Cancer Res Treat.
2020 Jan;52(1):284-291. 10.4143/crt.2019.200.
Osimertinib in Patients with T790M-Positive Advanced Non-small Cell Lung Cancer: Korean Subgroup Analysis from Phase II Studies
- Affiliations
-
- 1Division of Hematology-Oncology, Department of Medicine, Samsung Medical Center, Sungkyunkwan University School of Medicine, Seoul, Korea
- 2Center for Lung Cancer, National Cancer Center, Goyang, Korea
- 3Department of Internal Medicine, Seoul National University College of Medicine, Seoul, Korea
- 4Division of Medical Oncology, Department of Internal Medicine, Yonsei Cancer Center, Yonsei University College of Medicine, Seoul, Korea
- 5Division of Medical Oncology, Department of Internal Medicine, Seoul St. Mary’s Hospital, College of Medicine, The Catholic University of Korea, Seoul, Korea
- 6Department of Oncology, Asan Medical Center, University of Ulsan College of Medicine, Seoul, Korea
- 7National Taiwan University and National Taiwan University Hospital, Taipei, Taiwan
- 8Kindai University Faculty of Medicine, Osaka-Sayama, Japan
- 9Division of Hematology and Medical Oncology, Department of Internal Medicine, Seoul National University Bundang Hospital, Seongnam, Korea
- KMID: 2501220
- DOI: http://doi.org/10.4143/crt.2019.200
Abstract
- Purpose
Osimertinib is a third-generation, irreversible, oral epidermal growth factor receptor (EGFR) tyrosine kinase inhibitor (TKI) that potently and selectively inhibits both EGFR sensitizing mutation and EGFR T790M and has demonstrated efficacy in non-small cell lung cancer (NSCLC) central nervous system (CNS) metastases. We present results of a subgroup analysis of Korean patients from the pooled data of two global phase II trials: AURA extension (NCT01802632) and AURA2 (NCT02094261).
Materials and Methods
Enrolled patients had EGFR T790M-positive NSCLC and disease progression during or after EGFR-TKI therapy. Patients received osimertinib 80 mg orally once daily until disease progression. The primary endpoint was objective response rate (ORR).
Results
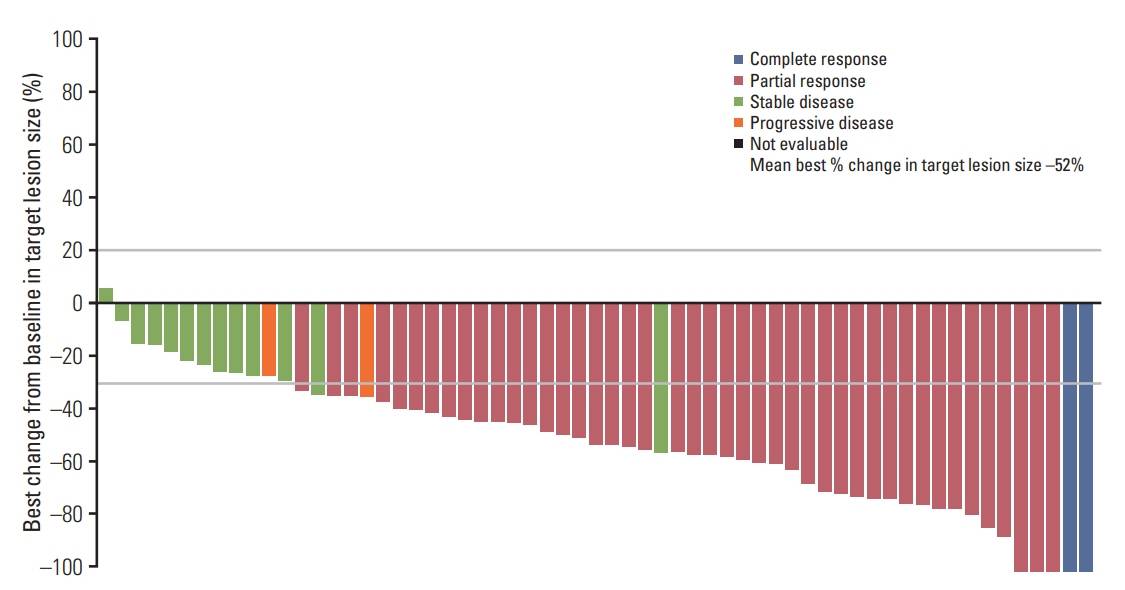
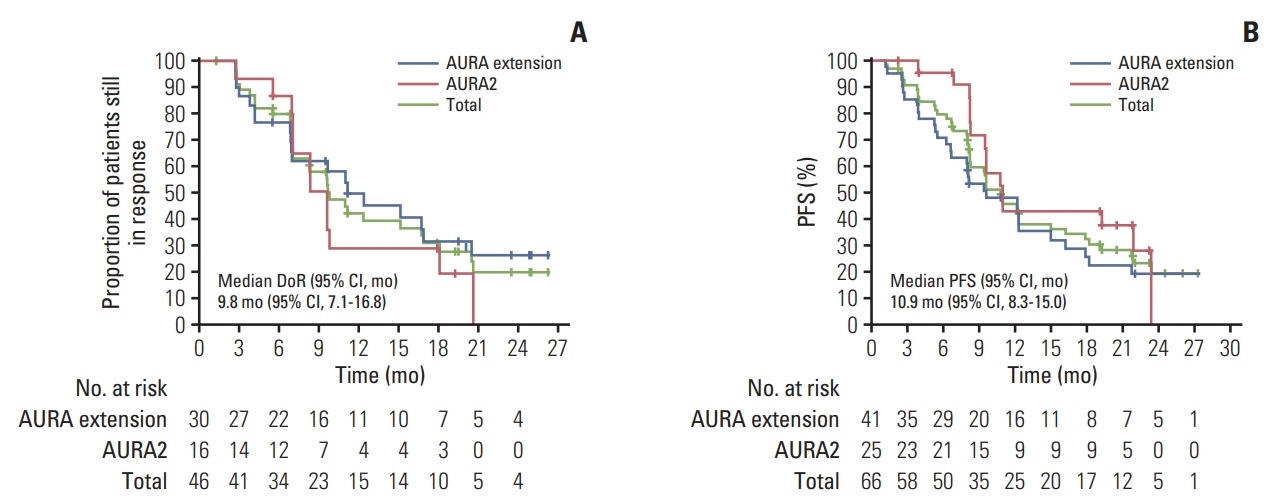
In total, 66 Korean patients received osimertinib treatment with a median treatment duration of 19 months. In the evaluable-for-response population (n=62), ORR was 74% (95% confidence interval [CI], 61.5 to 84.5) and median duration of response was 9.8 months (95% CI, 7.1 to 16.8). In the full analysis set (n=66), median progression-free survival was 10.9 months (95% CI, 8.3 to 15.0; data cutoff November 1, 2016), and median overall survival was 29.2 months (95% CI, 24.8 to 35.7; data cutoff May 1, 2018). Eight patients with CNS metastases were evaluable for response, none of whom showed CNS progression. The most common adverse events were rash (53%), cough (33%), paronychia, diarrhea, and decreased appetite (each 32%).
