Int J Stem Cells.
2020 Mar;13(1):163-175. 10.15283/ijsc18066.
Effect of Stem Cells, Ascorbic Acid and SERCA1a Gene Transfected Stem Cells in Experimentally Induced Type I Diabetic Myopathy
- Affiliations
-
- 1Department of Medical Histology and Cell Biology, Faculty of Medicine, Cairo University, Giza, Egypt
- 2Faculty of Oral and Dental Medicine, Future University in Egypt (FUE), New Cairo City, Egypt
- 3Faculty of Medicine, Cairo University, Giza, Egypt
- 4Faculty of Pharmacy and Biotechnology, German University in Cairo (GUC), New Cairo City, Egypt
- 5Faculty of Pharmacy, Near East University, North Cyprus, Cyprus
- KMID: 2500575
- DOI: http://doi.org/10.15283/ijsc18066
Abstract
- Background and Objectives
Sarco/endoplasmic reticulum Ca2+-ATPase (SERCA) inhibition was proved in streptozotocin (STZ)-diabetic rats. The present study aimed at investigating and comparing the therapeutic effect of bone marrow mesenchymal stem cells (BMMSCs), BMMSCs combined with ascorbic acid (AA) and SERCA1a gene transfected BMMSCs in induced type I diabetic myopathy of male albino rat.
Methods and Results
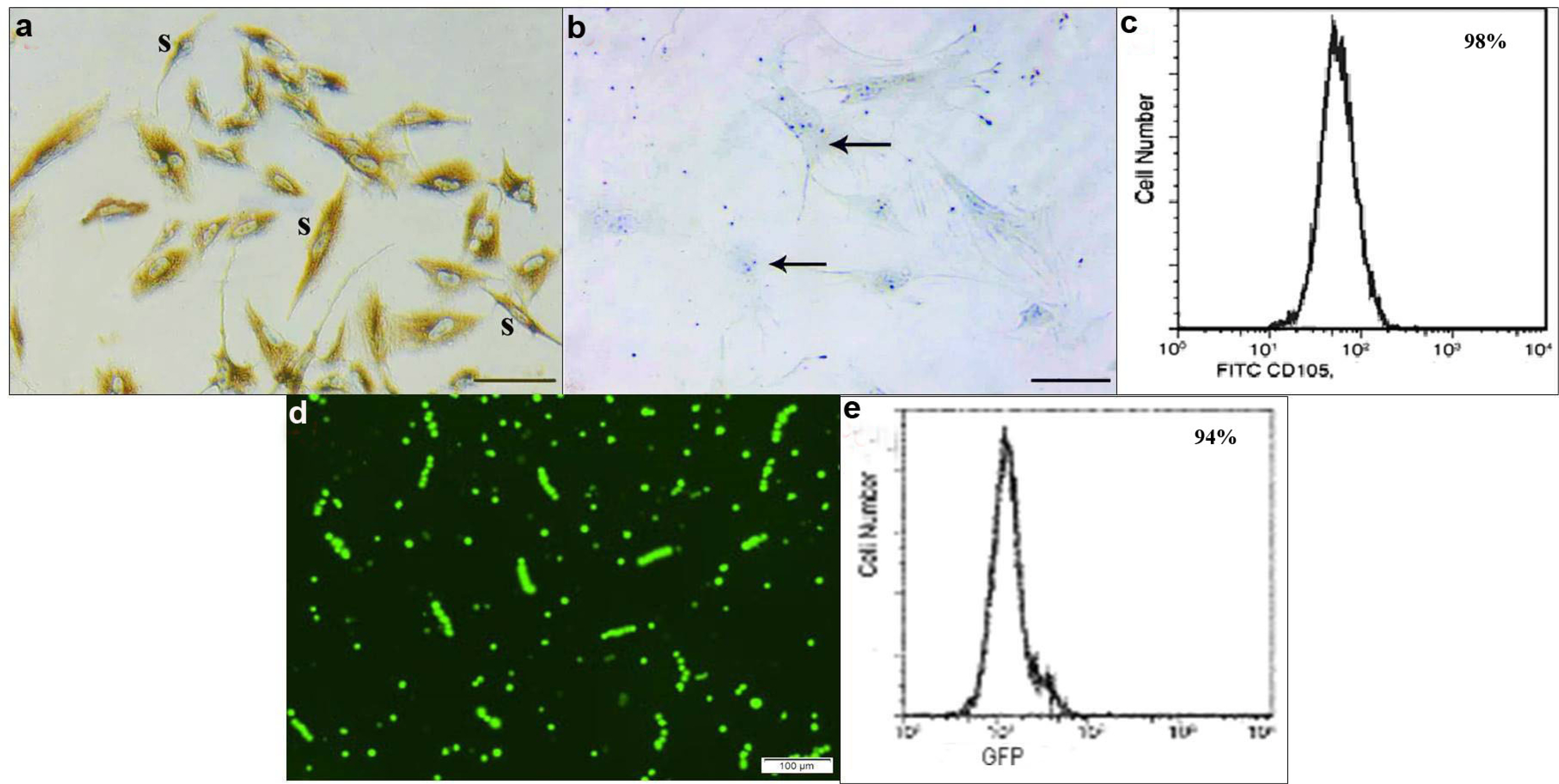
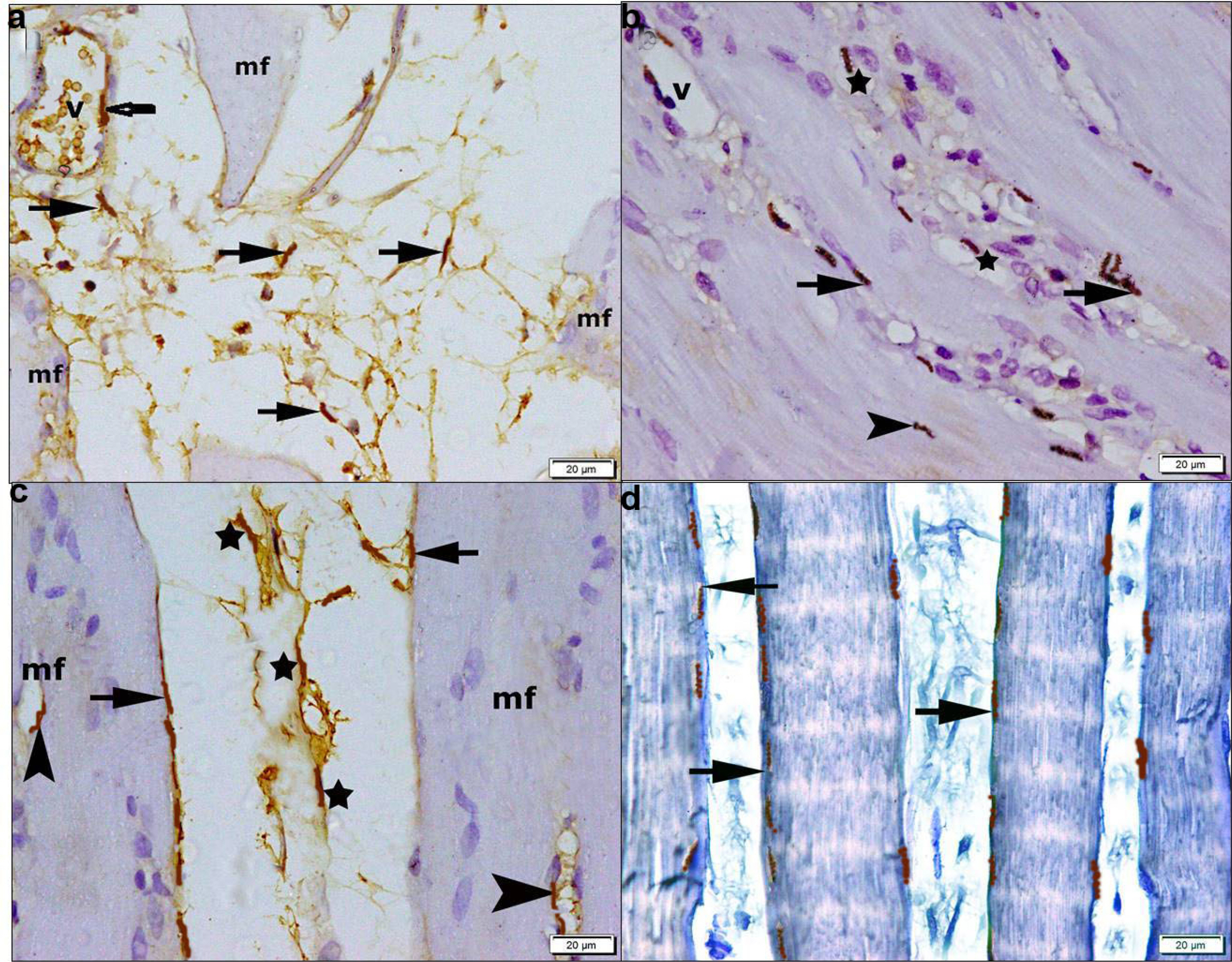
54 rats were divided into donor group of 6 rats for isolation, propagation and characterization of BMMSCs and SERCA1a transfected BMMSCs, groups I∼V 48 rats. Group I of 8 control rats, group II (Diabetic) of 10 rats given STZ 50 mg/kg intraperitoneal, group III (BMMSCs) of 10 rats given STZ and BMMSCs intravenous (IV), group IV (BMMSCs and AA) of 10 rats given STZ, BMMSCs IV and AA 500 mg/kg and group V (SERCA 1a transfected BMMSCs) of 10 rats given STZ and SERCA1a