Korean J Radiol.
2020 Mar;21(3):332-340. 10.3348/kjr.2019.0053.
Thyroid-Associated Orbitopathy: Evaluating Microstructural Changes of Extraocular Muscles and Optic Nerves Using Readout-Segmented Echo-Planar Imaging-Based Diffusion Tensor Imaging
- Affiliations
-
- 1Department of Endocrinology, The First Affiliated Hospital of Nanjing Medical University, Nanjing, China. njmu_yt@sina.com
- 2Department of Radiology, The First Affiliated Hospital of Nanjing Medical University, Nanjing, China.
- KMID: 2470757
- DOI: http://doi.org/10.3348/kjr.2019.0053
Abstract
OBJECTIVE
We aimed to investigate the ability of readout-segmented echo-planar imaging (rs-EPI)-based diffusion tensor imaging (DTI) in assessing the microstructural change of extraocular muscles (EOMs) and optic nerves in patients with thyroid-associated orbitopathy (TAO) as well as in evaluating disease activity.
MATERIALS AND METHODS
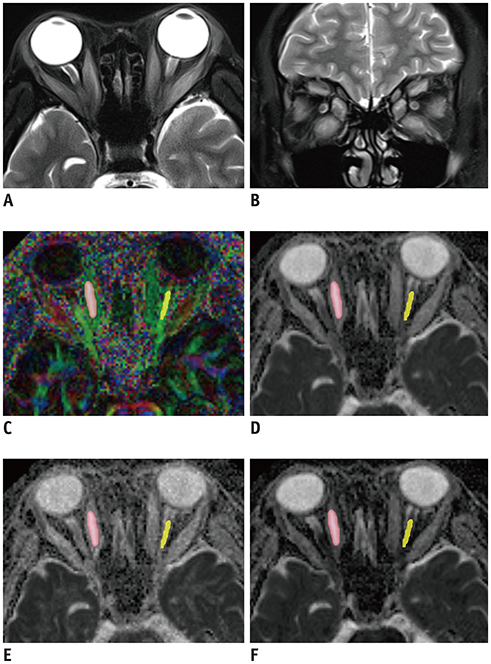
We enrolled 35 TAO patients and 22 healthy controls (HCs) who underwent pre-treatment rs-EPI-based DTI. Mean, axial, and radial diffusivity (MD, AD, and RD) and fractional anisotropy (FA) of the medial and lateral EOMs and optic nerve for each orbit were calculated and compared between TAO and HC groups and between