J Korean Orthop Assoc.
2020 Feb;55(1):9-28. 10.4055/jkoa.2020.55.1.9.
Current Guidelines for Non-Steroidal Anti-Inflammatory Drugs
- Affiliations
-
- 1Department of Orthopedic Surgery, Ewha Womans University Mokdong Hospital, Seoul, Korea. koreanknee@gmail.com
- KMID: 2470765
- DOI: http://doi.org/10.4055/jkoa.2020.55.1.9
Abstract
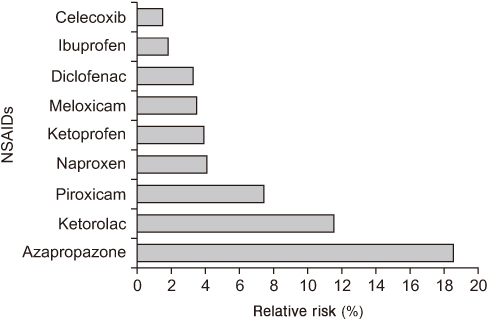
- Non-steroidal anti-inflammatory drugs (NSAIDs) are the most commonly used drugs worldwide for chronic pain, such as arthritis, and there are many different types depending on their composition and mechanism. After long-term use, various side effects can occur, such as gastrointestinal and cardiovascular complications. With a similar analgesic effect to that of traditional non-selective NSAIDs, cyclooxygenase-2-selective NSAIDs have been highly anticipated, because they could complement gastrointestinal tolerance. On the other hand, because of concerns about cardiovascular safety in 2004 and 2005, and the license withdrawals of rofecoxib and valdecoxib, the interest in the side effects of NSAIDs is increasing. Therefore, it is important to use the necessary drugs at a minimum, considering the side effects and interactions of each drug. This study examined the side effects and characteristics of each NSAID that may occur and reviewed the recent research and guidelines related to the use of non-selective NSAIDs and cyclooxygenase-2-selective NSAIDs.
Keyword
MeSH Terms
Figure
Reference
-
1. Abdulla A, Adams N, Bone M, et al. Guidance on the management of pain in older people. Age Ageing. 2013; 42 Suppl 1:i1–i57.2. Harirforoosh S, Asghar W, Jamali F. Adverse effects of nonsteroidal antiinflammatory drugs: an update of gastrointestinal, cardiovascular and renal complications. J Pharm Pharm Sci. 2013; 16:821–847.
Article3. Day RO, Graham GG, Williams KM. Pharmacokinetics of non-steroidal anti-inflammatory drugs. Baillieres Clin Rheumatol. 1988; 2:363–393.
Article4. Davies NM, Anderson KE. Clinical pharmacokinetics of naproxen. Clin Pharmacokinet. 1997; 32:268–293.
Article5. Davies NM, McLachlan AJ, Day RO, Williams KM. Clinical pharmacokinetics and pharmacodynamics of celecoxib: a selective cyclo-oxygenase-2 inhibitor. Clin Pharmacokinet. 2000; 38:225–242.6. Rainsford KD. Ibuprofen: pharmacology, efficacy and safety. Inflammopharmacology. 2009; 17:275–342.
Article7. Chou R, Helfand M, Peterson K, Dana T, Roberts C. Comparative effectiveness and safety of analgesics for osteoarthritis [Internet]. 2006. cited 2019 Jun 10. Available from: http://www.ncbi.nlm.nih.gov/books/NBK42994/.8. Chou R, Helfand M, Peterson K, Dana T, Roberts C. Drug class review: cyclo-oxygenase (COX)-2 inhibitors and non-steroidal anti-inflammatory drugs (NSAIDs): final report update 3 [Internet]. 2006. cited 2019 Jun 10. Available from: http://www.ncbi.nlm.nih.gov/books/NBK10620/.9. Chen YF, Jobanputra P, Barton P, et al. Cyclooxygenase-2 selective non-steroidal anti-inflammatory drugs (etodolac, meloxicam, celecoxib, rofecoxib, etoricoxib, valdecoxib and lumiracoxib) for osteoarthritis and rheumatoid arthritis: a systematic review and economic evaluation. Health Technol Assess. 2008; 12:1–278. iii
Article10. Bjordal JM, Ljunggren AE, Klovning A, Slørdal L. Non-steroidal anti-inflammatory drugs, including cyclo-oxygenase-2 inhibitors, in osteoarthritic knee pain: meta-analysis of randomised placebo controlled trials. BMJ. 2004; 329:1317.
