Tuberc Respir Dis.
2020 Apr;83(2):107-115. 10.4046/trd.2020.0003.
Recent Advances in Molecular Basis of Lung Aging and Its Associated Diseases
- Affiliations
-
- 1Section of Pulmonary, Critical Care and Sleep Medicine, Department of Internal Medicine, Yale University School of Medicine, New Haven, CT, USA. min-jong.kang@yale.edu
- KMID: 2471886
- DOI: http://doi.org/10.4046/trd.2020.0003
Abstract
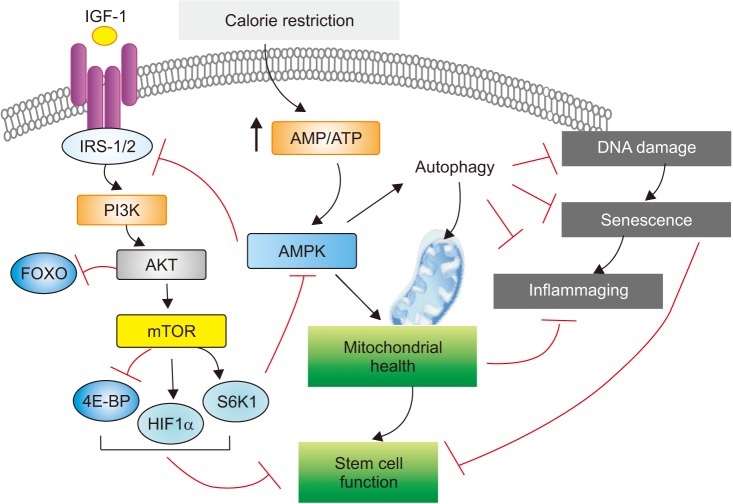
- Aging is often viewed as a progressive decline in fitness due to cumulative deleterious alterations of biological functions in the living system. Recently, our understanding of the molecular mechanisms underlying aging biology has significantly advanced. Interestingly, many of the pivotal molecular features of aging biology are also found to contribute to the pathogenesis of chronic lung disorders such as chronic obstructive pulmonary disease and idiopathic pulmonary fibrosis, for which advanced age is the most crucial risk factor. Thus, an enhanced understanding of how molecular features of aging biology are intertwined with the pathobiology of these aging-related lung disorders has paramount significance and may provide an opportunity for the development of novel therapeutics for these major unmet medical needs. To serve the purpose of integrating molecular understanding of aging biology with pulmonary medicine, in this review, recent findings obtained from the studies of aging-associated lung disorders are summarized and interpreted through the perspective of molecular biology of aging.
Keyword
MeSH Terms
Figure
Reference
-
1. Kaeberlein M, Rabinovitch PS, Martin GM. Healthy aging: the ultimate preventative medicine. Science. 2015; 350:1191–1193. PMID: 26785476.
Article2. Gladyshev VN. Aging: progressive decline in fitness due to the rising deleteriome adjusted by genetic, environmental, and stochastic processes. Aging Cell. 2016; 15:594–602. PMID: 27060562.
Article3. Kirkwood TB. Understanding the odd science of aging. Cell. 2005; 120:437–447. PMID: 15734677.
Article4. Fukuchi Y. The aging lung and chronic obstructive pulmonary disease: similarity and difference. Proc Am Thorac Soc. 2009; 6:570–572. PMID: 19934351.
Article5. Lowery EM, Brubaker AL, Kuhlmann E, Kovacs EJ. The aging lung. Clin Interv Aging. 2013; 8:1489–1496. PMID: 24235821.6. Skloot GS. The effects of aging on lung structure and function. Clin Geriatr Med. 2017; 33:447–457. PMID: 28991643.
Article7. Thannickal VJ, Murthy M, Balch WE, Chandel NS, Meiners S, Eickelberg O, et al. Blue journal conference: aging and susceptibility to lung disease. Am J Respir Crit Care Med. 2015; 191:261–269. PMID: 25590812.
Article8. Budinger GR, Kohanski RA, Gan W, Kobor MS, Amaral LA, Armanios M, et al. The intersection of aging biology and the pathobiology of lung diseases: a Joint NHLBI/NIA Workshop. J Gerontol A Biol Sci Med Sci. 2017; 72:1492–1500. PMID: 28498894.
Article9. MacNee W. Is chronic obstructive pulmonary disease an accelerated aging disease? Ann Am Thorac Soc. 2016; 13 Suppl 5:S429–S437. PMID: 28005421.
