J Lipid Atheroscler.
2020 Jan;9(1):66-78. 10.12997/jla.2020.9.1.66.
CD36 (SR-B2) as a Target to Treat Lipid Overload-Induced Cardiac Dysfunction
- Affiliations
-
- 1Department of Genetics & Cell Biology, Faculty of Health, Medicine and Life Sciences, Maastricht University, Maastricht, the Netherlands. glatz@maastrichtuniversity.nl
- KMID: 2470801
- DOI: http://doi.org/10.12997/jla.2020.9.1.66
Abstract
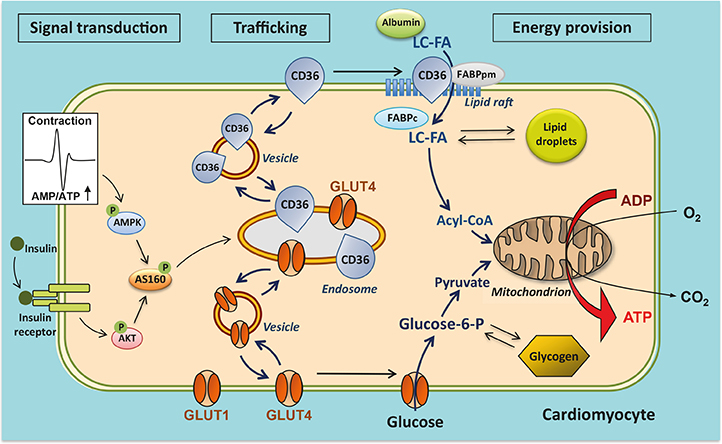
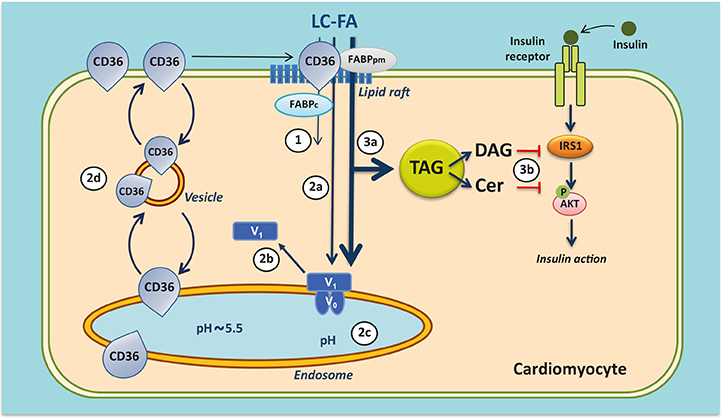
- The heart faces the challenge of adjusting the rate of fatty acid uptake to match myocardial demand for energy provision at any given moment, avoiding both too low uptake rates, which could elicit an energy deficit, and too high uptake rates, which pose the risk of excess lipid accumulation and lipotoxicity. The transmembrane glycoprotein cluster of differentiation 36 (CD36), a scavenger receptor (B2), serves many functions in lipid metabolism and signaling. In the heart, CD36 is the main sarcolemmal lipid transporter involved in the rate-limiting kinetic step in cardiac lipid utilization. The cellular fatty acid uptake rate is determined by the presence of CD36 at the cell surface, which is regulated by subcellular vesicular recycling from endosomes to the sarcolemma. CD36 has been implicated in dysregulated fatty acid and lipid metabolism in pathophysiological conditions, particularly high-fat diet-induced insulin resistance and diabetic cardiomyopathy. Thus, in conditions of chronic lipid overload, high levels of CD36 are moved to the sarcolemma, setting the heart on a route towards increased lipid uptake, excessive lipid accumulation, insulin resistance, and eventually contractile dysfunction. Insight into the subcellular trafficking machinery of CD36 will provide novel targets to treat the lipid-overloaded heart. A screen for CD36-dedicated trafficking proteins found that vacuolar-type Hâº-ATPase and specific vesicle-associated membrane proteins, among others, were uniquely involved in CD36 recycling. Preliminary data suggest that these proteins may offer clues on how to manipulate myocardial lipid uptake, and thus could be promising targets for metabolic intervention therapy to treat the failing heart.
Keyword
MeSH Terms
Figure
Reference
-
1. Longo M, Zatterale F, Naderi J, Parrillo L, Formisano P, Raciti GA, et al. Adipose tissue dysfunction as determinant of obesity-associated metabolic complications. Int J Mol Sci. 2019; 20:E2358.
Article2. Sharma S, Adrogue JV, Golfman L, Uray I, Lemm J, Youker K, et al. Intramyocardial lipid accumulation in the failing human heart resembles the lipotoxic rat heart. FASEB J. 2004; 18:1692–1700.