Conclusion
Efficacy and safety profiles of osimertinib in this subgroup are consistent with the global phase II pooled population, which supports osimertinib as a recommended treatment for Korean patients with T790M positive NSCLC.
Keyword
Figure
Reference
-
References
1. Tan DS, Yom SS, Tsao MS, Pass HI, Kelly K, Peled N, et al. The International Association for the Study of Lung Cancer Consensus Statement on optimizing management of EGFR mutation-positive non-small cell lung cancer: status in 2016. J Thorac Oncol. 2016; 11:946–63.
Article2. Hanna N, Johnson D, Temin S, Baker S Jr, Brahmer J, Ellis PM, et al. Systemic therapy for stage IV non-small-cell lung cancer: American Society of Clinical Oncology Clinical Practice Guideline update. J Clin Oncol. 2017; 35:3484–515.
Article3. Planchard D, Popat S, Kerr K, Novello S, Smit EF, Faivre-Finn C, et al. Metastatic non-small cell lung cancer: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol. 2018; 29(Suppl 4):iv192–237.
Article4. Rosell R, Carcereny E, Gervais R, Vergnenegre A, Massuti B, Felip E, et al. Erlotinib versus standard chemotherapy as firstline treatment for European patients with advanced EGFR mutation-positive non-small-cell lung cancer (EURTAC): a multicentre, open-label, randomised phase 3 trial. Lancet Oncol. 2012; 13:239–46.5. Mok TS, Wu YL, Thongprasert S, Yang CH, Chu DT, Saijo N, et al. Gefitinib or carboplatin-paclitaxel in pulmonary adenocarcinoma. N Engl J Med. 2009; 361:947–57.
Article6. Sequist LV, Yang JC, Yamamoto N, O'Byrne K, Hirsh V, Mok T, et al. Phase III study of afatinib or cisplatin plus pemetrexed in patients with metastatic lung adenocarcinoma with EGFR mutations. J Clin Oncol. 2013; 31:3327–34.
Article7. Wang ZF, Ren SX, Li W, Gao GH. Frequency of the acquired resistant mutation T790 M in non-small cell lung cancer patients with active exon 19Del and exon 21 L858R: a systematic review and meta-analysis. BMC Cancer. 2018; 18:148.
Article8. Cross DA, Ashton SE, Ghiorghiu S, Eberlein C, Nebhan CA, Spitzler PJ, et al. AZD9291, an irreversible EGFR TKI, overcomes T790M-mediated resistance to EGFR inhibitors in lung cancer. Cancer Discov. 2014; 4:1046–61.