Article10. Mora AL, Bueno M, Rojas M. Mitochondria in the spotlight of aging and idiopathic pulmonary fibrosis. J Clin Invest. 2017; 127:405–414. PMID: 28145905.
Article11. Lopez-Otin C, Blasco MA, Partridge L, Serrano M, Kroemer G. The hallmarks of aging. Cell. 2013; 153:1194–1217. PMID: 23746838.
Article12. Szilard L. On the nature of the aging process. Proc Natl Acad Sci U S A. 1959; 45:30–45. PMID: 16590351.
Article13. Moskalev AA, Shaposhnikov MV, Plyusnina EN, Zhavoronkov A, Budovsky A, Yanai H, et al. The role of DNA damage and repair in aging through the prism of Koch-like criteria. Ageing Res Rev. 2013; 12:661–684. PMID: 22353384.
Article14. Niedernhofer LJ, Gurkar AU, Wang Y, Vijg J, Hoeijmakers JH, Robbins PD. Nuclear genomic instability and aging. Annu Rev Biochem. 2018; 87:295–322. PMID: 29925262.
Article15. Blackburn EH, Epel ES, Lin J. Human telomere biology: a contributory and interactive factor in aging, disease risks, and protection. Science. 2015; 350:1193–1198. PMID: 26785477.
Article16. Brunet A, Berger SL. Epigenetics of aging and aging-related disease. J Gerontol A Biol Sci Med Sci. 2014; 69 Suppl 1:S17–S20. PMID: 24833581.
Article17. Sen P, Shah PP, Nativio R, Berger SL. Epigenetic mechanisms of longevity and aging. Cell. 2016; 166:822–839. PMID: 27518561.
Article18. Kaushik S, Cuervo AM. Proteostasis and aging. Nat Med. 2015; 21:1406–1415. PMID: 26646497.
Article19. Most J, Tosti V, Redman LM, Fontana L. Calorie restriction in humans: an update. Ageing Res Rev. 2017; 39:36–45. PMID: 27544442.
Article20. Redman LM, Smith SR, Burton JH, Martin CK, Il'yasova D, Ravussin E. Metabolic slowing and reduced oxidative damage with sustained caloric restriction support the rate of living and oxidative damage theories of aging. Cell Metab. 2018; 27:805–815. PMID: 29576535.
Article21. Fontana L, Partridge L, Longo VD. Extending healthy life span: from yeast to humans. Science. 2010; 328:321–326. PMID: 20395504.22. Rizza W, Veronese N, Fontana L. What are the roles of calorie restriction and diet quality in promoting healthy longevity? Ageing Res Rev. 2014; 13:38–45. PMID: 24291541.
Article23. Sun N, Youle RJ, Finkel T. The mitochondrial basis of aging. Mol Cell. 2016; 61:654–666. PMID: 26942670.
Article24. Akbari M, Kirkwood TB, Bohr VA. Mitochondria in the signaling pathways that control longevity and health span. Ageing Res Rev. 2019; 54:100940. PMID: 31415807.
Article25. Dela Cruz CS, Kang MJ. Mitochondrial dysfunction and damage associated molecular patterns (DAMPs) in chronic inflammatory diseases. Mitochondrion. 2018; 41:37–44. PMID: 29221810.
Article26. Calcinotto A, Kohli J, Zagato E, Pellegrini L, Demaria M, Alimonti A. Cellular senescence: aging, cancer, and injury. Physiol Rev. 2019; 99:1047–1078. PMID: 30648461.
Article27. Gorgoulis V, Adams PD, Alimonti A, Bennett DC, Bischof O, Bishop C, et al. Cellular senescence: defining a path forward. Cell. 2019; 179:813–827. PMID: 31675495.
Article28. He S, Sharpless NE. Senescence in health and disease. Cell. 2017; 169:1000–1011. PMID: 28575665.
Article29. Prata L, Ovsyannikova IG, Tchkonia T, Kirkland JL. Senescent cell clearance by the immune system: emerging therapeutic opportunities. Semin Immunol. 2018; 40:101275. PMID: 31088710.
Article30. Brunauer R, Alavez S, Kennedy BK. Stem cell models: a guide to understand and mitigate aging? Gerontology. 2017; 63:84–90. PMID: 27760416.
Article31. Schultz MB, Sinclair DA. When stem cells grow old: phenotypes and mechanisms of stem cell aging. Development. 2016; 143:3–14. PMID: 26732838.
Article32. Neves J, Sousa-Victor P, Jasper H. Rejuvenating strategies for stem cell-based therapies in aging. Cell Stem Cell. 2017; 20:161–175. PMID: 28157498.
