Intest Res.
2020 Apr;18(2):184-191. 10.5217/ir.2019.00094.
Exclusive enteral nutrition for induction of remission in anti-tumor necrosis factor refractory adult Crohn’s disease: the Indian experience
- Affiliations
-
- 1Department of Gastroenterology, Dayanand Medical College, Ludhiana, India
- 2Department of Dietetics, Dayanand Medical College, Ludhiana, India
- 3Department of Internal Medicine, Dayanand Medical College, Ludhiana, India
- 4Department of Pharmacology, Dayanand Medical College, Ludhiana, India
- KMID: 2501383
- DOI: http://doi.org/10.5217/ir.2019.00094
Abstract
- Background/Aims
Exclusive enteral nutrition (EEN) is recommended for induction of remission in pediatric Crohn’s disease (CD). However, it is not currently recommended for inducing remission in adults. This report describes the use of 12-week EEN for induction of remission in anti-tumor necrosis factor (anti-TNF) refractory adult CD.
Methods
This is a retrospective analysis of adults with moderate to severe active (Crohn’s Disease Activity Index [CDAI] >220) anti-TNF refractory CD, who received EEN for 12 weeks between April 2018 and March 2019 at Dayanand Medical College and Hospital, Ludhiana, India. Primary outcomes included achievement of clinical remission and fistula healing at 12 weeks. Improvement in inflammatory markers and nutritional status were the secondary end points.
Results
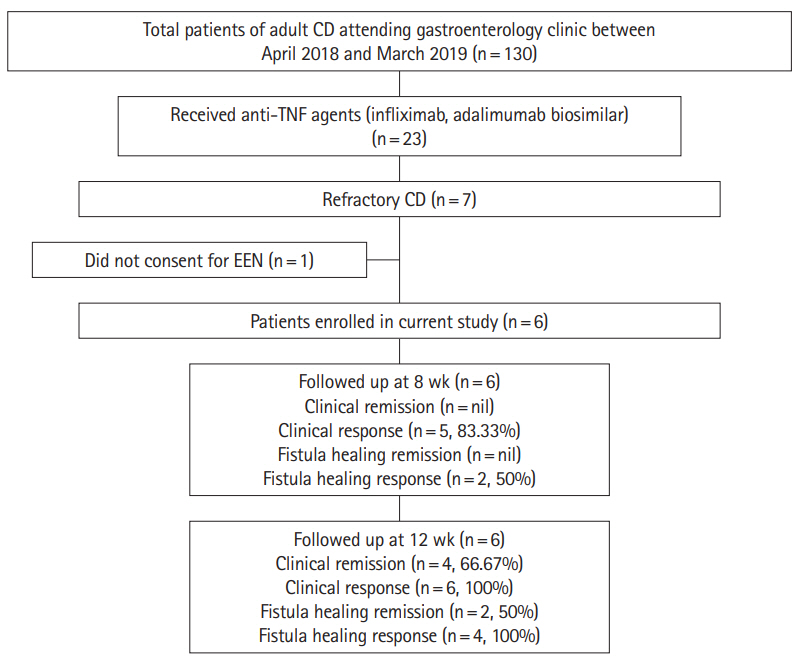
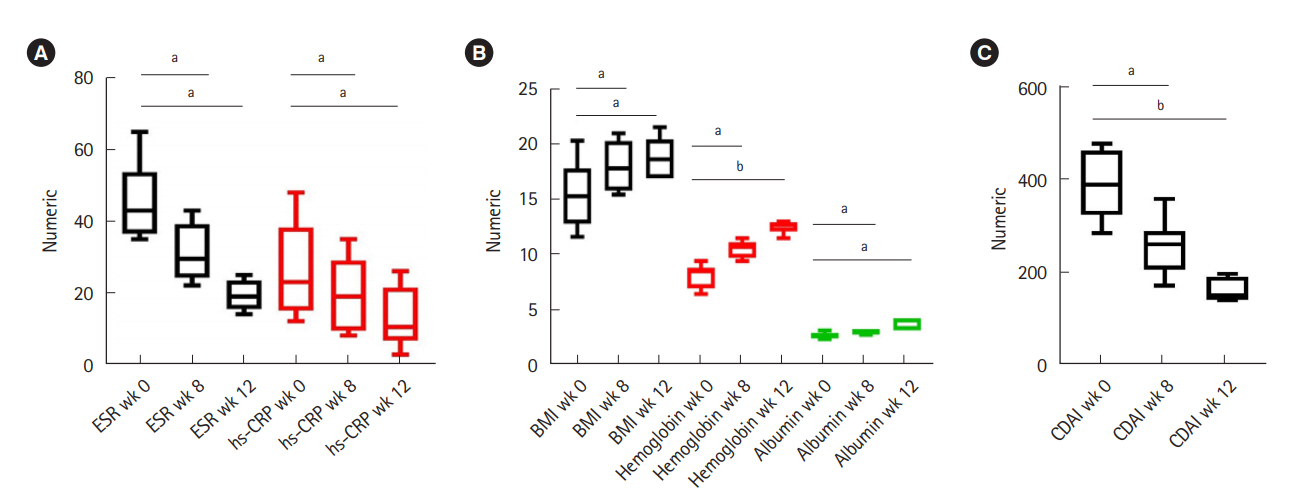
Out of 23 patients who received anti-TNF agents, 7 (30.4%) were refractory and were offered EEN as a salvage therapy. Six patients (66.7% females, mean age 25.6±6.5 years) consented. Four patients (66.6%) achieved clinical remission (CDAI <150). Mean CDAI of patients decreased significantly after 12 weeks of EEN (388.8±74.8 vs. 160.0±25.2, P<0.001). Perianal fistulas showed clinical response (drainage decreased by >50%), though none achieved