Article9. Soria JC, Ohe Y, Vansteenkiste J, Reungwetwattana T, Chewaskulyong B, Lee KH, et al. Osimertinib in untreated EGFRmutated advanced non-small-cell lung cancer. N Engl J Med. 2018; 378:113–25.10. Wu YL, Ahn MJ, Garassino MC, Han JY, Katakami N, Kim HR, et al. CNS efficacy of osimertinib in patients with T790Mpositive advanced non-small-cell lung cancer: data from a randomized phase III trial (AURA3). J Clin Oncol. 2018; 36:2702–9.
Article11. Reungwetwattana T, Nakagawa K, Cho BC, Cobo M, Cho EK, Bertolini A, et al. CNS response to osimertinib versus standard epidermal growth factor receptor tyrosine kinase inhibitors in patients with untreated EGFR-mutated advanced non-small-cell lung cancer. J Clin Oncol. 2018; 36:3290–7.
Article12. Mok TS, Wu YL, Ahn MJ, Garassino MC, Kim HR, Ramalingam SS, et al. Osimertinib or platinum-pemetrexed in EGFR T790M-positive lung cancer. N Engl J Med. 2017; 376:629–40.
Article13. Yang JC, Ahn MJ, Kim DW, Ramalingam SS, Sequist LV, Su WC, et al. Osimertinib in pretreated T790M-positive advanced non-small-cell lung cancer: AURA study phase II extension component. J Clin Oncol. 2017; 35:1288–96.
Article14. Goss G, Tsai CM, Shepherd FA, Bazhenova L, Lee JS, Chang GC, et al. Osimertinib for pretreated EGFR Thr790Met-positive advanced non-small-cell lung cancer (AURA2): a multicentre, open-label, single-arm, phase 2 study. Lancet Oncol. 2016; 17:1643–52.
Article15. Ahn MJ, Tsai CM, Shepherd FA, Bazhenova L, Sequist LV, Hida T, et al. Osimertinib in patients with T790M mutationpositive, advanced non-small cell lung cancer: long-term follow-up from a pooled analysis of 2 phase 2 studies. Cancer. 2019; 125:892–901.
Article16. Mok T, Ahn MJ, Han JY, Kang JH, Katakami N, Kim H, et al. CNS response to osimertinib in patients (pts) with T790M-positive advanced NSCLC: data from a randomized phase III trial (AURA3). J Clin Oncol. 2017; 35(15 Suppl):9005.
Article17. Novello S, Barlesi F, Califano R, Cufer T, Ekman S, Levra MG, et al. Metastatic non-small-cell lung cancer: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol. 2016; 27(suppl 5):v1–27.
Article18. Douillard JY, Ostoros G, Cobo M, Ciuleanu T, McCormack R, Webster A, et al. First-line gefitinib in Caucasian EGFR mutation-positive NSCLC patients: a phase-IV, open-label, singlearm study. Br J Cancer. 2014; 110:55–62.
Article19. Shigematsu H, Lin L, Takahashi T, Nomura M, Suzuki M, Wistuba II, et al. Clinical and biological features associated with epidermal growth factor receptor gene mutations in lung cancers. J Natl Cancer Inst. 2005; 97:339–46.
Article20. Lee SH, Kim WS, Choi YD, Seo JW, Han JH, Kim MJ, et al. Analysis of mutations in epidermal growth factor receptor gene in Korean patients with non-small cell lung cancer: summary of a nationwide survey. J Pathol Transl Med. 2015; 49:481–8.
Article21. Goss G, Tsai CM, Shepherd FA, Ahn MJ, Bazhenova L, Crino L, et al. CNS response to osimertinib in patients with T790Mpositive advanced NSCLC: pooled data from two phase II trials. Ann Oncol. 2018; 29:687–93.
Article22. Rangachari D, Yamaguchi N, VanderLaan PA, Folch E, Mahadevan A, Floyd SR, et al. Brain metastases in patients with EGFR-mutated or ALK-rearranged non-small-cell lung cancers. Lung Cancer. 2015; 88:108–11.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Assessment of Anti-tumor Efficacy of Osimertinib in Non-Small Cell Lung Cancer Patients by Liquid Biopsy Using Bronchoalveolar Lavage Fluid, Plasma, or Pleural Effusion
- A Phase II Trial of Osimertinib in the Second-Line Treatment of Non-small Cell Lung Cancer with the EGFR T790M Mutation, Detected from Circulating Tumor DNA: LiquidLung-O-Cohort 2
- Osimertinib Combined with Systemic Chemotherapy for EGFR Mutant, T790M-Negative, Non–Small Cell Lung Cancer Patients Who Develop Leptomeningeal Metastases with Extracranial Progression to Prior EGFR TKI
- Lazertinib: on the Way to Its Throne
- Rare Mechanism of Acquired Resistance to Osimertinib in Korean Patients with EGFR-mutated Non-small Cell Lung Cancer