS, Park JB, Park ER, Kim KJ, Moon G, Yang SH. Epidemiology of inflammatory bowel disease in the Songpa-Kangdong district, Seoul, Korea, 1986-2005: a KASID study. Inflamm Bowel Dis. 2008; 814:542–549.
Article5. Kraus TA, Toy L, Chan L, Childs J, Cheifetz A, Mayer L. Failure to induce oral tolerance in Crohn's and ulcerative colitis patients: possible genetic risk. Ann N Y Acad Sci. 2004; 1029:225–238.
Article6. Chernysh S, Kim SI, Bekker G, Pleskach VA, Filatova NA, Anikin VB, Platonov VG, Bulet P. Antiviral and antitumor peptides from insects. Proc Natl Acad Sci U S A. 2002; 99:12628–12632.
Article7. Chernysh S, Irina K, Irina A. Anti-tumor activity of immunomodulatory peptide alloferon-1 in mouse tumor transplantation model. Int Immunopharmacol. 2012; 12:312–314.
Article8. Lee N, Bae S, Kim H, Kong JM, Kim HR, Cho BJ, Kim SJ, Seok SH, Hwang YI, Kim S, Kang JS, Lee WJ. Inhibition of lytic reactivation of Kaposi's sarcoma-associated herpesvirus by alloferon. Antivir Ther. 2011; 16:17–26.
Article9. Bae S, Oh K, Kim H, Kim Y, Kim HR, Hwang YI, Lee DS, Kang JS, Lee WJ. The effect of alloferon on the enhancement of NK cell cytotoxicity against cancer via the up-regulation of perforin/granzyme B secretion. Immunobiology. 2013; 218:1026–1033.
Article10. Kim Y, Lee SK, Bae S, Kim H, Park Y, Chu NK, Kim SG, Kim HR, Hwang YI, Kang JS, Lee WJ. The anti-inflammatory effect of alloferon on UVB-induced skin inflammation through the down-regulation of pro-inflammatory cytokines. Immunol Lett. 2013; 149:110–118.
Article11. Lee MJ, Lee JK, Choi JW, Lee CS, Sim JH, Cho CH, Lee KH, Cho IH, Chung MH, Kim HR, Ye SK. Interleukin-6 induces S100A9 expression in colonic epithelial cells through STAT3 activation in experimental ulcerative colitis. PLoS One. 2012; 7:e38801.
Article12. Sander LE, Obermeier F, Dierssen U, Kroy DC, Singh AK, Seidler U, Streetz KL, Lutz HH, Müller W, Tacke F, Trautwein C. Gp130 signaling promotes development of acute experimental colitis by facilitating early neutrophil/macrophage recruitment and activation. J Immunol. 2008; 181:3586–3594.
Article13. Lin Y, Yang X, Yue W, Xu X, Li B, Zou L, He R. Chemerin aggravates DSS-induced colitis by suppressing M2 macrophage polarization. Cell Mol Immunol. 2014; 11:355–366.
Article14. Li B, Alli R, Vogel P, Geiger TL. IL-10 modulates DSS-induced colitis through a macrophage-ROS-NO axis. Mucosal Immunol. 2014; 7:869–878.
Article15. Jarry A, Bossard C, Bou-Hanna C, Masson D, Espaze E, Denis MG, Laboisse CL. Mucosal IL-10 and TGF-beta play crucial roles in preventing LPS-driven, IFN-gamma-mediated epithelial damage in human colon explants. J Clin Invest. 2008; 118:1132–1142.16. Marrero JA, Matkowskyj KA, Yung K, Hecht G, Benya RV. Dextran sulfate sodium-induced murine colitis activates NF-kappaB and increases galanin-1 receptor expression. Am J Physiol Gastrointest Liver Physiol. 2000; 278:G797–G804.17. Andresen L, Jørgensen VL, Perner A, Hansen A, Eugen-Olsen J, Rask-Madsen J. Activation of nuclear factor kappaB in colonic mucosa from patients with collagenous and ulcerative colitis. Gut. 2005; 54:503–509.