active and inactive TAO groups. Factors such as age, sex, disease duration, mediation, and smoking history between groups were also compared. Logistic regression analysis was used to evaluate the predictive value of significant variables for disease activity.
RESULTS
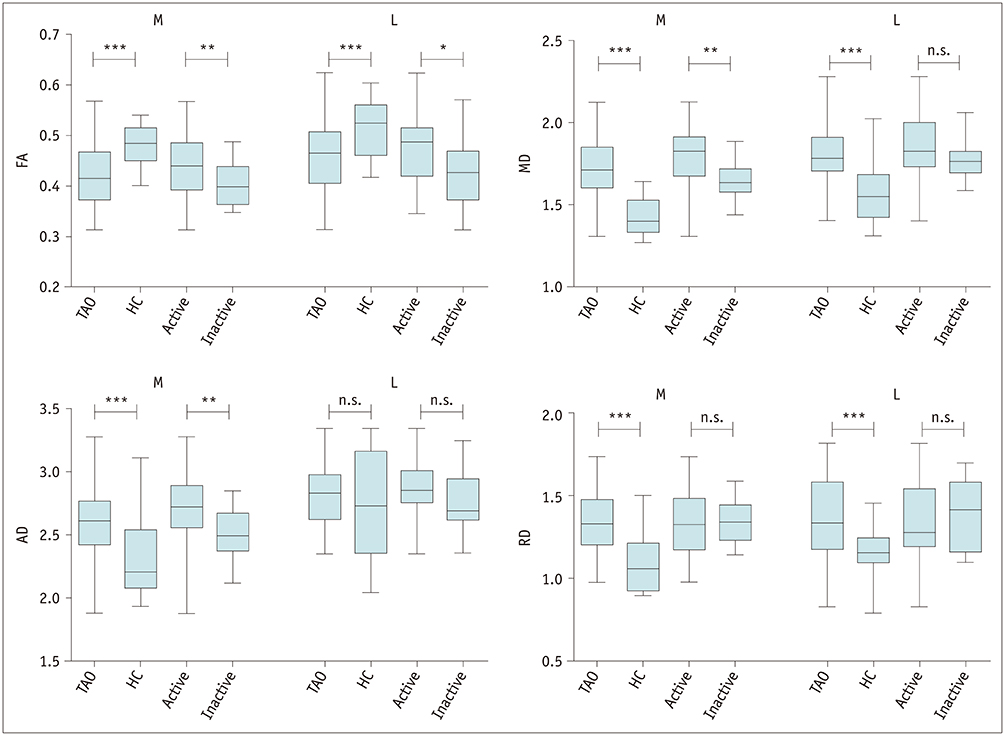
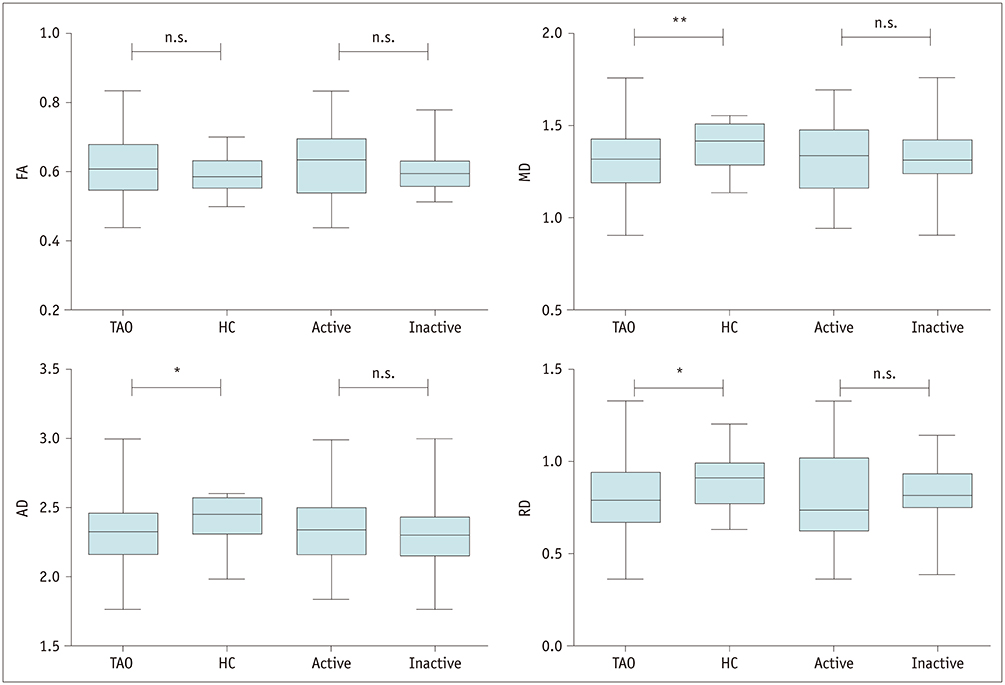
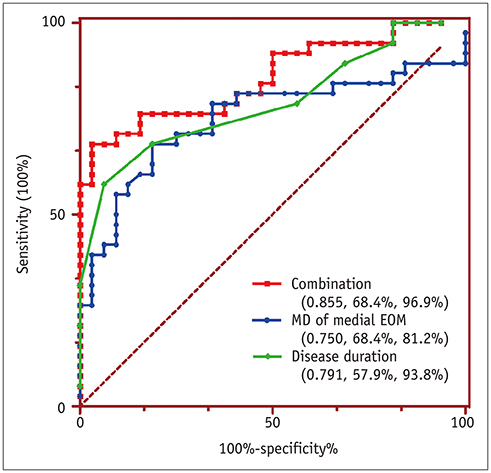
Disease duration was significantly shorter in active TAOs than in inactive ones (p < 0.001). TAO patients showed significantly lower FA and higher MD, AD, and RD than HCs for both medial and lateral EOMs (p < 0.001), but not the AD value of lateral EOMs (p = 0.619). Active patients had significantly higher FA, MD, and AD than inactive patients for medial EOMs (p < 0.005), whereas only FA differed significantly in the lateral EOMs (p = 0.018). The MD, AD, and RD of optic nerves were significantly lower in TAO patients than HCs (p < 0.05), except for FA (p = 0.129). Multivariate analysis showed that the MD of medial EOMs and disease duration were significant predictors for disease activity. The combination of these two parameters showed optimal diagnostic efficiency for disease activity (area under the curve, 0.855; sensitivity, 68.4%; specificity, 96.9%).
