Anat Cell Biol.
2020 Jun;53(2):216-227. 10.5115/acb.20.011.
Aster ageratoides Turcz. extract attenuates Alzheimer’s disease-associated cognitive deficits and vascular dementia-associated neuronal death
- Affiliations
-
- 1Department of Anatomy, College of Medicine, Konyang University, Daejeon, Korea
- 2Department of Herbal Crop Research, National Institute of Horticultural and Herbal Science, Rural Development Administration, Eumseong, Korea
- KMID: 2503451
- DOI: http://doi.org/10.5115/acb.20.011
Abstract
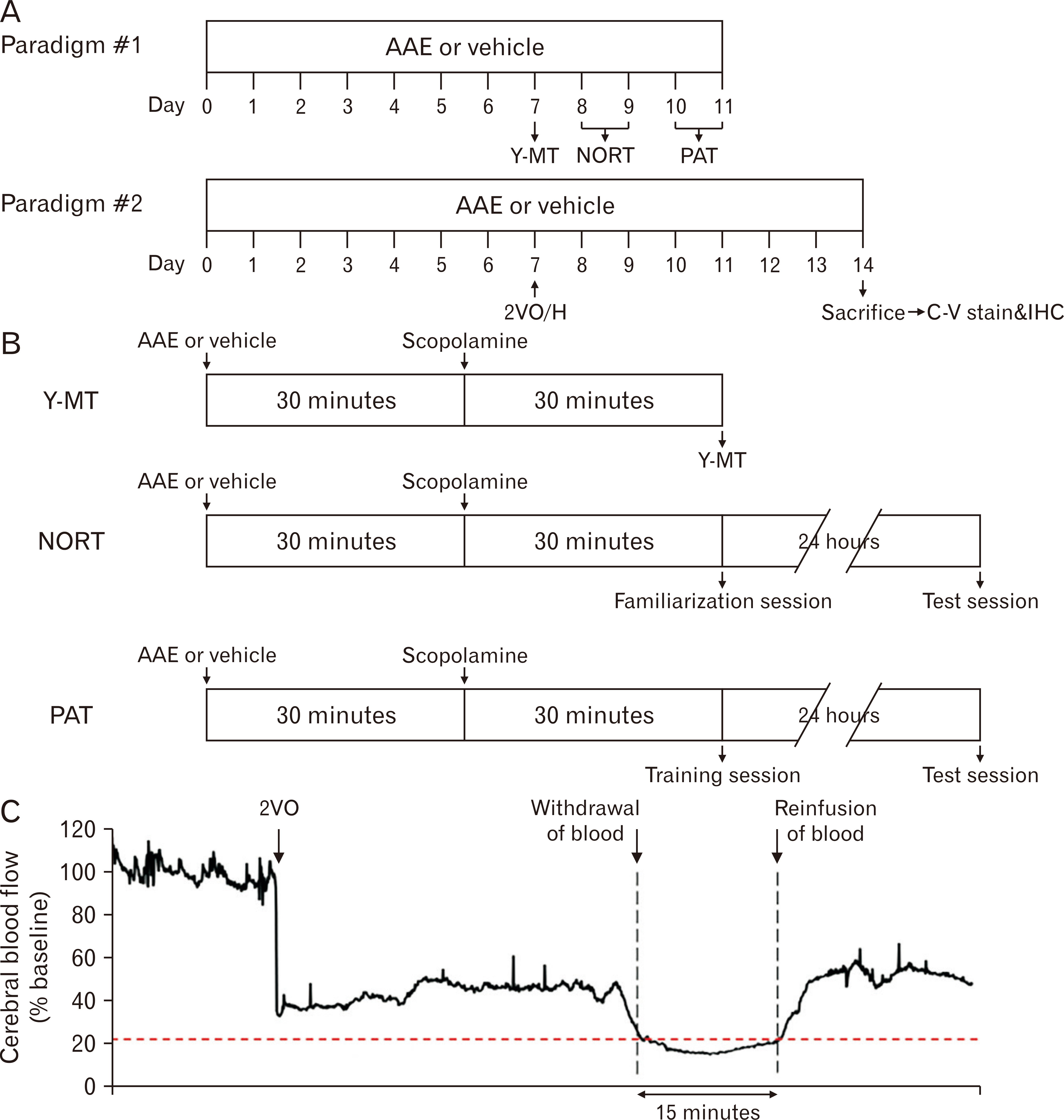
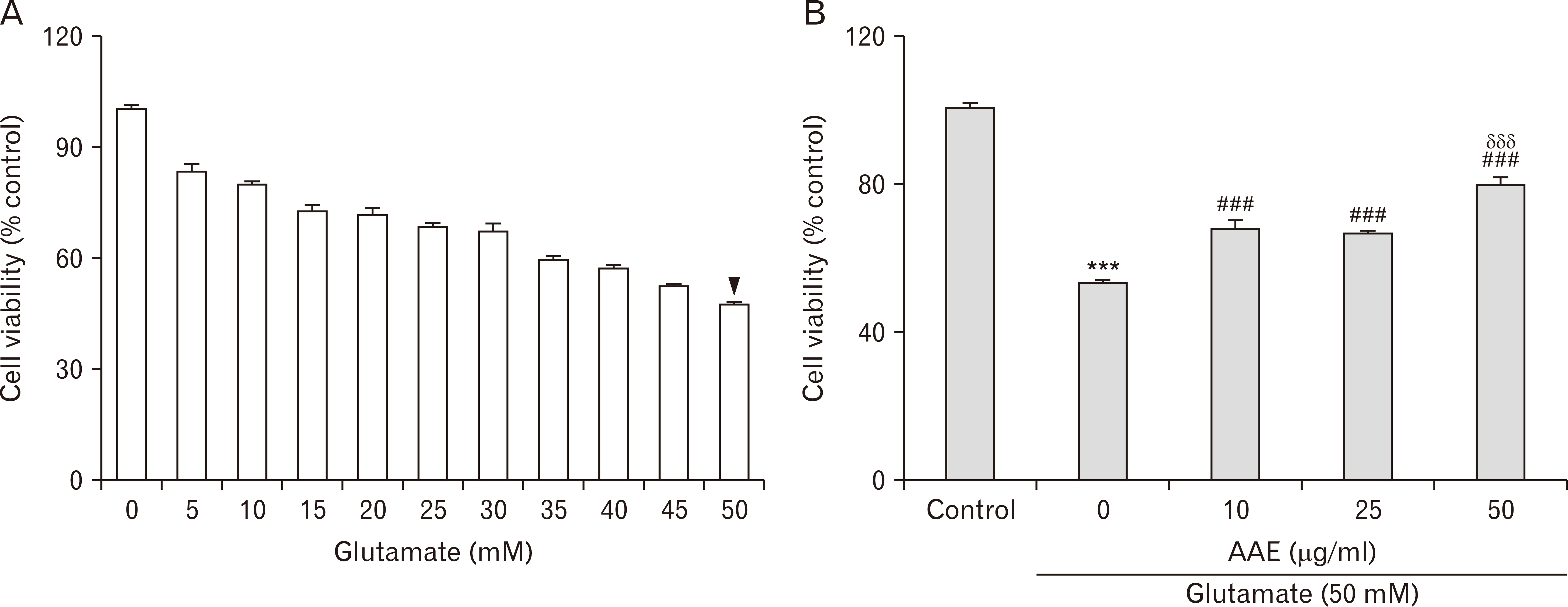
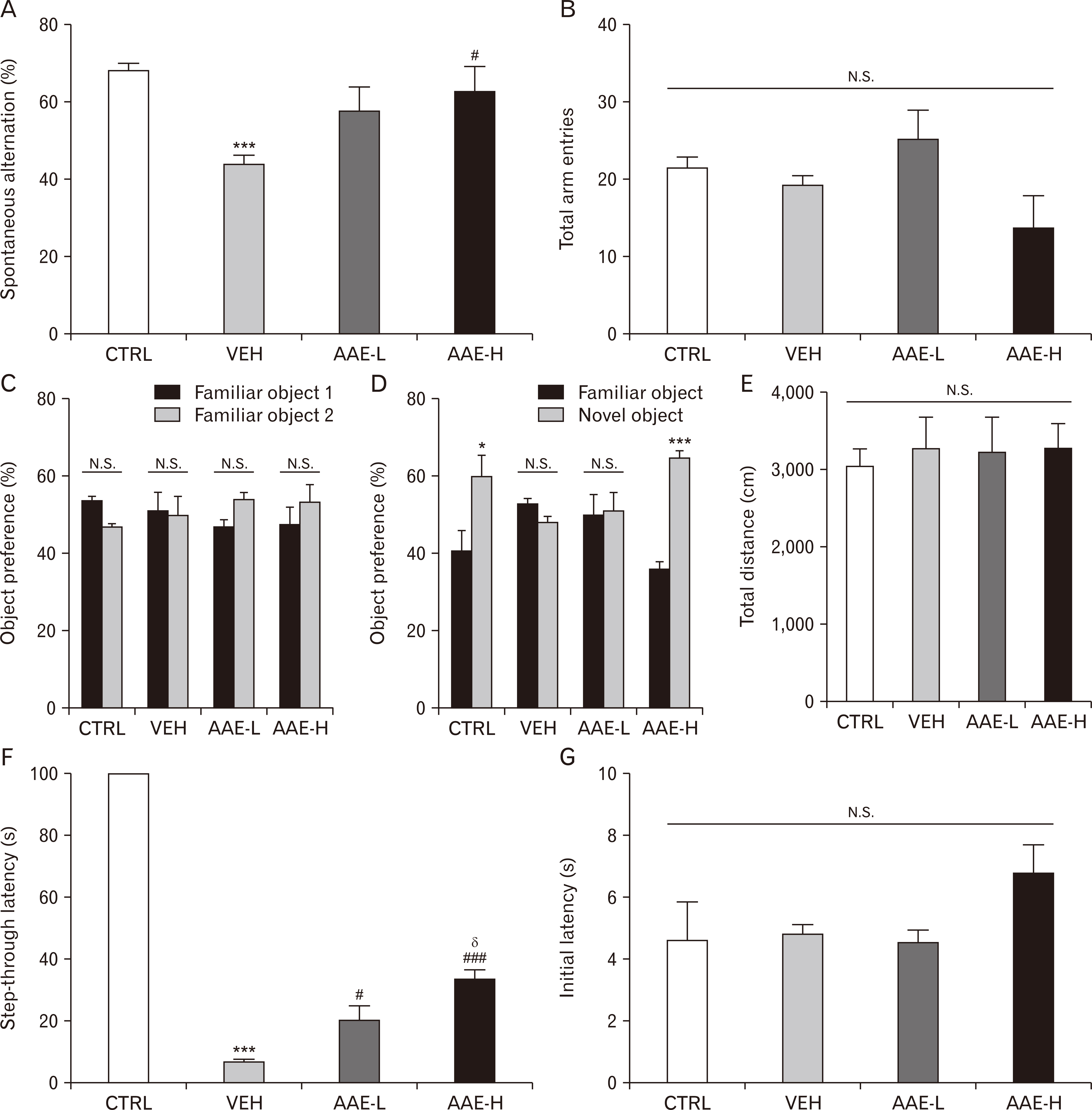
- Dementia is the common neurodegenerative disorder affecting the elderly, with a progressive cognitive decline and memory loss. Since Alzheimer’s disease (AD) and vascular