Article3. Boudina S, Abel ED. Diabetic cardiomyopathy, causes and effects. Rev Endocr Metab Disord. 2010; 11:31–39.
Article4. Zlobine I, Gopal K, Ussher JR. Lipotoxicity in obesity and diabetes-related cardiac dysfunction. Biochim Biophys Acta. 2016; 1861:1555–1568.
Article5. Stanley WC, Recchia FA, Lopaschuk GD. Myocardial substrate metabolism in the normal and failing heart. Physiol Rev. 2005; 85:1093–1129.
Article6. Glatz JF, Luiken JJ. From fat to FAT (CD36/SR-B2): understanding the regulation of cellular fatty acid uptake. Biochimie. 2017; 136:21–26.
Article7. Glatz JFC, Luiken JJFP. Dynamic role of the transmembrane glycoprotein CD36 (SR-B2) in cellular fatty acid uptake and utilization. J Lipid Res. 2018; 59:1084–1093.
Article8. Prabhudas M, Bowdish D, Drickamer K, Febbraio M, Herz J, Kobzik L, et al. Standardizing scavenger receptor nomenclature. J Immunol. 2014; 192:1997–2006.
Article9. Stremmel W, Lotz G, Strohmeyer G, Berk PD. Identification, isolation, and partial characterization of a fatty acid binding protein from rat jejunal microvillous membranes. J Clin Invest. 1985; 75:1068–1076.
Article10. Kazantzis M, Stahl A. Fatty acid transport proteins, implications in physiology and disease. Biochim Biophys Acta. 2012; 1821:852–857.
Article11. Hsieh FL, Turner L, Bolla JR, Robinson CV, Lavstsen T, Higgins MK. The structural basis for CD36 binding by the malaria parasite. Nat Commun. 2016; 7:12837.
Article12. Hamilton JA. New insights into the roles of proteins and lipids in membrane transport of fatty acids. Prostaglandins Leukot Essent Fatty Acids. 2007; 77:355–361.
Article13. Luiken JJ, Willems J, van der Vusse GJ, Glatz JF. Electrostimulation enhances FAT/CD36-mediated long-chain fatty acid uptake by isolated rat cardiac myocytes. Am J Physiol Endocrinol Metab. 2001; 281:E704–E712.
Article14. Luiken JJ, Coort SL, Willems J, Coumans WA, Bonen A, van der Vusse GJ, et al. Contraction-induced fatty acid translocase/CD36 translocation in rat cardiac myocytes is mediated through AMP-activated protein kinase signaling. Diabetes. 2003; 52:1627–1634.
Article15. Bonen A, Luiken JJ, Arumugam Y, Glatz JF, Tandon NN. Acute regulation of fatty acid uptake involves the cellular redistribution of fatty acid translocase. J Biol Chem. 2000; 275:14501–14508.
Article16. van Oort MM, van Doorn JM, Bonen A, Glatz JF, van der Horst DJ, Rodenburg KW, et al. Insulin-induced translocation of CD36 to the plasma membrane is reversible and shows similarity to that of GLUT4. Biochim Biophys Acta. 2008; 1781:61–71.
Article17. Klip A, McGraw TE, James DE. Thirty sweet years of GLUT4. J Biol Chem. 2019; 294:11369–11381.
Article18. Glatz JF, Luiken JJ, Bonen A. Membrane fatty acid transporters as regulators of lipid metabolism: implications for metabolic disease. Physiol Rev. 2010; 90:367–417.
Article19. Habets DD, Coumans WA, Voshol PJ, den Boer MA, Febbraio M, Bonen A, et al. AMPK-mediated increase in myocardial long-chain fatty acid uptake critically depends on sarcolemmal CD36. Biochem Biophys Res Commun. 2007; 355:204–210.
Article20. Kerner J, Hoppel C. Fatty acid import into mitochondria. Biochim Biophys Acta. 2000; 1486:1–17.
Article21. Glatz JFC, Luiken JJFP. Control of myocardial fatty acid uptake. In : Lopaschuk GD, Dhalla NS, editors. Cardiac energy metabolism in health and disease. New York (NY): Springer Science Inc.;2014. p. 49–67.22. Luiken JJ, Niessen HE, Coort SL, Hoebers N, Coumans WA, Schwenk RW, et al. Etomoxir-induced partial carnitine palmitoyltransferase-I (CPT-I) inhibition in vivo does not alter cardiac long-chain fatty acid uptake and oxidation rates. Biochem J. 2009; 419:447–455.