transfected BMMSCs IV. The rats were sacrificed after 8 weeks. Gastrocnemius specimens were subjected to biochemical, histological, morphometric and statistical studies. Diabetic rats revealed inflammatory and degenerative muscle changes, a significant increase in blood glucose level, mean DNA fragmentation and mean MDA values and a significant decrease in mean GSH and catalase values, area of pale nuclei, area% of CD105 and CD34 +ve cells, SERCA1a protein and gene values. The morphological changes regressed by therapy. In group III significant decrease in DNA fragmentation and MDA, significant increase in GSH and catalase, significant increase in the mean area of pale nuclei, area % of CD105 and CD34 +ve cells versus diabetic group. In group IV, same findings as group III versus diabetic and BMMSCs groups. In group V, same findings as group IV versus diabetic and treated groups. Western blot and PCR proved a mean value of SERCA1a protein and gene comparable to the control group. Mean calcium concentration values revealed a significant increase in the diabetic group, in BMMSCs and AA group versus control and SERCA1a group.
Conclusions
SERCA1a transfected BMMSCs proved a definite therapeutic effect, more remarkable than BMMSCs combined with AA. This effect was evidenced histologically and confirmed by significant changes in the biochemical tests indicating oxidative stress, muscle calcium concentration, morphometric parameters and PCR values of SERCA1a.
Keyword
Figure
Reference
-
References
