Korean J Physiol Pharmacol.
2020 May;24(3):223-232. 10.4196/kjpp.2020.24.3.223.
Sesamin induces A549 cell mitophagy and mitochondrial apoptosis via a reactive oxygen species-mediated reduction in mitochondrial membrane potential
- Affiliations
-
- 1Center of Morphological Experiment, Medical College of Yanbian University
- 2Department of Thoracic Surgery, Affiliated Hospital of Yanbian University, Yanji, Jilin 133000, China
- KMID: 2502504
- DOI: http://doi.org/10.4196/kjpp.2020.24.3.223
Abstract
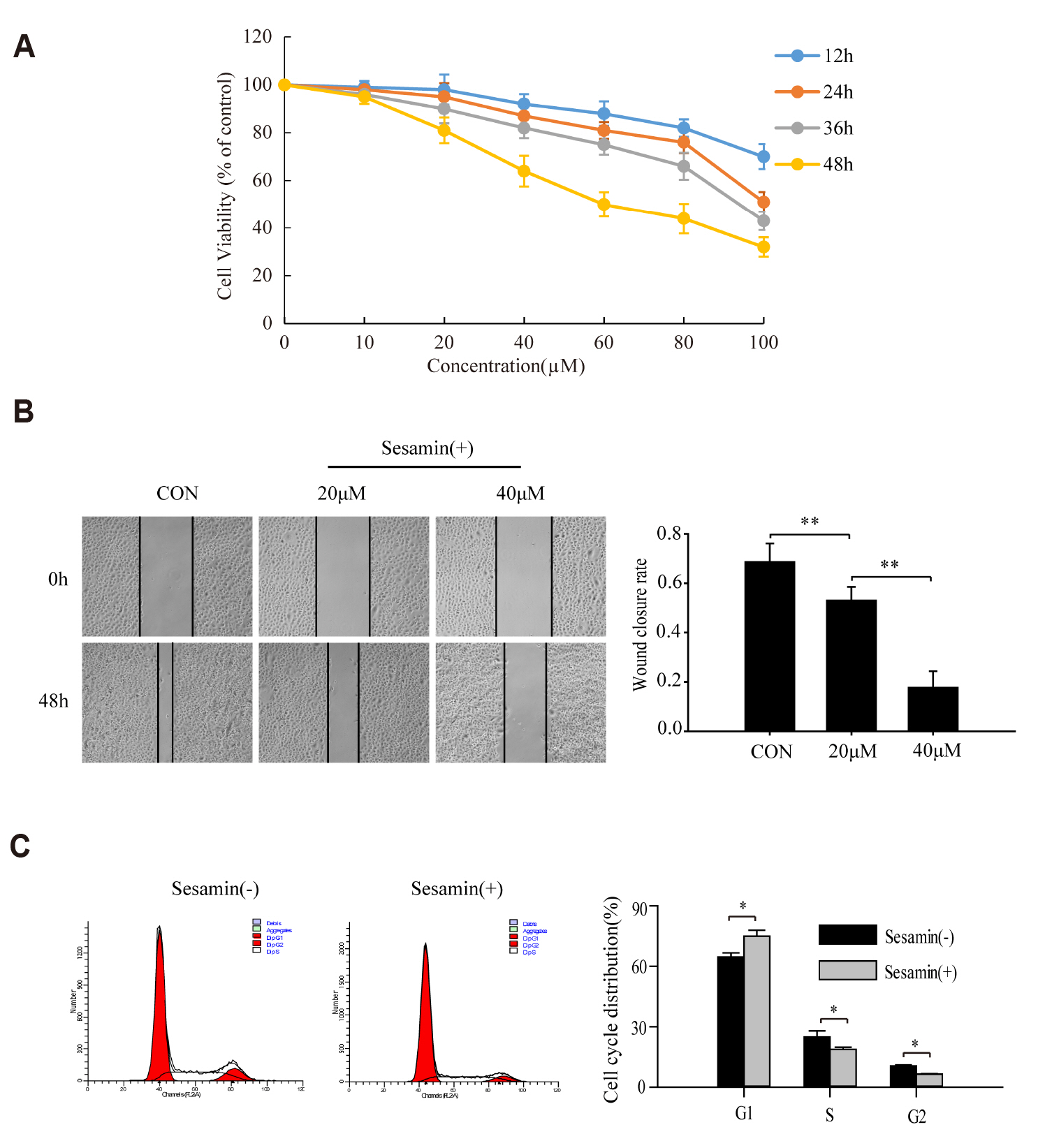
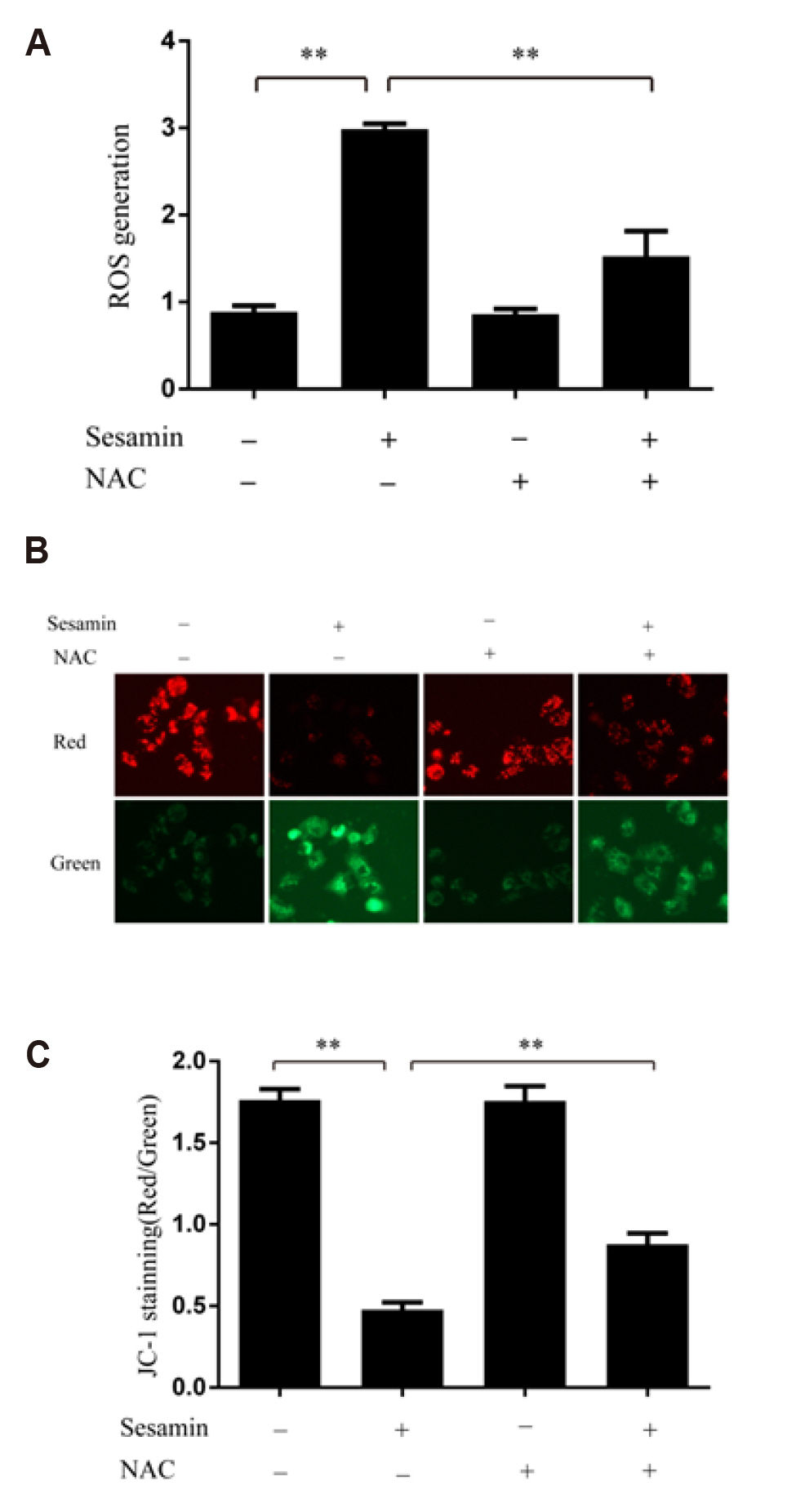
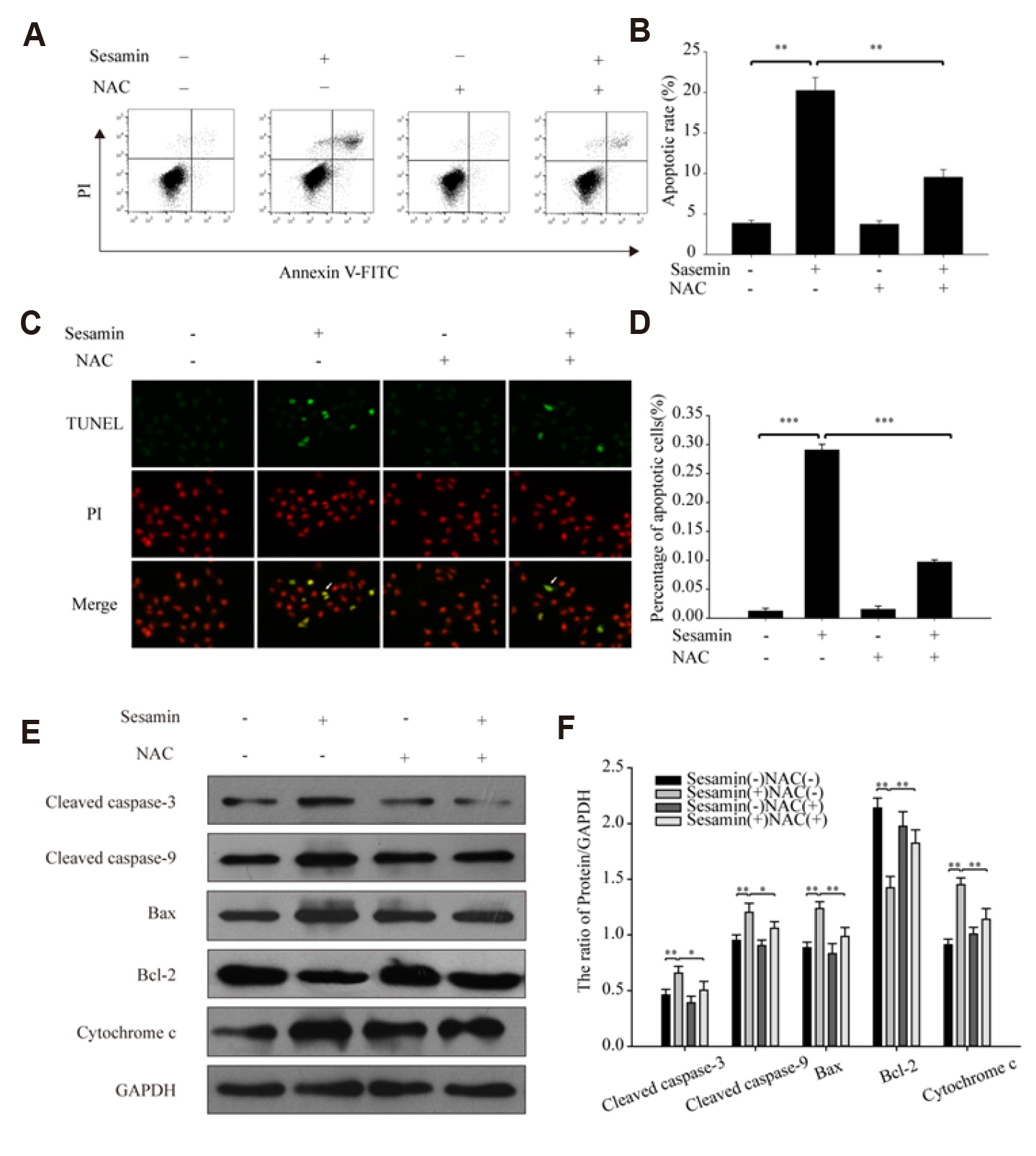
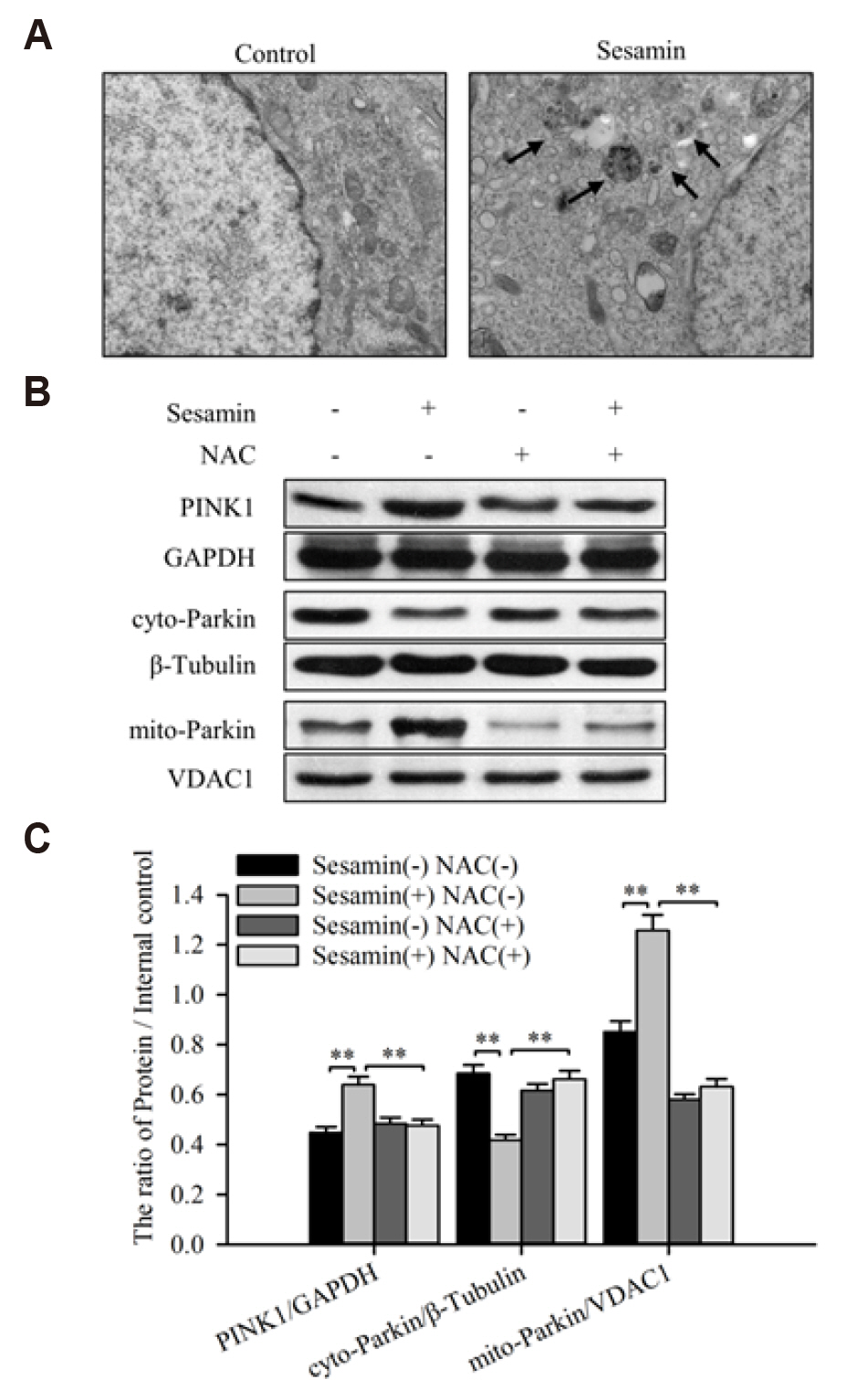
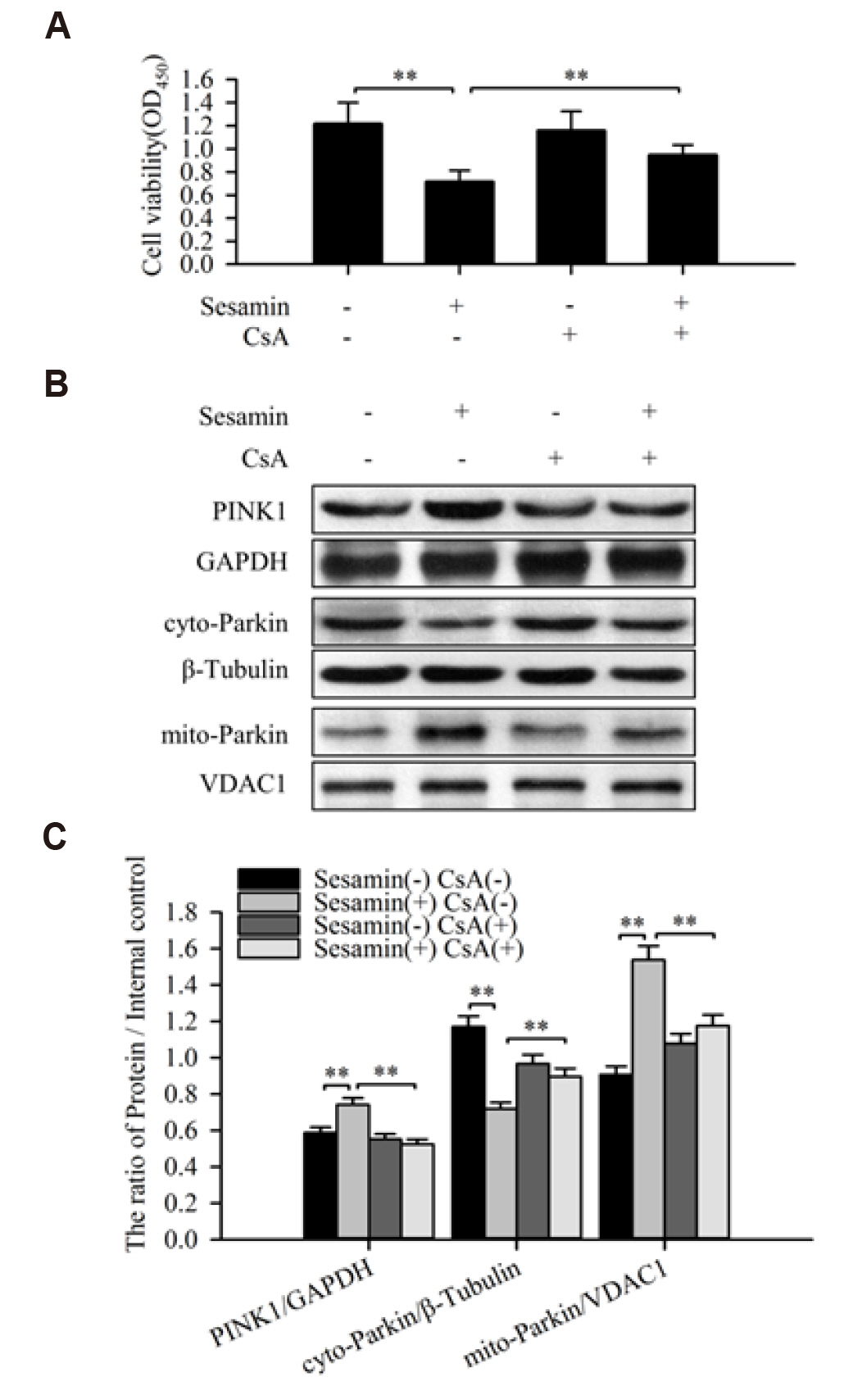
- Sesamin, a lipid-soluble lignin originally isolated from sesame seeds, which induces cancer cell apoptosis and autophagy. In the present study, has been reported that sesamin induces apoptosis via several pathways in human lung cancer cells. However, whether