Article33. Riera CE, Merkwirth C, De Magalhaes Filho CD, Dillin A. Signaling networks determining life span. Annu Rev Biochem. 2016; 85:35–64. PMID: 27294438.
Article34. Franceschi C, Bonafe M, Valensin S, Olivieri F, De Luca M, Ottaviani E, et al. Inflamm-aging: an evolutionary perspective on immunosenescence. Ann N Y Acad Sci. 2000; 908:244–254. PMID: 10911963.
Article35. Franceschi C, Garagnani P, Parini P, Giuliani C, Santoro A. Inflammaging: a new immune-metabolic viewpoint for age-related diseases. Nat Rev Endocrinol. 2018; 14:576–590. PMID: 30046148.
Article36. Fulop T, Witkowski JM, Olivieri F, Larbi A. The integration of inflammaging in age-related diseases. Semin Immunol. 2018; 40:17–35. PMID: 30287177.
Article37. Longo VD, Antebi A, Bartke A, Barzilai N, Brown-Borg HM, Caruso C, et al. Interventions to slow aging in humans: are we ready? Aging Cell. 2015; 14:497–510. PMID: 25902704.
Article38. Lopez-Lluch G, Navas P. Calorie restriction as an intervention in ageing. J Physiol. 2016; 594:2043–2060. PMID: 26607973.
Article39. Fontana L, Partridge L. Promoting health and longevity through diet: from model organisms to humans. Cell. 2015; 161:106–118. PMID: 25815989.
Article40. Hegab AE, Ozaki M, Meligy FY, Nishino M, Kagawa S, Ishii M, et al. Calorie restriction enhances adult mouse lung stem cells function and reverses several ageing-induced changes. J Tissue Eng Regen Med. 2019; 13:295–308. PMID: 30562419.
Article41. Hegab AE, Ozaki M, Meligy FY, Kagawa S, Ishii M, Betsuyaku T. High fat diet activates adult mouse lung stem cells and accelerates several aging-induced effects. Stem Cell Res. 2018; 33:25–35. PMID: 30308415.
Article42. Bishai JM, Mitzner W. Effect of severe calorie restriction on the lung in two strains of mice. Am J Physiol Lung Cell Mol Physiol. 2008; 295:L356–L362. PMID: 18515406.
Article43. Chung JH, Manganiello V, Dyck JR. Resveratrol as a calorie restriction mimetic: therapeutic implications. Trends Cell Biol. 2012; 22:546–554. PMID: 22885100.
Article44. Parikh N, Chakraborti AK. Phosphodiesterase 4 (PDE4) inhibitors in the treatment of COPD: promising dug candidates and future directions. Curr Med Chem. 2016; 23:129–141. PMID: 26572614.45. Teumer A, Qi Q, Nethander M, Aschard H, Bandinelli S, Beekman M, et al. Genomewide meta-analysis identifies loci associated with IGF-I and IGFBP-3 levels with impact on age-related traits. Aging Cell. 2016; 15:811–824. PMID: 27329260.46. Stormann S, Gutt B, Roemmler-Zehrer J, Bidlingmaier M, Huber RM, Schopohl J, et al. Assessment of lung function in a large cohort of patients with acromegaly. Eur J Endocrinol. 2017; 177:15–23. PMID: 28432267.47. Burdet L, de Muralt B, Schutz Y, Pichard C, Fitting JW. Administration of growth hormone to underweight patients with chronic obstructive pulmonary disease: a prospective, randomized, controlled study. Am J Respir Crit Care Med. 1997; 156:1800–1806. PMID: 9412558.48. Pape GS, Friedman M, Underwood LE, Clemmons DR. The effect of growth hormone on weight gain and pulmonary function in patients with chronic obstructive lung disease. Chest. 1991; 99:1495–1500. PMID: 2036835.
Article49. Krein PM, Winston BW. Roles for insulin-like growth factor I and transforming growth factor-beta in fibrotic lung disease. Chest. 2002; 122(6 Suppl):289S–293S. PMID: 12475802.50. Pilewski JM, Liu L, Henry AC, Knauer AV, Feghali-Bostwick CA. Insulin-like growth factor binding proteins 3 and 5 are overexpressed in idiopathic pulmonary fibrosis and contribute to extracellular matrix deposition. Am J Pathol. 2005; 166:399–407. PMID: 15681824.
Article51. Johnson SC, Rabinovitch PS, Kaeberlein M. mTOR is a key modulator of ageing and age-related disease. Nature. 2013; 493:338–345. PMID: 23325216.