remission. Entero-enteric fistulae showed complete healing. Mean body mass index improved from 15.6±3.1 to 18.9±1.9 kg/m2 at week 12 (P=0.003). Hemoglobin and serum albumin also improved from 8.2±1.1 g/dL and 2.8±0.3 g/dL at baseline to 12.6±0.6 g/dL and 3.6±0.5 g/dL post-EEN respectively (P<0.001 and P=0.006 respectively).
Conclusions
EEN appears to be an effective and well tolerated therapy for induction of remission in anti-TNF refractory adult CD. More data from prospective trials with larger number of patients is required.
Keyword
Figure
Cited by 1 articles
-
Enteral nutrition in the biologic era: learn from yesterday, live for today, hope for tomorrow
Tadakazu Hisamatsu
Intest Res. 2020;18(2):139-140. doi: 10.5217/ir.2019.09192.
Reference
-
1. Ha F, Khalil H. Crohn’s disease: a clinical update. Therap Adv Gastroenterol. 2015; 8:352–359.
Article2. Hanauer SB, Feagan BG, Lichtenstein GR, et al. Maintenance infliximab for Crohn’s disease: the ACCENT I randomised trial. Lancet. 2002; 359:1541–1549.
Article3. Colombel JF, Sandborn WJ, Rutgeerts P, et al. Adalimumab for maintenance of clinical response and remission in patients with Crohn’s disease: the CHARM trial. Gastroenterology. 2007; 132:52–65.
Article4. Schreiber S, Khaliq-Kareemi M, Lawrance IC, et al. Maintenance therapy with certolizumab pegol for Crohn’s disease. N Engl J Med. 2007; 357:239–250.
Article5. Sandborn WJ, Rutgeerts P, Enns R, et al. Adalimumab induction therapy for Crohn disease previously treated with infliximab: a randomized trial. Ann Intern Med. 2007; 146:829–838.
Article6. Sandborn WJ, Gasink C, Gao LL, et al. Ustekinumab induction and maintenance therapy in refractory Crohn’s disease. N Engl J Med. 2012; 367:1519–1528.
Article7. Fukuda Y, Matsui T, Suzuki Y, et al. Adsorptive granulocyte and monocyte apheresis for refractory Crohn’s disease: an open multicenter prospective study. J Gastroenterol. 2004; 39:1158–1164.