1. Chiu CY, Yang RS, Sheu ML, Chan DC, Yang TH, Tsai KS, Chiang CK, Liu SH. 2016; Advanced glycation end-products induce skeletal muscle atrophy and dysfunction in diabetic mice via a RAGE-mediated, AMPK-down-regulated, Akt pathway. J Pathol. 238:470–482. DOI: 10.1002/path.4674. PMID: 26586640.
Article2. Harmer AR, Ruell PA, Hunter SK, McKenna MJ, Thom JM, Chisholm DJ, Flack JR. 2014; Effects of type 1 diabetes, sprint training and sex on skeletal muscle sarcoplasmic reticulum Ca2+ uptake and Ca2+-ATPase activity. J Physiol. 592:523–535. DOI: 10.1113/jphysiol.2013.261172. PMID: 24297852. PMCID: PMC3930437.
Article3. Gamu D, Trinh A, Bombardier E, Tupling AR. 2015; Persistence of diet-induced obesity despite access to voluntary activity in mice lacking sarcolipin. Physiol Rep. 3:e12549. DOI: 10.14814/phy2.12549. PMID: 26400985. PMCID: PMC4600390.
Article4. Thompson MD, Mei Y, Weisbrod RM, Silver M, Shukla PC, Bolotina VM, Cohen RA, Tong X. 2014; Glutathione adducts on sarcoplasmic/endoplasmic reticulum Ca2+ ATPase Cys-674 regulate endothelial cell calcium stores and angiogenic function as well as promote ischemic blood flow recovery. J Biol Chem. 289:19907–19916. DOI: 10.1074/jbc.M114.554451. PMID: 24920669. PMCID: PMC4106311.
Article5. Masola B, Oguntibeju OO, Oyenihi AB. 2018; Centella asiatica ameliorates diabetes-induced stress in rat tissues via influences on antioxidants and inflammatory cytokines. Biomed Pharmacother. 101:447–457. DOI: 10.1016/j.biopha.2018.02.115. PMID: 29501767.
Article6. Oyarce K, Silva-Alvarez C, Ferrada L, Martínez F, Salazar K, Nualart F. 2018; SVCT2 is expressed by cerebellar precursor cells, which differentiate into neurons in response to ascorbic acid. Mol Neurobiol. 55:1136–1149. DOI: 10.1007/s12035-016-0366-5. PMID: 28097475.