Article11. Bezsheiko V. Comparative efficacy of nonsteroidal anti-inflammatory drugs (NSAIDs). Psychosom Med General Pract. 2017; 2:e020348.12. Smith SR, Deshpande BR, Collins JE, Katz JN, Losina E. Comparative pain reduction of oral non-steroidal anti-inflammatory drugs and opioids for knee osteoarthritis: systematic analytic review. Osteoarthritis Cartilage. 2016; 24:962–972.
Article13. Singh G, Fort JG, Goldstein JL, et al. Celecoxib versus naproxen and diclofenac in osteoarthritis patients: SUCCESS-I study. Am J Med. 2006; 119:255–266.
Article14. Goldstein JL, Hochberg MC, Fort JG, Zhang Y, Hwang C, Sostek M. Clinical trial: the incidence of NSAID-associated endoscopic gastric ulcers in patients treated with PN 400 (naproxen plus esomeprazole magnesium) vs. enteric-coated naproxen alone. Aliment Pharmacol Ther. 2010; 32:401–413.
Article15. Bensen WG, Zhao SZ, Burke TA, et al. Upper gastrointestinal tolerability of celecoxib, a COX-2 specific inhibitor, compared to naproxen and placebo. J Rheumatol. 2000; 27:1876–1883.16. Kivitz AJ, Moskowitz RW, Woods E, et al. Comparative efficacy and safety of celecoxib and naproxen in the treatment of osteoarthritis of the hip. J Int Med Res. 2001; 29:467–479.
Article17. McKenna F, Borenstein D, Wendt H, Wallemark C, Lefkowith JB, Geis GS. Celecoxib versus diclofenac in the management of osteoarthritis of the knee. Scand J Rheumatol. 2001; 30:11–18.18. Hochberg MC, Fort JG, Svensson O, Hwang C, Sostek M. Fixed-dose combination of enteric-coated naproxen and immediate-release esomeprazole has comparable efficacy to celecoxib for knee osteoarthritis: two randomized trials. Curr Med Res Opin. 2011; 27:1243–1253.
Article19. Bingham CO 3rd, Sebba AI, Rubin BR, et al. Efficacy and safety of etoricoxib 30 mg and celecoxib 200 mg in the treatment of osteoarthritis in two identically designed, randomized, placebo-controlled, non-inferiority studies. Rheumatology (Oxford). 2007; 46:496–507.
Article20. Wang R, Dasgupta A, Ward MM. Comparative efficacy of non-steroidal anti-inflammatory drugs in ankylosing spondylitis: a Bayesian network meta-analysis of clinical trials. Ann Rheum Dis. 2016; 75:1152–1160.
Article21. Lazzaroni M, Bianchi Porro G. Gastrointestinal side-effects of traditional non-steroidal anti-inflammatory drugs and new formulations. Aliment Pharmacol Ther. 2004; 20 Suppl 2:48–58.
Article22. Laine L. Nonsteroidal anti-inflammatory drug gastropathy. Gastrointest Endosc Clin N Am. 1996; 6:489–504.
Article23. Singh G, Triadafilopoulos G. Epidemiology of NSAID induced gastrointestinal complications. J Rheumatol Suppl. 1999; 56:18–24.24. Gabriel SE, Jaakkimainen L, Bombardier C. Risk for serious gastrointestinal complications related to use of nonsteroidal anti-inflammatory drugs. A meta-analysis. Ann Intern Med. 1991; 115:787–796.25. Russell RI. Defining patients at risk of non-steroidal anti-inflammatory drug gastropathy. Ital J Gastroenterol Hepatol. 1999; 31 Suppl 1:S14–S18.26. Silverstein FE, Faich G, Goldstein JL, et al. Gastrointestinal toxicity with celecoxib vs nonsteroidal anti-inflammatory drugs for osteoarthritis and rheumatoid arthritis: the CLASS study: a randomized controlled trial. Celecoxib long-term arthritis safety study. JAMA. 2000; 284:1247–1255.27. Castellsague J, Riera-Guardia N, Calingaert B, et al. Safety of Non-Steroidal Anti-Inflammatory Drugs (SOS) Project. Individual NSAIDs and upper gastrointestinal complications: a systematic review and meta-analysis of observational studies (the SOS project). Drug Saf. 2012; 35:1127–1146.28. Chan FKL, Ching JYL, Tse YK, et al. Gastrointestinal safety of celecoxib versus naproxen in patients with cardiothrombotic diseases and arthritis after upper gastrointestinal bleeding (CONCERN): an industry-independent, double-blind, double-dummy, randomised trial. Lancet. 2017; 389:2375–2382.
Article29. Laine L, Connors LG, Reicin A, et al. Serious lower gastrointestinal clinical events with nonselective NSAID or coxib use. Gastroenterology. 2003; 124:288–292.