Article8. Domènech E, Hinojosa J, Esteve-Comas M, et al. Granulocyteaphaeresis in steroid-dependent inflammatory bowel disease: a prospective, open, pilot study. Aliment Pharmacol Ther. 2004; 20:1347–1352.
Article9. Matsui T, Nishimura T, Matake H, Ohta T, Sakurai T, Yao T. Granulocytapheresis for Crohn’s disease: a report on seven refractory patients. Am J Gastroenterol. 2003; 98:511–512.
Article10. Weisshof R, Chermesh I. Micronutrient deficiencies in inflammatory bowel disease. Curr Opin Clin Nutr Metab Care. 2015; 18:576–581.
Article11. Nguyen GC, Munsell M, Harris ML. Nationwide prevalence and prognostic significance of clinically diagnosable proteincalorie malnutrition in hospitalized inflammatory bowel disease patients. Inflamm Bowel Dis. 2008; 14:1105–1111.
Article12. Goh J, O’Morain CA. Review article: nutrition and adult inflammatory bowel disease. Aliment Pharmacol Ther. 2003; 17:307320.13. Lochs H, Dejong C, Hammarqvist F, et al. ESPEN guidelines on enteral nutrition: gastroenterology. Clin Nutr. 2006; 25:260274.
Article14. Ruemmele FM, Veres G, Kolho KL, et al. Consensus guidelines of ECCO/ESPGHAN on the medical management of pediatric Crohn’s disease. J Crohns Colitis. 2014; 8:1179–1207.15. Day AS, Lopez RN. Exclusive enteral nutrition in children with Crohn’s disease. World J Gastroenterol. 2015; 21:6809–6816.
Article16. Takagi S, Utsunomiya K, Kuriyama S, et al. Effectiveness of an ‘half elemental diet’ as maintenance therapy for Crohn’s disease: a randomized-controlled trial. Aliment Pharmacol Ther. 2006; 24:1333–1340.
Article17. Esaki M, Matsumoto T, Hizawa K, et al. Preventive effect of nutritional therapy against postoperative recurrence of Crohn disease, with reference to findings determined by intra-operative enteroscopy. Scand J Gastroenterol. 2005; 40:1431–1437.
Article18. Teahon K, Bjarnason I, Pearson M, Levi AJ. Ten years’ experience with an elemental diet in the management of Crohn’s disease. Gut. 1990; 31:1133–1137.
Article19. Rigaud D, Cosnes J, Le Quintrec Y, René E, Gendre JP, Mignon M. Controlled trial comparing two types of enteral nutrition in treatment of active Crohn’s disease: elemental versus polymeric diet. Gut. 1991; 32:1492–1497.
Article20. Li Y, Zhu W, Gong J, et al. Influence of exclusive enteral nutrition therapy on visceral fat in patients with Crohn’s disease. Inflamm Bowel Dis. 2014; 20:1568–1574.
Article21. Wall CL, Gearry RB, Day AS. Treatment of active Crohn’s disease with exclusive and partial enteral nutrition: a pilot study in adults. Inflamm Intest Dis. 2018; 2:219–227.
Article22. Hu D, Ren J, Wang G, et al. Exclusive enteral nutritional therapy can relieve inflammatory bowel stricture in Crohn’s disease. J Clin Gastroenterol. 2014; 48:790–795.
Article23. Yang Q, Gao X, Chen H, et al. Efficacy of exclusive enteral nutrition in complicated Crohn’s disease. Scand J Gastroenterol. 2017; 52:995–1001.
Article24. Yamamoto T, Nakahigashi M, Umegae S, Matsumoto K. Enteral nutrition for the maintenance of remission in Crohn’s disease: a systematic review. Eur J Gastroenterol Hepatol. 2010; 22:1–8.
Article25. Lennard-Jones JE. Classification of inflammatory bowel disease. Scand J Gastroenterol Suppl. 1989; 170:2–6.
Article26. Satsangi J, Silverberg MS, Vermeire S, Colombel JF. The Montreal classification of inflammatory bowel disease: controversies, consensus, and implications. Gut. 2006; 55:749–753.
Article27. Gomollón F, Dignass A, Annese V, et al. 3rd European evidencebased consensus on the diagnosis and management of Crohn’s disease 2016: Part 1: diagnosis and medical management. J Crohns Colitis. 2017; 11:3–25.