mitophagy is involved in sesamin induced lung cancer cell apotosis remains unclear. This study, the anticancer activity of sesamin in lung cancer was studied by reactive oxygen species (ROS) and mitophagy. A549 cells were treated with sesamin, and cell viability, migration ability, and cell cycle were assessed using the CCK8 assay, scratch-wound test, and flow cytometry, respectively. ROS levels, mitochondrial membrane potential, and apoptosis were examined by flow cytometric detection of DCFH-DA fluorescence and by using JC-1 and TUNEL assays. The results indicated that sesamin treatment inhibited the cell viability and migration ability of A549 cells and induced G0/G1 phase arrest. Furthermore, sesamin induced an increase in ROS levels, a reduction in mitochondrial membrane potential, and apoptosis accompanied by an increase in cleaved caspase-3 and cleaved caspase-9. Additionally, sesamin triggered mitophagy and increased the expression of PINK1 and translocation of Parkin from the cytoplasm to the mitochondria. However, the antioxidant N-acetyl-L-cysteine clearly reduced the oxidative stress and mitophagy induced by sesamin. Furthermore, we found that cyclosporine A (an inhibitor of mitophagy) decreased the inhibitory effect of sesamin on A549 cell viability. Collectively, our data indicate that sesamin exerts lethal effects on lung cancer cells through the induction of ROS-mediated mitophagy and mitochondrial apoptosis.