Article30. Higuchi K, Umegaki E, Watanabe T, et al. Present status and strategy of NSAIDs-induced small bowel injury. J Gastroenterol. 2009; 44:879–888.
Article31. Maiden L, Thjodleifsson B, Theodors A, Gonzalez J, Bjarnason I. A quantitative analysis of NSAID-induced small bowel pathology by capsule enteroscopy. Gastroenterology. 2005; 128:1172–1178.
Article32. Graham DY, Opekun AR, Willingham FF, Qureshi WA. Visible small-intestinal mucosal injury in chronic NSAID users. Clin Gastroenterol Hepatol. 2005; 3:55–59.
Article33. Lanas A, Sopeña F. Nonsteroidal anti-inflammatory drugs and lower gastrointestinal complications. Gastroenterol Clin North Am. 2009; 38:333–352.
Article34. Lanas A, García-Rodríguez LA, Polo-Tomás M, et al. Time trends and impact of upper and lower gastrointestinal bleeding and perforation in clinical practice. Am J Gastroenterol. 2009; 104:1633–1641.
Article35. Marlicz W, Loniewski I, Grimes DS, Quigley EM. Nonsteroidal anti-inflammatory drugs, proton pump inhibitors, and gastrointestinal injury: contrasting interactions in the stomach and small intestine. Mayo Clin Proc. 2014; 89:1699–1709.
Article36. Watanabe T, Sugimori S, Kameda N, et al. Small bowel injury by low-dose enteric-coated aspirin and treatment with misoprostol: a pilot study. Clin Gastroenterol Hepatol. 2008; 6:1279–1282.
Article37. Goldstein JL, Eisen GM, Lewis B, Gralnek IM, Zlotnick S, Fort JG. Video capsule endoscopy to prospectively assess small bowel injury with celecoxib, naproxen plus omeprazole, and placebo. Clin Gastroenterol Hepatol. 2005; 3:133–141.
Article38. Goldstein JL, Eisen GM, Lewis B, et al. Small bowel mucosal injury is reduced in healthy subjects treated with celecoxib compared with ibuprofen plus omeprazole, as assessed by video capsule endoscopy. Aliment Pharmacol Ther. 2007; 25:1211–1222.
Article39. Lanza FL, Chan FK, Quigley EM. Practice Parameters Committee of the American College of Gastroenterology. Guidelines for prevention of NSAID-related ulcer complications. Am J Gastroenterol. 2009; 104:728–738.
Article40. Chan FK, Lanas A, Scheiman J, Berger MF, Nguyen H, Goldstein JL. Celecoxib versus omeprazole and diclofenac in patients with osteoarthritis and rheumatoid arthritis (CONDOR): a randomised trial. Lancet. 2010; 376:173–179.
Article41. Cryer B, Li C, Simon LS, Singh G, Stillman MJ, Berger MF. GI-REASONS: a novel 6-month, prospective, randomized, open-label, blinded endpoint (PROBE) trial. Am J Gastroenterol. 2013; 108:392–400.
Article42. Rostom A, Wells G, Tugwell P, Welch V, Dubé C, McGowan J. The prevention of chronic NSAID induced upper gastrointestinal toxicity: a Cochrane collaboration metaanalysis of randomized controlled trials. J Rheumatol. 2000; 27:2203–2214.43. Silverstein FE, Graham DY, Senior JR, et al. Misoprostol reduces serious gastrointestinal complications in patients with rheumatoid arthritis receiving nonsteroidal anti-inflammatory drugs. A randomized, double-blind, placebo-controlled trial. Ann Intern Med. 1995; 123:241–249.44. Bocanegra TS, Weaver AL, Tindall EA, et al. Diclofenac/misoprostol compared with diclofenac in the treatment of osteoarthritis of the knee or hip: a randomized, placebo controlled trial. Arthrotec Osteoarthritis Study Group. J Rheumatol. 1998; 25:1602–1611.45. Graham DY, Agrawal NM, Campbell DR, et al. NSAID-Associated Gastric Ulcer Prevention Study Group. Ulcer prevention in long-term users of nonsteroidal anti-inflammatory drugs: results of a double-blind, randomized, multicenter, active- and placebo-controlled study of misoprostol vs lansoprazole. Arch Intern Med. 2002; 162:169–175.46. Ray WA, Chung CP, Stein CM, et al. Risk of peptic ulcer hospitalizations in users of NSAIDs with gastroprotective cotherapy versus coxibs. Gastroenterology. 2007; 133:790–798.