Article23. Ouwens DM, Diamant M. Myocardial insulin action and the contribution of insulin resistance to the pathogenesis of diabetic cardiomyopathy. Arch Physiol Biochem. 2007; 113:76–86.
Article24. Bonen A, Jain SS, Snook LA, Han XX, Yoshida Y, Buddo KH, et al. Extremely rapid increase in fatty acid transport and intramyocellular lipid accumulation but markedly delayed insulin resistance after high fat feeding in rats. Diabetologia. 2015; 58:2381–2391.
Article25. Shulman GI. Cellular mechanisms of insulin resistance. J Clin Invest. 2000; 106:171–176.
Article26. Ouwens DM, Diamant M, Fodor M, Habets DD, Pelsers MM, El Hasnaoui M, et al. Cardiac contractile dysfunction in insulin-resistant rats fed a high-fat diet is associated with elevated CD36-mediated fatty acid uptake and esterification. Diabetologia. 2007; 50:1938–1948.
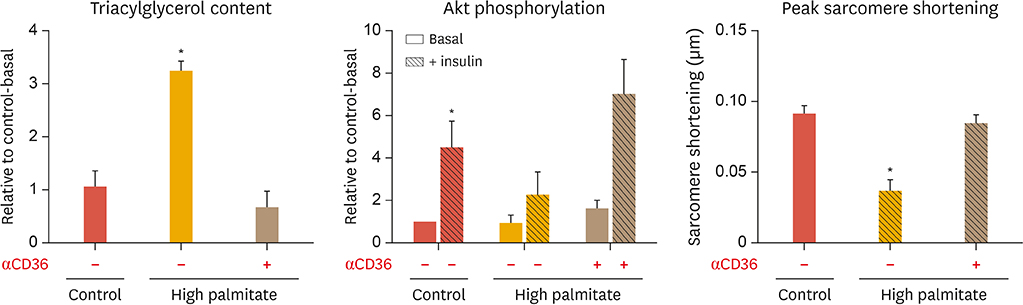
Article27. Angin Y, Steinbusch LK, Simons PJ, Greulich S, Hoebers NT, Douma K, et al. CD36 inhibition prevents lipid accumulation and contractile dysfunction in rat cardiomyocytes. Biochem J. 2012; 448:43–53.
Article28. Steinbusch LK, Luiken JJ, Vlasblom R, Chabowski A, Hoebers NT, Coumans WA, et al. Absence of fatty acid transporter CD36 protects against Western-type diet-related cardiac dysfunction following pressure overload in mice. Am J Physiol Endocrinol Metab. 2011; 301:E618–E627.
Article29. Sung MM, Koonen DP, Soltys CL, Jacobs RL, Febbraio M, Dyck JR. Increased CD36 expression in middle-aged mice contributes to obesity-related cardiac hypertrophy in the absence of cardiac dysfunction. J Mol Med (Berl). 2011; 89:459–469.
Article30. Zhu B, Li MY, Lin Q, Liang Z, Xin Q, Wang M, et al. Lipid oversupply induces CD36 sarcolemmal translocation via dual modulation of PKCζ and TBC1D1: an early event prior to insulin resistance. Theranostics. 2020; 10:1332–1354.
Article31. Silverstein RL, Febbraio M. CD36, a scavenger receptor involved in immunity, metabolism, angiogenesis, and behavior. Sci Signal. 2009; 2:re3.
Article32. Chi RJ, Harrison MS, Burd CG. Biogenesis of endosome-derived transport carriers. Cell Mol Life Sci. 2015; 72:3441–3455.
Article33. Glatz JF, Nabben M, Heather LC, Bonen A, Luiken JJ. Regulation of the subcellular trafficking of CD36, a major determinant of cardiac fatty acid utilization. Biochim Biophys Acta. 2016; 1861:1461–1471.
Article34. Steinbusch LK, Wijnen W, Schwenk RW, Coumans WA, Hoebers NT, Ouwens DM, et al. Differential regulation of cardiac glucose and fatty acid uptake by endosomal pH and actin filaments. Am J Physiol Cell Physiol. 2010; 298:C1549–C1559.