dementia (VD) share key pathologies including oxidative damage, oral supplement of phytochemical medicines, which are well-known for their antioxidant properties, can be a viable therapy for both types of dementia. In this study, the therapeutic potential of the Aster ageratoides extract (AAE), an oriental drug with multiple medicinal properties, was tested on experimental rat models of AD and VD. After confirming the in vitro attenuation of neuronal excitotoxicity by AAE, rats were orally administered with AAE for 7 days and subsequently tested under 2 different experimental paradigms: efficacy screening against #1 AD and #2 VD. For paradigm #1, the rats received intraperitoneal scopolamine and subsequently underwent 3 different behavior tests i.e., the Y-maze, novel object recognition, and passive avoidance tests. For paradigm #2, the rats were operated with the 2-vessel occlusion and hypovolemia (2VO/H) technique, and at postoperative day 7, their hippocampal neuronal viability and the neuroinflammatory changes were quantified. The results showed that the scopolamine-induced impairment of memory performance was significantly improved by AAE intake. Furthermore, while the 2VO/H operation induced marked hippocampal neuronal death and microglial activation, both these effects were significantly attenuated by AAE supplements. Some of the aforementioned effects of AAE intake were dose-dependent. These results provided evidence that AAE supplements can exert anti-AD and -VD efficacies and suggested that AAE might be used as an edible phytotherapeutic for the 2 major types of dementia.