Article47. Chan FK, Wong VW, Suen BY, et al. Combination of a cyclo-oxygenase-2 inhibitor and a proton-pump inhibitor for prevention of recurrent ulcer bleeding in patients at very high risk: a double-blind, randomised trial. Lancet. 2007; 369:1621–1626.
Article48. Laine L, Jensen DM. Management of patients with ulcer bleeding. Am J Gastroenterol. 2012; 107:345–360. quiz 361.
Article49. Bruyère O, Cooper C, Pelletier JP, et al. A consensus statement on the European Society for Clinical and Economic Aspects of Osteoporosis and Osteoarthritis (ESCEO) algorithm for the management of knee osteoarthritis-from evidence-based medicine to the real-life setting. Semin Arthritis Rheum. 2016; 45(4 Suppl):S3–S11.
Article50. Scheiman JM, Yeomans ND, Talley NJ, et al. Prevention of ulcers by esomeprazole in at-risk patients using non-selective NSAIDs and COX-2 inhibitors. Am J Gastroenterol. 2006; 101:701–710.
Article51. Sostek MB, Fort JG, Estborn L, Vikman K. Long-term safety of naproxen and esomeprazole magnesium fixed-dose combination: phase III study in patients at risk for NSAID-associated gastric ulcers. Curr Med Res Opin. 2011; 27:847–854.
Article52. Lee JH, Lee YC, Jeon SW, Kim JW, Lee SW. Korean College of Helicobacter and Upper Gastrointestinal Research. Korean Association of Gastroenterology. Guidelines of prevention and treatment for NSAID-related peptic ulcers. Korean J Gastroenterol. 2009; 54:309–317.
Article53. Rao P, Knaus EE. Evolution of nonsteroidal anti-inflammatory drugs (NSAIDs): cyclooxygenase (COX) inhibition and beyond. J Pharm Pharm Sci. 2008; 11:81s–110s.
Article54. Antman EM, Bennett JS, Daugherty A, Furberg C, Roberts H, Taubert KA. American Heart Association. Use of nonsteroidal antiinflammatory drugs: an update for clinicians: a scientific statement from the American Heart Association. Circulation. 2007; 115:1634–1642.55. Fitzgerald GA. Coxibs and cardiovascular disease. N Engl J Med. 2004; 351:1709–1711.
Article56. Kearney PM, Baigent C, Godwin J, Halls H, Emberson JR, Patrono C. Do selective cyclo-oxygenase-2 inhibitors and traditional non-steroidal anti-inflammatory drugs increase the risk of atherothrombosis? Meta-analysis of randomised trials. BMJ. 2006; 332:1302–1308.
Article57. Bombardier C, Laine L, Reicin A, et al. Comparison of upper gastrointestinal toxicity of rofecoxib and naproxen in patients with rheumatoid arthritis. VIGOR Study Group. N Engl J Med. 2000; 343:1520–1528.58. Bresalier RS, Sandler RS, Quan H, et al. Cardiovascular events associated with rofecoxib in a colorectal adenoma chemoprevention trial. N Engl J Med. 2005; 352:1092–1102.
Article59. U.S. Food and Drug Administration. Analysis and recommendations for agency action regarding nonsteroidal antiinflammatory drugs and cardiovascular risk. J Pain Palliat Care Pharmacother. 2005; 19:83–97.60. ADAPT Research Group. Meinert CL, McCaffrey LD, Breitner JC. Alzheimer's disease anti-inflammatory prevention trial: design, methods, and baseline results. Alzheimers Dement. 2009; 5:93–104.61. Bertagnolli MM, Eagle CJ, Zauber AG, et al. Adenoma Prevention with Celecoxib Study Investigators. Five-year efficacy and safety analysis of the Adenoma Prevention with Celecoxib Trial. Cancer Prev Res (Phila). 2009; 2:310–321.
Article62. Bertagnolli MM, Eagle CJ, Zauber AG, et al. APC Study Investigators. Celecoxib for the prevention of sporadic colorectal adenomas. N Engl J Med. 2006; 355:873–884.
Article63. Cannon CP, Curtis SP, FitzGerald GA, et al. MEDAL Steering Committee. Cardiovascular outcomes with etoricoxib and diclofenac in patients with osteoarthritis and rheumatoid arthritis in the Multinational Etoricoxib and Diclofenac Arthritis Long-term (MEDAL) programme: a randomised comparison. Lancet. 2006; 368:1771–1781.
Article64. White WB, West CR, Borer JS, et al. Risk of cardiovascular events in patients receiving celecoxib: a meta-analysis of randomized clinical trials. Am J Cardiol. 2007; 99:91–98.