Article18. Rogler G, Brand K, Vogl D, Page S, Hofmeister R, Andus T, Knuechel R, Baeuerle PA, Schölmerich J, Gross V. Nuclear factor kappaB is activated in macrophages and epithelial cells of inflamed intestinal mucosa. Gastroenterology. 1998; 115:357–369.
Article19. Kanarek N, London N, Schueler-Furman O, Ben-Neriah Y. Ubiquitination and degradation of the inhibitors of NF-kappaB. Cold Spring Harb Perspect Biol. 2010; 2:a000166.20. Chandel NS, Trzyna WC, McClintock DS, Schumacker PT. Role of oxidants in NF-kappa B activation and TNF-alpha gene transcription induced by hypoxia and endotoxin. J Immunol. 2000; 165:1013–1021.
Article21. Hanauer SB. Inflammatory bowel disease: epidemiology, pathogenesis, and therapeutic opportunities. Inflamm Bowel Dis. 2006; 12:Suppl 1. S3–S9.
Article22. Molodecky NA, Soon IS, Rabi DM, Ghali WA, Ferris M, Chernoff G, Benchimol EI, Panaccione R, Ghosh S, Barkema HW, Kaplan GG. Increasing incidence and prevalence of the inflammatory bowel diseases with time, based on systematic review. Gastroenterology. 2012; 142:46–54.
Article23. Ershov F, Kubanova A, Pinegin B, Shulzhenko A, Bekker G, Chernysh S. Allokine-α effect on the course of chronic genital herpes relapses. Materia Medica. 2003; 40:103–111.24. Chernysh S, Gordja N. The immune system of maggots of the blow fly (Calliphora vicina) as a source of medicinal drugs. J Evol Biochem Physiol. 2011; 47:524–533.
Article25. Taylor BS, de Vera ME, Ganster RW, Wang Q, Shapiro RA, Morris SM Jr, Billiar TR, Geller DA. Multiple NF-kappaB enhancer elements regulate cytokine induction of the human inducible nitric oxide synthase gene. J Biol Chem. 1998; 273:15148–15156.
Article26. Neurath MF, Finotto S, Fuss I, Boirivant M, Galle PR, Strober W. Regulation of T-cell apoptosis in inflammatory bowel disease: to die or not to die, that is the mucosal question. Trends Immunol. 2001; 22:21–26.
Article27. Grivennikov S, Karin E, Terzic J, Mucida D, Yu GY, Vallabhapurapu S, Scheller J, Rose-John S, Cheroutre H, Eckmann L, Karin M. IL-6 and Stat3 are required for survival of intestinal epithelial cells and development of colitis-associated cancer. Cancer Cell. 2009; 15:103–113.
Article28. Jin X, Zimmers TA, Zhang Z, Pierce RH, Koniaris LG. Interleukin-6 is an important in vivo inhibitor of intestinal epithelial cell death in mice. Gut. 2010; 59:186–196.
Article29. Triantafillidis JK, Nasioulas G, Kosmidis PA. Colorectal cancer and inflammatory bowel disease: epidemiology, risk factors, mechanisms of carcinogenesis and prevention strategies. Anticancer Res. 2009; 29:2727–2737.30. Ryu MJ, Anikin V, Hong SH, Jeon H, Yu YG, Yu MH, Chernysh S, Lee C. Activation of NF-kappaB by alloferon through down-regulation of antioxidant proteins and IkappaBalpha. Mol Cell Biochem. 2008; 313:91–102.
Article31. Cheng M, Chen Y, Xiao W, Sun R, Tian Z. NK cell-based immunotherapy for malignant diseases. Cell Mol Immunol. 2013; 10:230–252.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Histological Study of Experimental Colitis Induced by Dextran Sulfate Sodium
- Fine Structure of Goblet Cell Regeneration on Experimental Colitis Induced by Dextran Sulfate Sodium
- Treatment with Extracellular Vesicles from Giardia lamblia Alleviates Dextran Sulfate Sodium-Induced Colitis in C57BL/6 Mice
- Effects of Dextran Sulfate Sodium-Induced Ulcerative Colitis on the Disposition of Tofacitinib in Rats
- Anti-inflammatory effects of mulberry twig extracts on dextran sulfate sodium-induced colitis mouse model