Keyword
Figure
Reference
-
1. Cunningham EL, McGuinness B, Herron B, Passmore AP. 2015; Dementia. Ulster Med J. 84:79–87. DOI: 10.1093/med/9780198796046.003.0011. PMID: 29801122. PMCID: PMC5997966.2. Castellani RJ, Rolston RK, Smith MA. 2010; Alzheimer disease. Dis Mon. 56:484–546. DOI: 10.1016/j.disamonth.2010.06.001. PMID: 20831921. PMCID: PMC2941917.
Article3. Bowler JV. 2005; Vascular cognitive impairment. J Neurol Neurosurg Psychiatry. 76(Suppl 5):v35–44. DOI: 10.1136/jnnp.2005.082313. PMID: 16291920. PMCID: PMC1765709.
Article4. Bruun M, Rhodius-Meester HFM, Koikkalainen J, Baroni M, Gjerum L, Lemstra AW, Barkhof F, Remes AM, Urhemaa T, Tolonen A, Rueckert D, van Gils M, Frederiksen KS, Waldemar G, Scheltens P, Mecocci P, Soininen H, Lötjönen J, Hasselbalch SG, van der Flier WM. 2018; Evaluating combinations of diagnostic tests to discriminate different dementia types. Alzheimers Dement (Amst). 10:509–18. DOI: 10.1016/j.dadm.2018.07.003. PMID: 30320203. PMCID: PMC6180596.
Article5. Cummings J, Lee G, Ritter A, Zhong K. 2018; Alzheimer's disease drug development pipeline: 2018. Alzheimers Dement (N Y). 4:195–214. DOI: 10.1016/j.trci.2018.03.009. PMID: 29955663. PMCID: PMC6021548.
Article6. Smith EE, Cieslak A, Barber P, Chen J, Chen YW, Donnini I, Edwards JD, Frayne R, Field TS, Hegedus J, Hanganu V, Ismail Z, Kanji J, Nakajima M, Noor R, Peca S, Sahlas D, Sharma M, Sposato LA, Swartz RH, Zerna C, Black SE, Hachinski V. 2017; Therapeutic strategies and drug development for vascular cognitive impairment. J Am Heart Assoc. 6:e005568. DOI: 10.1161/JAHA.117.005568. PMID: 28476873. PMCID: PMC5524100.
Article7. Santos CY, Snyder PJ, Wu WC, Zhang M, Echeverria A, Alber J. 2017; Pathophysiologic relationship between Alzheimer's disease, cerebrovascular disease, and cardiovascular risk: a review and synthesis. Alzheimers Dement (Amst). 7:69–87. DOI: 10.1016/j.dadm.2017.01.005. PMID: 28275702. PMCID: PMC5328683.