Keyword
Figure
Reference
-
1. Ferlay J, Shin HR, Bray F, Forman D, Mathers C, Parkin DM. 2010; Estimates of worldwide burden of cancer in 2008: GLOBOCAN 2008. Int J Cancer. 127:2893–2917. DOI: 10.1002/ijc.25516. PMID: 21351269.
Article2. Wakelee H, Kelly K, Edelman MJ. 2014; 50 Years of progress in the systemic therapy of non-small cell lung cancer. Am Soc Clin Oncol Educ Book. 177–189. DOI: 10.14694/EdBook_AM.2014.34.177. PMID: 24857075. PMCID: PMC5600272.
Article3. Majdalawieh AF, Massri M, Nasrallah GK. 2017; A comprehensive review on the anti-cancer properties and mechanisms of action of sesamin, a lignan in sesame seeds (Sesamum indicum). Eur J Pharmacol. 815:512–521. DOI: 10.1016/j.ejphar.2017.10.020. PMID: 29032105.
Article4. Xu P, Cai F, Liu X, Guo L. 2015; Sesamin inhibits lipopolysaccharide-induced proliferation and invasion through the p38-MAPK and NF-κB signaling pathways in prostate cancer cells. Oncol Rep. 33:3117–3123. DOI: 10.3892/or.2015.3888. PMID: 25845399.
Article5. Siao AC, Hou CW, Kao YH, Jeng KC. 2015; Effect of sesamin on apoptosis and cell cycle arrest in human breast cancer mcf-7 cells. Asian Pac J Cancer Prev. 16:3779–3783. DOI: 10.7314/APJCP.2015.16.9.3779. PMID: 25987037.
Article6. Dou H, Yang S, Hu Y, Xu D, Liu L, Li X. 2018; Sesamin induces ER stress-mediated apoptosis and activates autophagy in cervical cancer cells. Life Sci. 200:87–93. DOI: 10.1016/j.lfs.2018.03.003. PMID: 29505783.
Article7. Wang HM, Cheng KC, Lin CJ, Hsu SW, Fang WC, Hsu TF, Chiu CC, Chang HW, Hsu CH, Lee AY. 2010; Obtusilactone A and (-)-sesamin induce apoptosis in human lung cancer cells by inhibiting mitochondrial Lon protease and activating DNA damage checkpoints. Cancer Sci. 101:2612–2620. DOI: 10.1111/j.1349-7006.2010.01701.x. PMID: 21077998.
Article8. Fang Q, Zhu Y, Wang Q, Song M, Gao G, Zhou Z. 2019; Suppression of cyclooxygenase 2 increases chemosensitivity to sesamin through the Akt PI3K signaling pathway in lung cancer cells. Int J Mol Med. 43:507–516. DOI: 10.3892/ijmm.2018.3939. PMID: 30365050.9. Chen Y, Li H, Zhang W, Qi W, Lu C, Huang H, Yang Z, Liu B, Zhang L. 2020; Sesamin suppresses NSCLC cell proliferation and induces apoptosis via Akt/p53 pathway. Toxicol Appl Pharmacol. 387:114848. DOI: 10.1016/j.taap.2019.114848. PMID: 31809756.
Article10. Chance B, Sies H, Boveris A. 1979; Hydroperoxide metabolism in mammalian organs. Physiol Rev. 59:527–605. DOI: 10.1152/physrev.1979.59.3.527. PMID: 37532.
Article11. Dawson TL, Gores GJ, Nieminen AL, Herman B, Lemasters JJ. 1993; Mitochondria as a source of reactive oxygen species during reductive stress in rat hepatocytes. Am J Physiol. 264(4 Pt 1):C961–C967. DOI: 10.1152/ajpcell.1993.264.4.C961. PMID: 8386454.
Article12. Lemasters JJ. 2005; Selective mitochondrial autophagy, or mitophagy, as a targeted defense against oxidative stress, mitochondrial dysfunction, and aging. Rejuvenation Res. 8:3–5. DOI: 10.1089/rej.2005.8.3. PMID: 15798367.