Article52. Saxton RA, Sabatini DM. mTOR signaling in growth, metabolism, and disease. Cell. 2017; 168:960–976. PMID: 28283069.
Article53. Yoshida T, Mett I, Bhunia AK, Bowman J, Perez M, Zhang L, et al. Rtp801, a suppressor of mTOR signaling, is an essential mediator of cigarette smoke-induced pulmonary injury and emphysema. Nat Med. 2010; 16:767–773. PMID: 20473305.
Article54. Houssaini A, Breau M, Kebe K, Abid S, Marcos E, Lipskaia L, et al. mTOR pathway activation drives lung cell senescence and emphysema. JCI Insight. 2018; 3:93203. PMID: 29415880.
Article55. Saito N, Araya J, Ito S, Tsubouchi K, Minagawa S, Hara H, et al. Involvement of lamin B1 reduction in accelerated cellular senescence during chronic obstructive pulmonary disease pathogenesis. J Immunol. 2019; 202:1428–1440. PMID: 30692212.
Article56. Wang Y, Liu J, Zhou JS, Huang HQ, Li ZY, Xu XC, et al. MTOR suppresses cigarette smoke-induced epithelial cell death and airway inflammation in chronic obstructive pulmonary disease. J Immunol. 2018; 200:2571–2580. PMID: 29507104.
Article57. Wu YF, Li ZY, Dong LL, Li WJ, Wu YP, Wang J, et al. Inactivation of MTOR promotes autophagy-mediated epithelial injury in particulate matter-induced airway inflammation. Autophagy. 2020; 16:435–450. PMID: 31203721.
Article58. Pasini E, Flati V, Comini L, Olivares A, Bertella E, Corsetti G, et al. Mammalian target of rapamycin: is it relevant to COPD pathogenesis or treatment? COPD. 2019; 16:89–92. PMID: 31056947.
Article59. Romero Y, Bueno M, Ramirez R, Alvarez D, Sembrat JC, Goncharova EA, et al. mTORC1 activation decreases autophagy in aging and idiopathic pulmonary fibrosis and contributes to apoptosis resistance in IPF fibroblasts. Aging Cell. 2016; 15:1103–1112. PMID: 27566137.60. Allen RJ, Guillen-Guio B, Oldham JM, Ma SF, Dressen A, Paynton ML, et al. Genome-wide association study of susceptibility to idiopathic pulmonary fibrosis. Am J Respir Crit Care Med. 2019; 11. 11. DOI: 10.1164/rccm.201905-1017OC. [Epub].61. Gokey JJ, Sridharan A, Xu Y, Green J, Carraro G, Stripp BR, et al. Active epithelial Hippo signaling in idiopathic pulmonary fibrosis. JCI Insight. 2018; 3:98738. PMID: 29563341.
Article62. Moya IM, Halder G. Hippo-YAP/TAZ signalling in organ regeneration and regenerative medicine. Nat Rev Mol Cell Biol. 2019; 20:211–226. PMID: 30546055.
Article63. Woodcock HV, Eley JD, Guillotin D, Plate M, Nanthakumar CB, Martufi M, et al. The mTORC1/4E-BP1 axis represents a critical signaling node during fibrogenesis. Nat Commun. 2019; 10:6. PMID: 30602778.
Article64. Lukey PT, Harrison SA, Yang S, Man Y, Holman BF, Rashidnasab A, et al. A randomised, placebo-controlled study of omipalisib (PI3K/mTOR) in idiopathic pulmonary fibrosis. Eur Respir J. 2019; 53:1801992. PMID: 30765508.
Article65. Mercer PF, Woodcock HV, Eley JD, Plate M, Sulikowski MG, Durrenberger PF, et al. Exploration of a potent PI3 kinase/mTOR inhibitor as a novel anti-fibrotic agent in IPF. Thorax. 2016; 71:701–711. PMID: 27103349.
Article66. Lawrence J, Nho R. The role of the mammalian target of rapamycin (mTOR) in pulmonary fibrosis. Int J Mol Sci. 2018; 19:E778. PMID: 29518028.
Article67. Herzig S, Shaw RJ. AMPK: guardian of metabolism and mitochondrial homeostasis. Nat Rev Mol Cell Biol. 2018; 19:121–135. PMID: 28974774.
Article68. O'Callaghan C, Vassilopoulos A. Sirtuins at the crossroads of stemness, aging, and cancer. Aging Cell. 2017; 16:1208–1218. PMID: 28994177.69. Lee SH, Lee JH, Lee HY, Min KJ. Sirtuin signaling in cellular senescence and aging. BMB Rep. 2019; 52:24–34. PMID: 30526767.