Article8. Govindpani K, McNamara LG, Smith NR, Vinnakota C, Waldvogel HJ, Faull RL, Kwakowsky A. 2019; Vascular dysfunction in Alzheimer's disease: a prelude to the pathological process or a consequence of it? J Clin Med. 8:E651. DOI: 10.3390/jcm8050651. PMID: 31083442. PMCID: PMC6571853.
Article9. Choi DW, Rothman SM. 1990; The role of glutamate neurotoxicity in hypoxic-ischemic neuronal death. Annu Rev Neurosci. 13:171–82. DOI: 10.1146/annurev.ne.13.030190.001131. PMID: 1970230.
Article10. Prentice H, Modi JP, Wu JY. 2015; Mechanisms of neuronal protection against excitotoxicity, endoplasmic reticulum stress, and mitochondrial dysfunction in stroke and neurodegenerative diseases. Oxid Med Cell Longev. 2015:964518. DOI: 10.1155/2015/964518. PMID: 26576229. PMCID: PMC4630664.
Article11. Luca M, Luca A, Calandra C. 2015; The role of oxidative damage in the pathogenesis and progression of Alzheimer's disease and vascular dementia. Oxid Med Cell Longev. 2015:504678. DOI: 10.1155/2015/504678. PMID: 26301043. PMCID: PMC4537746.
Article12. Alberdi E, Sánchez-Gómez MV, Ruiz A, Cavaliere F, Ortiz-Sanz C, Quintela-López T, Capetillo-Zarate E, Solé-Domènech S, Matute C. 2018; Mangiferin and morin attenuate oxidative stress, mitochondrial dysfunction, and neurocytotoxicity, induced by amyloid beta oligomers. Oxid Med Cell Longev. 2018:2856063. DOI: 10.1155/2018/2856063. PMID: 30013719. PMCID: PMC6022276.
Article13. Li Y, Zhang Z. 2015; Gastrodin improves cognitive dysfunction and decreases oxidative stress in vascular dementia rats induced by chronic ischemia. Int J Clin Exp Pathol. 8:14099–109. PMID: 26823723. PMCID: PMC4713509.14. Hussain T, Tan B, Yin Y, Blachier F, Tossou MC, Rahu N. 2016; Oxidative stress and inflammation: what polyphenols can do for us? Oxid Med Cell Longev. 2016:7432797. DOI: 10.1155/2016/7432797. PMID: 27738491. PMCID: PMC5055983.
Article15. Shin YH, Kim HJ, Ku JJ, Park KW, Choi K, Jeong HS, Kang SH. 2012; The folk plants in northern region of Chungcheongbuk-do. Korean J Plant Res. 25:707–18. DOI: 10.7732/kjpr.2012.25.6.707.
Article16. Clifford MN, Zheng W, Kuhnert N. 2006; Profiling the chlorogenic acids of aster by HPLC-MS(n). Phytochem Anal. 17:384–93. DOI: 10.1002/pca.935. PMID: 17144245.
Article17. Hwang SJ, Kim YW, Park Y, Lee HJ, Kim KW. 2014; Anti-inflammatory effects of chlorogenic acid in lipopolysaccharide-stimulated RAW 264.7 cells. Inflamm Res. 63:81–90. DOI: 10.1007/s00011-013-0674-4. PMID: 24127072.
Article18. Lou Z, Wang H, Zhu S, Ma C, Wang Z. 2011; Antibacterial activity and mechanism of action of chlorogenic acid. J Food Sci. 76:M398–403. DOI: 10.1111/j.1750-3841.2011.02213.x. PMID: 22417510.