Article13. Xiao B, Deng X, Lim GGY, Zhou W, Saw WT, Zhou ZD, Lim KL, Tan EK. 2017; p62-Mediated mitochondrial clustering attenuates apoptosis induced by mitochondrial depolarization. Biochim Biophys Acta Mol Cell Res. 1864:1308–1317. DOI: 10.1016/j.bbamcr.2017.04.009. PMID: 28433685.
Article14. Matsuda N, Sato S, Shiba K, Okatsu K, Saisho K, Gautier CA, Sou YS, Saiki S, Kawajiri S, Sato F, Kimura M, Komatsu M, Hattori N, Tanaka K. 2010; PINK1 stabilized by mitochondrial depolarization recruits Parkin to damaged mitochondria and activates latent Parkin for mitophagy. J Cell Biol. 189:211–221. DOI: 10.1083/jcb.200910140. PMID: 20404107. PMCID: PMC2856912.
Article15. Narendra D, Tanaka A, Suen DF, Youle RJ. 2008; Parkin is recruited selectively to impaired mitochondria and promotes their autophagy. J Cell Biol. 183:795–803. DOI: 10.1083/jcb.200809125. PMID: 19029340. PMCID: PMC2592826.
Article16. Narendra DP, Jin SM, Tanaka A, Suen DF, Gautier CA, Shen J, Cookson MR, Youle RJ. 2010; PINK1 is selectively stabilized on impaired mitochondria to activate Parkin. PLoS Biol. 8:e1000298. DOI: 10.1371/journal.pbio.1000298. PMID: 20126261. PMCID: PMC2811155.
Article17. Halestrap AP, Connern CP, Griffiths EJ, Kerr PM. 1997; Cyclosporin A binding to mitochondrial cyclophilin inhibits the permeability transition pore and protects hearts from ischaemia/reperfusion injury. Mol Cell Biochem. 174:167–172. DOI: 10.1023/A:1006879618176. PMID: 9309682.
Article18. Akl MR, Ayoub NM, Abuasal BS, Kaddoumi A, Sylvester PW. 2013; Sesamin synergistically potentiates the anticancer effects of γ-tocotrienol in mammary cancer cell lines. Fitoterapia. 84:347–359. DOI: 10.1016/j.fitote.2012.12.013. PMID: 23266736.
Article19. Li L, Tan J, Miao Y, Lei P, Zhang Q. 2015; ROS and autophagy: interactions and molecular regulatory mechanisms. Cell Mol Neurobiol. 35:615–621. DOI: 10.1007/s10571-015-0166-x. PMID: 25722131.
Article20. Yang Y, Karakhanova S, Hartwig W, D'Haese JG, Philippov PP, Werner J, Bazhin AV. 2016; Mitochondria and mitochondrial ROS in cancer: novel targets for anticancer therapy. J Cell Physiol. 231:2570–2581. DOI: 10.1002/jcp.25349. PMID: 26895995.
Article21. Kang D, Hamasaki N. 2003; Mitochondrial oxidative stress and mitochondrial DNA. Clin Chem Lab Med. 41:1281–1288. DOI: 10.1515/CCLM.2003.195. PMID: 14580153.
Article22. Zhang B, Bian W, Pal A, He Y. 2015; Macrophage apoptosis induced by aqueous C60 aggregates changing the mitochondrial membrane potential. Environ Toxicol Pharmacol. 39:237–246. DOI: 10.1016/j.etap.2014.11.013. PMID: 25531262.
Article23. Vagner T, Mouravlev A, Young D. 2015; A novel bicistronic sensor vector for detecting caspase-3 activation. J Pharmacol Toxicol Methods. 72:11–18. DOI: 10.1016/j.vascn.2014.11.006. PMID: 25482476.
Article24. Farias-de-Oliveira DA, Villa-Verde DM, Nunes Panzenhagen PH, Silva dos Santos D, Berbert LR, Savino W, de Meis J. 2013; Caspase-8 and caspase-9 mediate thymocyte apoptosis in Trypanosoma cruzi acutely infected mice. J Leukoc Biol. 93:227–234. DOI: 10.1189/jlb.1211589. PMID: 23159925.
Article25. Martínez-Fábregas J, Díaz-Moreno I, González-Arzola K, Janocha S, Navarro JA, Hervás M, Bernhardt R, Velázquez-Campoy A, Díaz-Quintana A, De la Rosa MA. 2014; Structural and functional analysis of novel human cytochrome C targets in apoptosis. Mol Cell Proteomics. 13:1439–1456. DOI: 10.1074/mcp.M113.034322. PMID: 24643968. PMCID: PMC4047465.