Article65. Caldwell B, Aldington S, Weatherall M, Shirtcliffe P, Beasley R. Risk of cardiovascular events and celecoxib: a systematic review and meta-analysis. J R Soc Med. 2006; 99:132–140.
Article66. Shin S. Safety of celecoxib versus traditional nonsteroidal anti-inflammatory drugs in older patients with arthritis. J Pain Res. 2018; 11:3211–3219.
Article67. Nissen SE, Yeomans ND, Solomon DH, et al. PRECISION Trial Investigators. Cardiovascular safety of celecoxib, naproxen, or ibuprofen for arthritis. N Engl J Med. 2016; 375:2519–2529.
Article68. FitzGerald GA. Imprecision: limitations to interpretation of a large randomized clinical trial. Circulation. 2017; 135:113–115.
Article69. Solomon SD, Wittes J, Finn PV, et al. Cross Trial Safety Assessment Group. Cardiovascular risk of celecoxib in 6 randomized placebo-controlled trials: the cross trial safety analysis. Circulation. 2008; 117:2104–2113.70. Coxib and traditional NSAID Trialists' (CNT) Collaboration. Bhala N, Emberson J, Merhi A, et al. Vascular and upper gastrointestinal effects of non-steroidal anti-inflammatory drugs: meta-analyses of individual participant data from randomised trials. Lancet. 2013; 382:769–779.71. Capone ML, Tacconelli S, Sciulli MG, et al. Clinical pharmacology of platelet, monocyte, and vascular cyclooxygenase inhibition by naproxen and low-dose aspirin in healthy subjects. Circulation. 2004; 109:1468–1471.
Article72. McGettigan P, Henry D. Cardiovascular risk and inhibition of cyclooxygenase: a systematic review of the observational studies of selective and nonselective inhibitors of cyclooxygenase 2. JAMA. 2006; 296:1633–1644.73. Schmidt M, Sørensen HT, Pedersen L. Diclofenac use and cardiovascular risks: series of nationwide cohort studies. BMJ. 2018; 362:k3426.
Article74. Fleisher LA, Beckman JA, Brown KA, et al. American College of Cardiology/American Heart Association Task Force on Practice Guidelines (Writing Committee to Revise the 2002 Guidelines on Perioperative Cardiovascular Evaluation for Noncardiac Surgery). American Society of Echocardiography. American Society of Nuclear Cardiology. Heart Rhythm Society. Society of Cardiovascular Anesthesiologists. Society for Cardiovascular Angiography and Interventions. Society for Vascular Medicine and Biology. Society for Vascular Surgery. ACC/AHA 2007 guidelines on perioperative cardiovascular evaluation and care for noncardiac surgery: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines (Writing Committee to revise the 2002 guidelines on perioperative cardiovascular evaluation for noncardiac surgery): developed in collaboration with the American Society of Echocardiography, American Society of Nuclear Cardiology, Heart Rhythm Society, Society of Cardiovascular Anesthesiologists, Society for Cardiovascular Angiography and Interventions, Society for Vascular Medicine and Biology, and Society for Vascular Surgery. Circulation. 2007; 116:e418–e499.75. Patrignani P, Tacconelli S, Bruno A, Sostres C, Lanas A. Managing the adverse effects of nonsteroidal anti-inflammatory drugs. Expert Rev Clin Pharmacol. 2011; 4:605–621.
Article76. Bell AD, Roussin A, Cartier R, et al. The use of antiplatelet therapy in the outpatient setting: Canadian Cardiovascular Society guidelines executive summary. Can J Cardiol. 2011; 27:208–221.
Article77. Scarpignato C, Lanas A, Blandizzi C, Lems WF, Hermann M, Hunt RH. International NSAID Consensus Group. Safe prescribing of non-steroidal anti-inflammatory drugs in patients with osteoarthritis--an expert consensus addressing benefits as well as gastrointestinal and cardiovascular risks. BMC Med. 2015; 13:55.
Article78. European Medicines Agency (EMA). Assessment report for non-steroidal anti-inflammatory drugs (NSAIDs) and cardiovascular risk. London: EMA;2012. 10. EMA/696137/2012.79. Maity P, Bindu S, Choubey V, et al. Lansoprazole protects and heals gastric mucosa from non-steroidal anti-inflammatory drug (NSAID)-induced gastropathy by inhibiting mitochondrial as well as Fas-mediated death pathways with concurrent induction of mucosal cell renewal. J Biol Chem. 2008; 283:14391–14401.
Article80. Gooch K, Culleton BF, Manns BJ, et al. NSAID use and progression of chronic kidney disease. Am J Med. 2007; 120:280.e1–280.e7.