Article7. de la Garza-Rodea AS, Boersma H, Dambrot C, de Vries AA, van Bekkum DW, Knaän-Shanzer S. 2015; Barriers in contribution of human mesenchymal stem cells to murine muscle regeneration. World J Exp Med. 5:140–153. DOI: 10.5493/wjem.v5.i2.140. PMID: 25992329. PMCID: PMC4436938.8. Mars T, Strazisar M, Mis K, Kotnik N, Pegan K, Lojk J, Grubic Z, Pavlin M. 2015; Electrotransfection and lipofection show comparable efficiency for in vitro gene delivery of primary human myoblasts. J Membr Biol. 248:273–283. DOI: 10.1007/s00232-014-9766-5. PMID: 25534347.
Article9. Hidaka R, Machida M, Fujimaki S, Terashima K, Asashima M, Kuwabara T. 2013; Monitoring neurodegeneration in diabetes using adult neural stem cells derived from the olfactory bulb. Stem Cell Res Ther. 4:51. DOI: 10.1186/scrt201. PMID: 23673084. PMCID: PMC3707061.
Article10. Bhansali S, Kumar V, Saikia UN, Medhi B, Jha V, Bhansali A, Dutta P. 2015; Effect of mesenchymal stem cells transplantation on glycaemic profile & their localization in streptozotocin induced diabetic Wistar rats. Indian J Med Res. 142:63–71. DOI: 10.4103/0971-5916.162116. PMID: 26261168. PMCID: PMC4557252.
Article11. Sassoli C, Nosi D, Tani A, Chellini F, Mazzanti B, Quercioli F, Zecchi-Orlandini S, Formigli L. 2014; Defining the role of mesenchymal stromal cells on the regulation of matrix metalloproteinases in skeletal muscle cells. Exp Cell Res. 323:297–313. DOI: 10.1016/j.yexcr.2014.03.003. PMID: 24631289.
Article12. Aboul-Fotouh GI, Zickri MB, Metwally HG, Ibrahim IR, Kamar SS, Sakr W. 2015; Therapeutic effect of adipose derived stem cells versus atorvastatin on amiodarone induced lung injury in male rat. Int J Stem Cells. 8:170–180. DOI: 10.15283/ijsc.2015.8.2.170. PMID: 26634065. PMCID: PMC4651281.
Article13. Picklo MJ, Thyfault JP. 2015; Vitamin E and vitamin C do not reduce insulin sensitivity but inhibit mitochondrial protein expression in exercising obese rats. Appl Physiol Nutr Metab. 40:343–352. DOI: 10.1139/apnm-2014-0302. PMID: 25761734. PMCID: PMC4713234.
Article14. Espina M, Jülke H, Brehm W, Ribitsch I, Winter K, Delling U. 2016; Evaluation of transport conditions for autologous bone marrow-derived mesenchymal stromal cells for therapeutic application in horses. PeerJ. 4:e1773. DOI: 10.7717/peerj.1773. PMID: 27019778. PMCID: PMC4806605.
Article15. Li H, Fu X, Ouyang Y, Cai C, Wang J, Sun T. 2006; Adult bone-marrow-derived mesenchymal stem cells contribute to wound healing of skin appendages. Cell Tissue Res. 326:725–736. DOI: 10.1007/s00441-006-0270-9. PMID: 16906419.
Article16. Ude CC, Shamsul BS, Ng MH, Chen HC, Norhamdan MY, Aminuddin BS, Ruszymah BH. 2012; Bone marrow and adipose stem cells can be tracked with PKH26 until post staining passage 6 in in vitro and in vivo. Tissue Cell. 44:156–163. DOI: 10.1016/j.tice.2012.02.001. PMID: 22402173.
Article17. Tupling AR, Bombardier E, Gupta SC, Hussain D, Vigna C, Bloemberg D, Quadrilatero J, Trivieri MG, Babu GJ, Backx PH, Periasamy M, MacLennan DH, Gramolini AO. 2011; Enhanced Ca2+ transport and muscle relaxation in skeletal muscle from sarcolipin-null mice. Am J Physiol Cell Physiol. 301:C841–C849. DOI: 10.1152/ajpcell.00409.2010. PMID: 21697544. PMCID: PMC3654932.
Article18. Gruber SJ, Cornea RL, Li J, Peterson KC, Schaaf TM, Gillispie GD, Dahl R, Zsebo KM, Robia SL, Thomas DD. 2014; Discovery of enzyme modulators via high-throughput time-resolved FRET in living cells. J Biomol Screen. 19:215–222. DOI: 10.1177/1087057113510740. PMID: 24436077. PMCID: PMC4013825.