Article28. Asteria CR, Ficari F, Bagnoli S, Milla M, Tonelli F. Treatment of perianal fistulas in Crohn’s disease by local injection of antibody to TNF-alpha accounts for a favourable clinical response in selected cases: a pilot study. Scand J Gastroenterol. 2006; 41:1064–1072.
Article29. Sidler MA, Lemberg DA, Day AS. Combination of nutritional therapy and medical therapy for the management of enterovesical fistula in paediatric Crohn’s disease: a case report. eSPEN. 2008; 3:e89. –e91.
Article30. Levine A, Wine E. Effects of enteral nutrition on Crohn’s disease: clues to the impact of diet on disease pathogenesis. Inflamm Bowel Dis. 2013; 19:1322–1329.31. Lionetti P, Callegari ML, Ferrari S, et al. Enteral nutrition and microflora in pediatric Crohn’s disease. JPEN J Parenter Enteral Nutr. 2005; 29(4 Suppl):S173–S175.
Article32. de Jong NS, Leach ST, Day AS. Polymeric formula has direct anti-inflammatory effects on enterocytes in an in vitro model of intestinal inflammation. Dig Dis Sci. 2007; 52:2029–2036.
Article33. Nahidi L, Corley SM, Wilkins MR, et al. The major pathway by which polymeric formula reduces inflammation in intestinal epithelial cells: a microarray-based analysis. Genes Nutr. 2015; 10:479.
Article34. Alhagamhmad MH, Day AS, Lemberg DA, Leach ST. Exploring and enhancing the anti-inflammatory properties of polymeric formula. JPEN J Parenter Enteral Nutr. 2017; 41:436–445.
Article35. de Bie C, Kindermann A, Escher J. Use of exclusive enteral nutrition in paediatric Crohn’s disease in The Netherlands. J Crohns Colitis. 2013; 7:263–270.
Article36. Afzal NA, Davies S, Paintin M, et al. Colonic Crohn’s disease in children does not respond well to treatment with enteral nutrition if the ileum is not involved. Dig Dis Sci. 2005; 50:14711475.
Article37. Nguyen DL, Palmer LB, Nguyen ET, McClave SA, Martindale RG, Bechtold ML. Specialized enteral nutrition therapy in Crohn’s disease patients on maintenance infliximab therapy: a metaanalysis. Therap Adv Gastroenterol. 2015; 8:168–175.
Article38. Hirai F, Ishihara H, Yada S, et al. Effectiveness of concomitant enteral nutrition therapy and infliximab for maintenance treatment of Crohn’s disease in adults. Dig Dis Sci. 2013; 58:13291334.
Article39. Kamata N, Oshitani N, Watanabe K, et al. Efficacy of concomitant elemental diet therapy in scheduled infliximab therapy in patients with Crohn’s disease to prevent loss of response. Dig Dis Sci. 2015; 60:1382–1388.
Article40. Hisamatsu T, Kunisaki R, Nakamura S, et al. Effect of elemental diet combined with infliximab dose escalation in patients with Crohn’s disease with loss of response to infliximab: CERISIER trial. Intest Res. 2018; 16:494–498.
Article41. Narula N, Dhillon A, Zhang D, Sherlock ME, Tondeur M, Zachos M. Enteral nutritional therapy for induction of remission in Crohn’s disease. Cochrane Database Syst Rev. 2018; 4–CD000542.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- An overview of the role of exclusive enteral nutrition for complicated Crohn's disease
- A Case of Crohn's Disease Showing Favorable Response to Induction and Maintenance Therapy with Methotrexate after Failure of Anti-tumor Necrosis Factor Therapy
- Therapeutic Efficacy of Exclusive Enteral Nutrition with Specific Polymeric Diet in Pediatric Crohn's Disease
- Therapeutic Efficacy of Oral Enteral Nutrition in Pediatric Crohn's Disease: A Single Center Non-Comparative Retrospective Study
- Infliximab: The Benefit for Refractory Crohn Disease andTop-down Induction Therapy in Severe Crohn Disease