Article19. Xu R, Kang Q, Ren J, Li Z, Xu X. 2013; Antitumor molecular mechanism of chlorogenic acid on inducting genes GSK-3 β and APC and inhibiting gene β -catenin. J Anal Methods Chem. 2013:951319. DOI: 10.1155/2013/951319. PMID: 23844319. PMCID: PMC3697783.20. Sato Y, Itagaki S, Kurokawa T, Ogura J, Kobayashi M, Hirano T, Sugawara M, Iseki K. 2011; In vitro and in vivo antioxidant properties of chlorogenic acid and caffeic acid. Int J Pharm. 403:136–8. DOI: 10.1016/j.ijpharm.2010.09.035. PMID: 20933071.21. Rebai O, Belkhir M, Sanchez-Gomez MV, Matute C, Fattouch S, Amri M. 2017; Differential molecular targets for neuroprotective effect of chlorogenic acid and its related compounds against glutamate induced excitotoxicity and oxidative stress in rat cortical neurons. Neurochem Res. 42:3559–72. DOI: 10.1007/s11064-017-2403-9. PMID: 28948515.
Article22. Carbone L. 2012; Pain management standards in the eighth edition of the Guide for the Care and Use of Laboratory Animals. J Am Assoc Lab Anim Sci. 51:322–8.23. Smith ML, Bendek G, Dahlgren N, Rosén I, Wieloch T, Siesjö BK. 1984; Models for studying long-term recovery following forebrain ischemia in the rat. 2. A 2-vessel occlusion model. Acta Neurol Scand. 69:385–401. DOI: 10.1111/j.1600-0404.1984.tb07822.x. PMID: 6464670.
Article24. Schneider CA, Rasband WS, Eliceiri KW. 2016; NIH Image to ImageJ: 25 years of image analysis. Nat Methods. 9:671–5. DOI: 10.1038/nmeth.2089. PMID: 22930834. PMCID: PMC5554542.
Article25. Jawaid T, Shakya AK, Siddiqui HH, Kamal M. 2014; Evaluation of Cucurbita maxima extract against scopolamine-induced amnesia in rats: implication of tumour necrosis factor alpha. Z Naturforsch C J Biosci. 69:407–17. DOI: 10.5560/znc.2014-0003. PMID: 25711042.26. Zhang ZY, Liu Z, Deng HH, Chen Q. 2018; Effects of acupuncture on vascular dementia (VD) animal models: a systematic review and meta-analysis. BMC Complement Altern Med. 18:302. DOI: 10.1186/s12906-018-2345-z. PMID: 30424749. PMCID: PMC6234685.
Article27. Glass CK, Saijo K, Winner B, Marchetto MC, Gage FH. 2010; Mechanisms underlying inflammation in neurodegeneration. Cell. 140:918–34. DOI: 10.1016/j.cell.2010.02.016. PMID: 20303880. PMCID: PMC2873093.
Article28. Dong XX, Wang Y, Qin ZH. 2009; Molecular mechanisms of excitotoxicity and their relevance to pathogenesis of neurodegenerative diseases. Acta Pharmacol Sin. 30:379–87. DOI: 10.1038/aps.2009.24. PMID: 19343058. PMCID: PMC4002277.
Article29. Carvajal FJ, Mattison HA, Cerpa W. 2016; Role of NMDA receptor-mediated glutamatergic signaling in chronic and acute neuropathologies. Neural Plast. 2016:2701526. DOI: 10.1155/2016/2701526. PMID: 27630777. PMCID: PMC5007376.
Article30. Shen KZ, Johnson SW. 2010; Ca2+ influx through NMDA-gated channels activates ATP-sensitive K+ currents through a nitric oxide-cGMP pathway in subthalamic neurons. J Neurosci. 30:1882–93. DOI: 10.1523/JNEUROSCI.3200-09.2010. PMID: 20130197. PMCID: PMC2824890.
Article31. Hiebl V, Ladurner A, Latkolik S, Dirsch VM. 2018; Natural products as modulators of the nuclear receptors and metabolic sensors LXR, FXR and RXR. Biotechnol Adv. 36:1657–98. DOI: 10.1016/j.biotechadv.2018.03.003. PMID: 29548878.
Article32. Luo X, Yu X, Liu S, Deng Q, Liu X, Peng S, Li H, Liu J, Cao Y. 2015; The role of targeting kinase activity by natural products in cancer chemoprevention and chemotherapy (Review). Oncol Rep. 34:547–54. DOI: 10.3892/or.2015.4029. PMID: 26044950.