Article19. Kraitchman DL, Heldman AW, Atalar E, Amado LC, Martin BJ, Pittenger MF, Hare JM, Bulte JW. 2003; In vivo magnetic resonance imaging of mesenchymal stem cells in myocardial infarction. Circulation. 107:2290–2293. DOI: 10.1161/01.CIR.0000070931.62772.4E. PMID: 12732608.
Article20. Golshan Iranpour F, Kheiri S. 2016; Coadministration of calcium chloride with lead acetate can improve motility of cauda epididymal spermatozoa in Swiss white mice. Int J Reprod Biomed (Yazd). 14:141–144. DOI: 10.29252/ijrm.14.2.141. PMID: 27200429. PMCID: PMC4869154.
Article21. Nicoletti I, Migliorati G, Pagliacci MC, Grignani F, Riccardi C. 1991; A rapid and simple method for measuring thymocyte apoptosis by propidium iodide staining and flow cytometry. J Immunol Methods. 139:271–279. DOI: 10.1016/0022-1759(91)90198-O. PMID: 1710634.
Article22. Brancaccio P, Lippi G, Maffulli N. 2010; Biochemical markers of muscular damage. Clin Chem Lab Med. 48:757–767. DOI: 10.1515/CCLM.2010.179. PMID: 20518645.
Article23. Aziz MT, El-Asmar MF, Rezq AM, Wassef MA, Fouad H, Roshdy NK, Ahmed HH, Rashed LA, Sabry D, Taha FM, Hassouna A. 2014; Effects of a novel curcumin derivative on insulin synthesis and secretion in streptozotocin-treated rat pancreatic islets in vitro. Chin Med. 9:3. DOI: 10.1186/1749-8546-9-3. PMID: 24422903. PMCID: PMC3896850.
Article24. Steinert AF, Kunz M, Prager P, Göbel S, Klein-Hitpass L, Ebert R, Nöth U, Jakob F, Gohlke F. 2015; Characterization of bursa subacromialis-derived mesenchymal stem cells. Stem Cell Res Ther. 6:114. DOI: 10.1186/s13287-015-0104-3. PMID: 26036250. PMCID: PMC4479225.
Article25. Jarajapu YP, Hazra S, Segal M, Li Calzi S, Jadhao C, Qian K, Mitter SK, Raizada MK, Boulton ME, Grant MB. 2014; Vasoreparative dysfunction of CD34+ cells in diabetic individuals involves hypoxic desensitization and impaired autocrine/paracrine mechanisms. PLoS One. 9:e93965. DOI: 10.1371/journal.pone.0093965. PMID: 24713821. PMCID: PMC3979711.
Article26. Trost SU, Belke DD, Bluhm WF, Meyer M, Swanson E, Dillmann WH. 2002; Overexpression of the sarcoplasmic reticulum Ca(2+)-ATPase improves myocardial contractility in diabetic cardiomyopathy. Diabetes. 51:1166–1171. DOI: 10.2337/diabetes.51.4.1166. PMID: 11916940.
Article27. Pu T, Guo P, Qiu Y, Chen S, Yang L, Sun L, Ye F, Bu H. 2015; Quantitative real-time polymerase chain reaction is an alternative method for the detection of HER-2 amplification in formalin-fixed paraffin-embedded breast cancer samples. Int J Clin Exp Pathol. 8:10565–10574. PMID: 26617766. PMCID: PMC4637581.28. Li W, Du SN, Shi MJ, Sun ZZ. 2016; Spontaneous and transient predinner hyperglycemia in some patients with diabetes: dusk phenomenon. Medicine (Baltimore). 95:e5440. DOI: 10.1097/MD.0000000000005440. PMID: 27893687. PMCID: PMC5134880.29. Beyfuss K, Hood DA. 2018; A systematic review of p53 regulation of oxidative stress in skeletal muscle. Redox Rep. 23:100–117. DOI: 10.1080/13510002.2017.1416773. PMID: 29298131.
Article30. Marrocco V, Fiore P, Benedetti A, Pisu S, Rizzuto E, Musarò A, Madaro L, Lozanoska-Ochser B, Bouché M. 2017; Pharmacological inhibition of PKCθ counteracts muscle disease in a mouse model of duchenne muscular dystrophy. EBioMedicine. 16:150–161. DOI: 10.1016/j.ebiom.2017.01.001. PMID: 28089792. PMCID: PMC5474428.