Article26. Li J, Wu DD, Zhang JX, Wang J, Ma JJ, Hu X, Dong WG. 2018; Mitochondrial pathway mediated by reactive oxygen species involvement in α-hederin-induced apoptosis in hepatocellular carcinoma cells. World J Gastroenterol. 24:1901–1910. DOI: 10.3748/wjg.v24.i17.1901. PMID: 29740205. PMCID: PMC5937207.
Article27. Laulier C, Lopez BS. 2012; The secret life of Bcl-2: apoptosis-independent inhibition of DNA repair by Bcl-2 family members. Mutat Res. 751:247–257. DOI: 10.1016/j.mrrev.2012.05.002. PMID: 22677530.
Article28. Chen Q, Lesnefsky EJ. 2011; Blockade of electron transport during ischemia preserves bcl-2 and inhibits opening of the mitochondrial permeability transition pore. FEBS Lett. 585:921–926. DOI: 10.1016/j.febslet.2011.02.029. PMID: 21354418. PMCID: PMC3076511.
Article29. Pan LL, Wang AY, Huang YQ, Luo Y, Ling M. 2014; Mangiferin induces apoptosis by regulating Bcl-2 and Bax expression in the CNE2 nasopharyngeal carcinoma cell line. Asian Pac J Cancer Prev. 15:7065–7068. DOI: 10.7314/APJCP.2014.15.17.7065. PMID: 25227792.
Article30. Youle RJ, Narendra DP. 2011; Mechanisms of mitophagy. Nat Rev Mol Cell Biol. 12:9–14. DOI: 10.1038/nrm3028. PMID: 21179058. PMCID: PMC4780047.
Article31. Bertolin G, Ferrando-Miguel R, Jacoupy M, Traver S, Grenier K, Greene AW, Dauphin A, Waharte F, Bayot A, Salamero J, Lombès A, Bulteau AL, Fon EA, Brice A, Corti O. 2013; The TOMM machinery is a molecular switch in PINK1 and PARK2/PARKIN-dependent mitochondrial clearance. Autophagy. 9:1801–1817. DOI: 10.4161/auto.25884. PMID: 24149440.
Article32. Jin SM, Youle RJ. 2012; PINK1- and Parkin-mediated mitophagy at a glance. J Cell Sci. 125(Pt 4):795–799. DOI: 10.1242/jcs.093849. PMID: 22448035. PMCID: PMC3656616.
Article33. Altshuler-Keylin S, Shinoda K, Hasegawa Y, Ikeda K, Hong H, Kang Q, Yang Y, Perera RM, Debnath J, Kajimura S. 2016; Beige adipocyte maintenance is regulated by autophagy-induced mitochondrial clearance. Cell Metab. 24:402–419. DOI: 10.1016/j.cmet.2016.08.002. PMID: 27568548. PMCID: PMC5023491.
Article34. Kimura T, Takabatake Y, Takahashi A, Isaka Y. 2013; Chloroquine in cancer therapy: a double-edged sword of autophagy. Cancer Res. 73:3–7. DOI: 10.1158/0008-5472.CAN-12-2464. PMID: 23288916.
Article35. Kim JS, He L, Lemasters JJ. 2003; Mitochondrial permeability transition: a common pathway to necrosis and apoptosis. Biochem Biophys Res Commun. 304:463–470. DOI: 10.1016/S0006-291X(03)00618-1. PMID: 12729580.
Article36. Kim JS, Qian T, Lemasters JJ. 2003; Mitochondrial permeability transition in the switch from necrotic to apoptotic cell death in ischemic rat hepatocytes. Gastroenterology. 124:494–503. DOI: 10.1053/gast.2003.50059. PMID: 12557154.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Eugenol Induces a Reactive Oxygen Species-mediated Apoptosis in HL-60 Human Promyelocytic Leukemia Cells
- Dysfunctional Mitochondria Clearance in Situ: Mitophagy in Obesity and Diabetes-Associated Cardiometabolic Diseases
- Mitochondrial Reactive Oxygen Species Production Mediated by Romo1 Expression
- Anti-tumor activity and mitochondrial stability of disulfiram in HL-60 cells
- Cellular NAD⺠Level: A Key Determinant of Mitochondrial Quality and Health