Article81. Hemmelgarn BR, Culleton BF, Ghali WA. Derivation and validation of a clinical index for prediction of rapid progression of kidney dysfunction. QJM. 2007; 100:87–92.
Article82. Yarger S, Nwokeji E, Trice S, et al. Puk4 cumulative exposure to nonsteroidal anti-inflammatory drugs (NSAIDS) and the progression of chronic kidney disease (CKD). Value Health. 2011; 14:A74–A75.
Article83. Akhund L, Quinet RJ, Ishaq S. Celecoxib-related renal papillary necrosis. Arch Intern Med. 2003; 163:114–115.
Article84. Rocha JL, Fernández-Alonso J. Acute tubulointerstitial nephritis associated with the selective COX-2 enzyme inhibitor, rofecoxib. Lancet. 2001; 357:1946–1947.
Article85. Höcherl K, Endemann D, Kammerl MC, Grobecker HF, Kurtz A. Cyclo-oxygenase-2 inhibition increases blood pressure in rats. Br J Pharmacol. 2002; 136:1117–1126.
Article86. Perazella MA, Tray K. Selective cyclooxygenase-2 inhibitors: a pattern of nephrotoxicity similar to traditional nonsteroidal anti-inflammatory drugs. Am J Med. 2001; 111:64–67.
Article87. Ungprasert P, Cheungpasitporn W, Crowson CS, Matteson EL. Individual non-steroidal anti-inflammatory drugs and risk of acute kidney injury: a systematic review and meta-analysis of observational studies. Eur J Intern Med. 2015; 26:285–291.
Article88. Nussmeier NA, Whelton AA, Brown MT, et al. Complications of the COX-2 inhibitors parecoxib and valdecoxib after cardiac surgery. N Engl J Med. 2005; 352:1081–1091.89. Zhang J, Ding EL, Song Y. Adverse effects of cyclooxygenase 2 inhibitors on renal and arrhythmia events: meta-analysis of randomized trials. JAMA. 2006; 296:1619–1632.90. Verrico MM, Weber RJ, McKaveney TP, Ansani NT, Towers AL. Adverse drug events involving COX-2 inhibitors. Ann Pharmacother. 2003; 37:1203–1213.
Article91. Hochberg MC, Altman RD, April KT, et al. American College of Rheumatology. American College of Rheumatology 2012 recommendations for the use of nonpharmacologic and pharmacologic therapies in osteoarthritis of the hand, hip, and knee. Arthritis Care Res (Hoboken). 2012; 64:465–474.
Article92. Sriuttha P, Sirichanchuen B, Permsuwan U. Hepatotoxicity of nonsteroidal anti-inflammatory drugs: a systematic review of randomized controlled trials. Int J Hepatol. 2018; 2018:5253623.
Article93. Riley TR 3rd, Smith JP. Ibuprofen-induced hepatotoxicity in patients with chronic hepatitis C: a case series. Am J Gastroenterol. 1998; 93:1563–1565.
Article94. Riley TR, Smith JP. Preventive care in chronic liver disease. J Gen Intern Med. 1999; 14:699–704.
Article95. Schink T, Kollhorst B, Varas Lorenzo C, et al. Risk of ischemic stroke and the use of individual non-steroidal anti-inflammatory drugs: a multi-country European database study within the SOS Project. PLoS One. 2018; 13:e0203362.
Article96. Nakhai-Pour HR, Broy P, Sheehy O, Bérard A. Use of nonaspirin nonsteroidal anti-inflammatory drugs during pregnancy and the risk of spontaneous abortion. CMAJ. 2011; 183:1713–1720.
Article97. Risser A, Donovan D, Heintzman J, Page T. NSAID prescribing precautions. Am Fam Physician. 2009; 80:1371–1378.98. Kauffman RE, Banner W, Berlin CM, et al. The transfer of drugs and other chemicals into human milk. Pediatrics. 1994; 93:137–150. https://pediatrics.aappublications.org/content/pediatrics/93/1/137.full.pdf.
Article99. Kowalski ML, Makowska JS, Blanca M, et al. Hypersensitivity to nonsteroidal anti-inflammatory drugs (NSAIDs) - classification, diagnosis and management: review of the EAACI/ENDA(#) and GA2LEN/HANNA*. Allergy. 2011; 66:818–829.
Article100. Jenkins C, Costello J, Hodge L. Systematic review of prevalence of aspirin induced asthma and its implications for clinical practice. BMJ. 2004; 328:434.