Article70. Canto C, Gerhart-Hines Z, Feige JN, Lagouge M, Noriega L, Milne JC, et al. AMPK regulates energy expenditure by modulating NAD+ metabolism and SIRT1 activity. Nature. 2009; 458:1056–1060. PMID: 19262508.
Article71. Rajendrasozhan S, Yang SR, Kinnula VL, Rahman I. SIRT1, an antiinflammatory and antiaging protein, is decreased in lungs of patients with chronic obstructive pulmonary disease. Am J Respir Crit Care Med. 2008; 177:861–870. PMID: 18174544.
Article72. Yao H, Chung S, Hwang JW, Rajendrasozhan S, Sundar IK, Dean DA, et al. SIRT1 protects against emphysema via FOXO3-mediated reduction of premature senescence in mice. J Clin Invest. 2012; 122:2032–2045. PMID: 22546858.
Article73. Takasaka N, Araya J, Hara H, Ito S, Kobayashi K, Kurita Y, et al. Autophagy induction by SIRT6 through attenuation of insulin-like growth factor signaling is involved in the regulation of human bronchial epithelial cell senescence. J Immunol. 2014; 192:958–968. PMID: 24367027.
Article74. Minagawa S, Araya J, Numata T, Nojiri S, Hara H, Yumino Y, et al. Accelerated epithelial cell senescence in IPF and the inhibitory role of SIRT6 in TGF-beta-induced senescence of human bronchial epithelial cells. Am J Physiol Lung Cell Mol Physiol. 2011; 300:L391–L401. PMID: 21224216.75. Wyman AE, Noor Z, Fishelevich R, Lockatell V, Shah NG, Todd NW, et al. Sirtuin 7 is decreased in pulmonary fibrosis and regulates the fibrotic phenotype of lung fibroblasts. Am J Physiol Lung Cell Mol Physiol. 2017; 312:L945–L958. PMID: 28385812.
Article76. Hwang JW, Yao H, Caito S, Sundar IK, Rahman I. Redox regulation of SIRT1 in inflammation and cellular senescence. Free Radic Biol Med. 2013; 61:95–110. PMID: 23542362.
Article77. Piskovatska V, Stefanyshyn N, Storey KB, Vaiserman AM, Lushchak O. Metformin as a geroprotector: experimental and clinical evidence. Biogerontology. 2019; 20:33–48. PMID: 30255224.
Article78. Foretz M, Guigas B, Bertrand L, Pollak M, Viollet B. Metformin: from mechanisms of action to therapies. Cell Metab. 2014; 20:953–966. PMID: 25456737.
Article79. Bishwakarma R, Zhang W, Lin YL, Kuo YF, Cardenas VJ, Sharma G. Metformin use and health care utilization in patients with coexisting chronic obstructive pulmonary disease and diabetes mellitus. Int J Chron Obstruct Pulmon Dis. 2018; 13:793–800. PMID: 29551895.
Article80. Zhu A, Teng Y, Ge D, Zhang X, Hu M, Yao X. Role of metformin in treatment of patients with chronic obstructive pulmonary disease: a systematic review. J Thorac Dis. 2019; 11:4371–4378. PMID: 31737323.
Article81. Rangarajan S, Bone NB, Zmijewska AA, Jiang S, Park DW, Bernard K, et al. Metformin reverses established lung fibrosis in a bleomycin model. Nat Med. 2018; 24:1121–1127. PMID: 29967351.
Article82. Kheirollahi V, Wasnick RM, Biasin V, Vazquez-Armendariz AI, Chu X, Moiseenko A, et al. Metformin induces lipogenic differentiation in myofibroblasts to reverse lung fibrosis. Nat Commun. 2019; 10:2987. PMID: 31278260.
Article83. Spagnolo P, Kreuter M, Maher TM, Wuyts W, Bonella F, Corte TJ, et al. Metformin does not affect clinically relevant outcomes in patients with idiopathic pulmonary fibrosis. Respiration. 2018; 96:314–322. PMID: 30025392.
Article84. Tchkonia T, Zhu Y, van Deursen J, Campisi J, Kirkland JL. Cellular senescence and the senescent secretory phenotype: therapeutic opportunities. J Clin Invest. 2013; 123:966–972. PMID: 23454759.
Article85. Royce GH, Brown-Borg HM, Deepa SS. The potential role of necroptosis in inflammaging and aging. Geroscience. 2019; 41:795–811. PMID: 31721033.
Article