Article33. Busch C, Burkard M, Leischner C, Lauer UM, Frank J, Venturelli S. 2015; Epigenetic activities of flavonoids in the prevention and treatment of cancer. Clin Epigenetics. 7:64. DOI: 10.1186/s13148-015-0095-z. PMID: 26161152. PMCID: PMC4497414.
Article34. Nobili S, Lippi D, Witort E, Donnini M, Bausi L, Mini E, Capaccioli S. 2009; Natural compounds for cancer treatment and prevention. Pharmacol Res. 59:365–78. DOI: 10.1016/j.phrs.2009.01.017. PMID: 19429468.
Article35. Srivastava P, Yadav RS. 2016; Efficacy of natural compounds in neurodegenerative disorders. Adv Neurobiol. 12:107–23. DOI: 10.1007/978-3-319-28383-8_7. PMID: 27651251.
Article36. Shao R, Xiao J. 2013; Natural products for treatment of Alzheimer's disease and related diseases: understanding their mechanism of action. Curr Neuropharmacol. 11:337. DOI: 10.2174/1570159X11311040001. PMID: 24381527. PMCID: PMC3744900.37. Olivares D, Deshpande VK, Shi Y, Lahiri DK, Greig NH, Rogers JT, Huang X. 2012; N-methyl D-aspartate (NMDA) receptor antagonists and memantine treatment for Alzheimer's disease, vascular dementia and Parkinson's disease. Curr Alzheimer Res. 9:746–58. DOI: 10.2174/156720512801322564. PMID: 21875407. PMCID: PMC5002349.
Article38. Gilles C, Ertlé S. 2000; Pharmacological models in Alzheimer's disease research. Dialogues Clin Neurosci. 2:247–55. PMID: 22034060. PMCID: PMC3181609.
Article39. Yoo DY, Woo YJ, Kim W, Nam SM, Lee BH, Yeun GH, Yoon YS, Won MH, Park JH, Hwang IK. 2011; Effects of a new synthetic butyrylcholinesterase inhibitor, HBU-39, on cell proliferation and neuroblast differentiation in the hippocampal dentate gyrus in a scopolamine-induced amnesia animal model. Neurochem Int. 59:722–8. DOI: 10.1016/j.neuint.2011.06.021. PMID: 21771623.
Article40. Venkat P, Chopp M, Chen J. 2015; Models and mechanisms of vascular dementia. Exp Neurol. 272:97–108. DOI: 10.1016/j.expneurol.2015.05.006. PMID: 25987538. PMCID: PMC4631710.
Article41. Eng LF, Ghirnikar RS. 1994; GFAP and astrogliosis. Brain Pathol. 4:229–37. DOI: 10.1111/j.1750-3639.1994.tb00838.x. PMID: 7952264.
Article42. Kempuraj D, Thangavel R, Natteru PA, Selvakumar GP, Saeed D, Zahoor H, Zaheer S, Iyer SS, Zaheer A. 2016; Neuroinflammation induces neurodegeneration. J Neurol Neurosurg Spine. 1:1003. PMID: 28127589. PMCID: PMC5260818.43. Tjalkens RB, Popichak KA, Kirkley KA. 2017; Inflammatory activation of microglia and astrocytes in manganese neurotoxicity. Adv Neurobiol. 18:159–81. DOI: 10.1007/978-3-319-60189-2_8. PMID: 28889267. PMCID: PMC6462217.
Article44. LeBlanc AC. 2005; The role of apoptotic pathways in Alzheimer's disease neurodegeneration and cell death. Curr Alzheimer Res. 2:389–402. DOI: 10.2174/156720505774330573. PMID: 16248844.
Article45. Onyango IG, Dennis J, Khan SM. 2016; Mitochondrial dysfunction in Alzheimer's disease and the rationale for bioenergetics based therapies. Aging Dis. 7:201–14. DOI: 10.14336/AD.2015.1007. PMID: 27114851. PMCID: PMC4809610.
Article