Article101. Kowalski ML, Asero R, Bavbek S, et al. Classification and practical approach to the diagnosis and management of hypersensitivity to nonsteroidal anti-inflammatory drugs. Allergy. 2013; 68:1219–1232.
Article102. Settipane RA, Schrank PJ, Simon RA, Mathison DA, Christiansen SC, Stevenson DD. Prevalence of cross-sensitivity with acetaminophen in aspirin-sensitive asthmatic subjects. J Allergy Clin Immunol. 1995; 96:480–485.
Article103. By the American Geriatrics Society 2015 Beers Criteria Update Expert Panel. American Geriatrics Society 2015 updated beers criteria for potentially inappropriate medication use in older adults. J Am Geriatr Soc. 2015; 63:2227–2246.104. Moore N, Pollack C, Butkerait P. Adverse drug reactions and drug-drug interactions with over-the-counter NSAIDs. Ther Clin Risk Manag. 2015; 11:1061–1075.105. Colebatch AN, Marks JL, van der, Edwards CJ. Safety of nonsteroidal antiinflammatory drugs and/or paracetamol in people receiving methotrexate for inflammatory arthritis: a Cochrane systematic review. J Rheumatol Suppl. 2012; 90:62–73.
Article106. Koffeman AR, Van Buul AR, Valkhoff VE, et al. Adverse drug reactions in a primary care population prescribed non-steroidal anti-inflammatory drugs. Scand J Prim Health Care. 2015; 33:163–169.
Article107. Avouac B, Combe B, Darné B. [Prescription of NSAIDs in patients treatment with platelet inhibitors or anticoagulants]. Presse Med. 2003; 32(37 Pt 2):S38–S43. French.108. Masclee GM, Valkhoff VE, Coloma PM, et al. Risk of upper gastrointestinal bleeding from different drug combinations. Gastroenterology. 2014; 147:784–792.e9.
Article109. Solomon DH, Goodson NJ. The cardiovascular system in rheumatic disease: the newest “extraarticular” manifestation? J Rheumatol. 2005; 32:1415–1417.110. Renda G, Tacconelli S, Capone ML, et al. Celecoxib, ibuprofen, and the antiplatelet effect of aspirin in patients with osteoarthritis and ischemic heart disease. Clin Pharmacol Ther. 2006; 80:264–274.
Article111. Bhatt DL, Scheiman J, Abraham NS, et al. American College of Cardiology Foundation Task Force on Clinical Expert Consensus Documents. ACCF/ACG/AHA 2008 expert consensus document on reducing the gastrointestinal risks of antiplatelet therapy and NSAID use: a report of the American College of Cardiology Foundation Task Force on Clinical Expert Consensus Documents. Circulation. 2008; 118:1894–1909.112. Anzellotti P, Capone ML, Jeyam A, et al. Low-dose naproxen interferes with the antiplatelet effects of aspirin in healthy subjects: recommendations to minimize the functional consequences. Arthritis Rheum. 2011; 63:850–859.
Article113. Mellemkjaer L, Blot WJ, Sørensen HT, et al. Upper gastrointestinal bleeding among users of NSAIDs: a population-based cohort study in Denmark. Br J Clin Pharmacol. 2002; 53:173–181.
Article114. Shorr RI, Ray WA, Daugherty JR, Griffin MR. Concurrent use of nonsteroidal anti-inflammatory drugs and oral anticoagulants places elderly persons at high risk for hemorrhagic peptic ulcer disease. Arch Intern Med. 1993; 153:1665–1670.
Article115. Douketis JD, Berger PB, Dunn AS, et al. The perioperative management of antithrombotic therapy: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines (8th Edition). Chest. 2008; 133:6 Suppl. 299S–339S.116. Carmichael JC, Keller DS, Baldini G, et al. Clinical practice guidelines for enhanced recovery after colon and rectal surgery from the American Society of Colon and Rectal Surgeons and Society of American Gastrointestinal and Endoscopic Surgeons. Dis Colon Rectum. 2017; 60:761–784.
Article117. National Institute for Health and Care Excellence (NICE). Gastro-oesophageal reflux disease and dyspepsia in adults: investigation and management. London: NICE;2014. p. 38.118. Card R, Sawyer M, Degnan B, et al. Perioperative protocol. Updated march 2014. Bloomington (MN): Institute for Clinical Systems Improvement;2014. p. 124.119. Jordan KM, Arden NK, Doherty M, et al. Standing Committee for International Clinical Studies Including Therapeutic Trials ESCISIT. EULAR recommendations 2003: an evidence based approach to the management of knee osteoarthritis: Report of a Task Force of the Standing Committee for International Clinical Studies Including Therapeutic Trials (ESCISIT). Ann Rheum Dis. 2003; 62:1145–1155.