Article31. Vono R, Fuoco C, Testa S, Pirrò S, Maselli D, Ferland McCollough D, Sangalli E, Pintus G, Giordo R, Finzi G, Sessa F, Cardani R, Gotti A, Losa S, Cesareni G, Rizzi R, Bearzi C, Cannata S, Spinetti G, Gargioli C, Madeddu P. 2016; Activation of the pro-oxidant PKCβII-p66Shc signaling pathway contributes to pericyte dysfunction in skeletal muscles of patients with diabetes with critical limb ischemia. Diabetes. 65:3691–3704. DOI: 10.2337/db16-0248. PMID: 27600065.
Article32. Funai K, Lodhi IJ, Spears LD, Yin L, Song H, Klein S, Semenkovich CF. 2016; Skeletal muscle phospholipid metabolism regulates insulin sensitivity and contractile function. Diabetes. 65:358–370. DOI: 10.2337/db15-0659. PMID: 26512026. PMCID: PMC4747455.
Article33. Anderluh M, Kocic G, Tomovic K, Kocic R, Deljanin-Ilic M, Smelcerovic A. 2016; Cross-talk between the dipeptidyl peptidase-4 and stromal cell-derived factor-1 in stem cell homing and myocardial repair: potential impact of dipeptidyl peptidase-4 inhibitors. Pharmacol Ther. 167:100–107. DOI: 10.1016/j.pharmthera.2016.07.009. PMID: 27484974.
Article34. Souidi N, Stolk M, Rudeck J, Strunk D, Schallmoser K, Volk HD, Seifert M. 2017; Stromal cells act as guardians for endothelial progenitors by reducing their immunogenicity after co-transplantation. Stem Cells. 35:1233–1245. DOI: 10.1002/stem.2573. PMID: 28100035.
Article35. Subramani B, Subbannagounder S, Ramanathanpullai C, Palanivel S, Ramasamy R. 2017; Impaired redox environment modulates cardiogenic and ion-channel gene expression in cardiac-resident and non-resident mesenchymal stem cells. Exp Biol Med (Maywood). 242:645–656. DOI: 10.1177/1535370216688568. PMID: 28092181. PMCID: PMC5685259.
Article36. El Haddad M, Notarnicola C, Evano B, El Khatib N, Blaquière M, Bonnieu A, Tajbakhsh S, Hugon G, Vernus B, Mercier J, Carnac G. 2017; Retinoic acid maintains human skeletal muscle progenitor cells in an immature state. Cell Mol Life Sci. 74:1923–1936. DOI: 10.1007/s00018-016-2445-1. PMID: 28025671.
Article37. De Jesus NM, Wang L, Lai J, Rigor RR, Francis Stuart SD, Bers DM, Lindsey ML, Ripplinger CM. 2017; Antiarrhythmic effects of interleukin 1 inhibition after myocardial infarction. Heart Rhythm. 14:727–736. DOI: 10.1016/j.hrthm.2017.01.027. PMID: 28111350. PMCID: PMC5403568.
Article38. Cui C, Merritt R, Fu L, Pan Z. 2017; Targeting calcium signaling in cancer therapy. Acta Pharm Sin B. 7:3–17. DOI: 10.1016/j.apsb.2016.11.001. PMID: 28119804. PMCID: PMC5237760.
Article39. Kang S, Dahl R, Hsieh W, Shin A, Zsebo KM, Buettner C, Hajjar RJ, Lebeche D. 2016; Small molecular allosteric activator of the Sarco/Endoplasmic Reticulum Ca2+-ATPase (SERCA) attenuates diabetes and metabolic disorders. J Biol Chem. 291:5185–5198. DOI: 10.1074/jbc.M115.705012. PMID: 26702054. PMCID: PMC4777852.
Article40. Allard B. 2018; From excitation to intracellular Ca2+ movements in skeletal muscle: basic aspects and related clinical disorders. Neuromuscul Disord. 28:394–401. DOI: 10.1016/j.nmd.2018.03.004. PMID: 29627324.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Effect of Stem Cells and Gene Transfected Stem Cells Therapy on the Pancreas of Experimentally Induced Type 1 Diabetes
- Questions Surrounding iPS Cells in Japan
- Current Concepts of Stem Cell Therapy
- Clinical Applications of Neural Stem Cells for the Treatment of Peripheral Neuropathy
- Stem Cell Properties of Therapeutic Potential