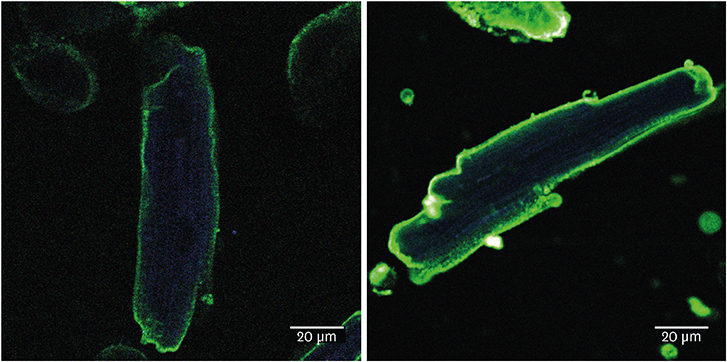
Article35. Liu Y, Steinbusch LK, Nabben M, Kapsokalyvas D, van Zandvoort M, Schönleitner P, et al. Palmitate-induced vacuolar-type H+-ATPase inhibition feeds forward into insulin resistance and contractile dysfunction. Diabetes. 2017; 66:1521–1534.
Article36. Kane PM. Targeting reversible disassembly as a mechanism of controlling V-ATPase activity. Curr Protein Pept Sci. 2012; 13:117–123.
Article37. McGuire C, Stransky L, Cotter K, Forgac M. Regulation of V-ATPase activity. Front Biosci (Landmark Ed). 2017; 22:609–622.
Article38. Bond S, Forgac M. The Ras/cAMP/protein kinase A pathway regulates glucose-dependent assembly of the vacuolar (H+)-ATPase in yeast. J Biol Chem. 2008; 283:36513–36521.
Article39. Schwenk RW, Dirkx E, Coumans WA, Bonen A, Klip A, Glatz JF, et al. Requirement for distinct vesicle-associated membrane proteins in insulin- and AMP-activated protein kinase (AMPK)-induced translocation of GLUT4 and CD36 in cultured cardiomyocytes. Diabetologia. 2010; 53:2209–2219.
Article40. Sabra MM, Damcott C, Fu M, Ott S, O'Connell JR, Mitchell BD, et al. Vesicle-associated membrane protein 4, a positional candidate gene on 1q24-q25, is not associated with type 2 diabetes in the Old Order Amish. Mol Genet Metab. 2005; 85:133–139.
Article41. Geraets IM, Glatz JF, Luiken JJ, Nabben M. Pivotal role of membrane substrate transporters on the metabolic alterations in the pressure-overloaded heart. Cardiovasc Res. 2019; 115:1000–1012.
Article42. Glatz JFC, Nabben M, Young ME, Schulze PC, Taegtmeyer H, Luiken JJFP. Re-balancing cellular energy substrate metabolism to mend the failing heart. Biochim Biophys Acta Mol Basis Dis. 2020.
Article43. Luiken JJ, Chanda D, Nabben M, Neumann D, Glatz JF. Post-translational modifications of CD36 (SR-B2): implications for regulation of myocellular fatty acid uptake. Biochim Biophys Acta. 2016; 1862:2253–2258.
Article44. Son NH, Basu D, Samovski D, Pietka TA, Peche VS, Willecke F, et al. Endothelial cell CD36 optimizes tissue fatty acid uptake. J Clin Invest. 2018; 128:4329–4342.
Article45. Keizer HA, Schaart G, Tandon NN, Glatz JF, Luiken JJ. Subcellular immunolocalisation of fatty acid translocase (FAT)/CD36 in human type-1 and type-2 skeletal muscle fibres. Histochem Cell Biol. 2004; 121:101–107.
Article46. Puchalska P, Crawford PA. Multi-dimensional roles of ketone bodies in fuel metabolism, signaling, and therapeutics. Cell Metab. 2017; 25:262–284.
Article47. Aquilani R, La Rovere MT, Corbellini D, Pasini E, Verri M, Barbieri A, et al. Plasma amino acid abnormalities in chronic heart failure. Mechanisms, potential risks and targets in human myocardium metabolism. Nutrients. 2017; 9:E1251.
Article48. Chen L, Song J, Hu S. Metabolic remodeling of substrate utilization during heart failure progression. Heart Fail Rev. 2019; 24:143–154.
Article49. Wende AR, Brahma MK, McGinnis GR, Young ME. Metabolic origins of heart failure. JACC Basic Transl Sci. 2017; 2:297–310.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Cilostazol Attenuates 4-hydroxynonenal-enhanced CD36 Expression on Murine Macrophages via Inhibition of NADPH Oxidase-derived Reactive Oxygen Species Production
- Comparing the Effects of Carvedilol Enantiomers on Regression of Established Cardiac Hypertrophy Induced by Pressure Overload
- The Role of CD36 in Type 2 Diabetes Mellitus: β-Cell Dysfunction and Beyond
- Lipid A as a Drug Target and Therapeutic Molecule
- CD36, a scavenger receptor implicated in atherosclerosis