Article120. Bradley JD, Brandt KD, Katz BP, Kalasinski LA, Ryan SI. Comparison of an antiinflammatory dose of ibuprofen, an analgesic dose of ibuprofen, and acetaminophen in the treatment of patients with osteoarthritis of the knee. N Engl J Med. 1991; 325:87–91.
Article121. Williams HJ, Ward JR, Egger MJ, et al. Comparison of naproxen and acetaminophen in a two-year study of treatment of osteoarthritis of the knee. Arthritis Rheum. 1993; 36:1196–1206.122. Oxford analgesic league table [Internet]. London: The Royal College of Anaesthetists;2019. cited 2019 Jun 5. Available from: https://www.rcoa.ac.uk/faculty-of-pain-medicine/opioids-aware/oxford-league-table.123. Graham GG, Davies MJ, Day RO, Mohamudally A, Scott KF. The modern pharmacology of paracetamol: therapeutic actions, mechanism of action, metabolism, toxicity and recent pharmacological findings. Inflammopharmacology. 2013; 21:201–232.
Article124. Solomon DH, Rassen JA, Glynn RJ, et al. The comparative safety of opioids for nonmalignant pain in older adults. Arch Intern Med. 2010; 170:1979–1986.
Article125. McPherson ML, Cimino NM. Topical NSAID formulations. Pain Med. 2013; 14 Suppl 1:S35–S39.
Article126. Altman RD, Barthel HR. Topical therapies for osteoarthritis. Drugs. 2011; 71:1259–1279.
Article127. Jorge LL, Feres CC, Teles VE. Topical preparations for pain relief: efficacy and patient adherence. J Pain Res. 2010; 4:11–24.128. Yates AJ Jr, McGrory BJ, Starz TW, Vincent KR, McCardel B, Golightly YM. AAOS appropriate use criteria: optimizing the non-arthroplasty management of osteoarthritis of the knee. J Am Acad Orthop Surg. 2014; 22:261–267.129. Klinge SA, Sawyer GA. Effectiveness and safety of topical versus oral nonsteroidal anti-inflammatory drugs: a comprehensive review. Phys Sportsmed. 2013; 41:64–74.
Article130. Brühlmann P, de Vathaire F, Dreiser RL, Michel BA. Short-term treatment with topical diclofenac epolamine plaster in patients with symptomatic knee osteoarthritis: pooled analysis of two randomised clinical studies. Curr Med Res Opin. 2006; 22:2429–2438.
Article131. Barthel HR, Haselwood D, Longley S 3rd, Gold MS, Altman RD. Randomized controlled trial of diclofenac sodium gel in knee osteoarthritis. Semin Arthritis Rheum. 2009; 39:203–212.
Article132. Derry S, Conaghan P, Da Silva JA, Wiffen PJ, Moore RA. Topical NSAIDs for chronic musculoskeletal pain in adults. Cochrane Database Syst Rev. 2016; 4:CD007400.
Article133. Scarpignato C, Gatta L, Zullo A, Blandizzi C. SIF-AIGO-FIMMG Group. Italian Society of Pharmacology, the Italian Association of Hospital Gastroenterologists, and the Italian Federation of General Practitioners. Effective and safe proton pump inhibitor therapy in acid-related diseases - a position paper addressing benefits and potential harms of acid suppression. BMC Med. 2016; 14:179.
Article134. Freedberg DE, Kim LS, Yang YX. The risks and benefits of long-term use of proton pump inhibitors: expert review and best practice advice from the American Gastroenterological Association. Gastroenterology. 2017; 152:706–715.
Article135. Park HR, Cho SK, Im SG, et al. Treatment patterns of knee osteoarthritis patients in Korea. Korean J Intern Med. 2019; 34:1145–1153.
Article136. Lee SH, Han CD, Yang IH, Ha CW. Prescription pattern of NSAIDs and the prevalence of NSAID-induced gastrointestinal risk factors of orthopaedic patients in clinical practice in Korea. J Korean Med Sci. 2011; 26:561–567.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Updates of Spondyloarthrothy Treatment
- Diagnosis and treatment of Helicobacter pylori infection in patients treated with non-steroidal anti-inflammatory drugs
- Efficacy of disease-modifying osteoarthritis drugs in the treatment of osteoarthritis
- Multiple Gastrointestinal Drug Induced Ulcers Associated with Aspirin and Non-steroidal Anti-inflammatory Drugs: A Case Report and Review of the Literature
- Chemoprevention of Gastric Cancer: Non-steroidal Anti-inflammatory Drugs Including Aspirin