CONCLUSION
rs-EPI-based DTI is promising in assessing microstructural changes of EOMs and optic nerves and can help to indicate the disease activity of TAO, especially through the MD of medial EOMs.
Keyword
MeSH Terms
Figure
Reference
-
1. Ludgate M, Baker G. Unlocking the immunological mechanisms of orbital inflammation in thyroid eye disease. Clin Exp Immunol. 2002; 127:193–198.
Article2. Carlé A, Pedersen IB, Knudsen N, Perrild H, Ovesen L, Rasmussen LB, et al. Epidemiology of subtypes of hyperthyroidism in Denmark: a population-based study. Eur J Endocrinol. 2011; 164:801–809.
Article3. Bartalena L, Pinchera A, Marcocci C. Management of Graves' ophthalmopathy: reality and perspectives. Endocr Rev. 2000; 21:168–199.
Article4. Blandford AD, Zhang D, Chundury RV, Perry JD. Dysthyroid optic neuropathy: update on pathogenesis, diagnosis, and management. Expert Rev Ophthalmol. 2017; 12:111–121.
Article5. Gonçalves AC, Gebrim EM, Monteiro ML. Imaging studies for diagnosing Graves' orbitopathy and dysthyroid optic neuropathy. Clinics (Sao Paulo). 2012; 67:1327–1334.
Article6. Rodríguez-González N, Pérez-Rico C, López-Para Giménez R, Arévalo-Serrano J, Del Amo García B, Calzada Domingo L, et al. [Short-tau inversion-recovery (STIR) sequence magnetic resonance imaging evaluation of orbital structures in Graves' orbitopathy]. Arch Soc Esp Oftalmol. 2011; 86:351–357.
Article7. Szucs-Farkas Z, Toth J, Balazs E, Galuska L, Burman KD, Karanyi Z, et al. Using morphologic parameters of extraocular muscles for diagnosis and follow-up of Graves' ophthalmopathy: diameters, areas, or volumes? AJR Am J Roentgenol. 2002; 179:1005–1010.
Article8. Kirsch EC, Kaim AH, De Oliveira MG, von Arx G. Correlation of signal intensity ratio on orbital MRI-TIRM and clinical activity score as a possible predictor of therapy response in Graves' orbitopathy—a pilot study at 1.5 T. Neuroradiology. 2010; 52:91–97.9. Hu H, Xu XQ, Wu FY, Chen HH, Su GY, Shen J, et al. Diagnosis and stage of Graves' ophthalmopathy: efficacy of quantitative measurements of the lacrimal gland based on 3-T magnetic resonance imaging. Exp Ther Med. 2016; 12:725–729.
Article10. Min J, Park M, Choi JW, Jahng GH, Moon WJ. Inter-vendor and inter-session reliability of diffusion tensor imaging: implications for multicenter clinical imaging studies. Korean J Radiol. 2018; 19:777–782.
Article11. Wang MY, Wu K, Xu JM, Dai J, Qin W, Liu J, et al. Quantitative 3-T diffusion tensor imaging in detecting optic nerve degeneration in patients with glaucoma: association with retinal nerve fiber layer thickness and clinical severity. Neuroradiology. 2013; 55:493–498.
Article12. Wang MY, Qi PH, Shi DP. Diffusion tensor imaging of the optic nerve in subacute anterior ischemic optic neuropathy at 3T. AJNR Am J Neuroradiol. 2011; 32:1188–1194.
Article13. van der Walt A, Kolbe SC, Wang YE, Klistorner A, Shuey N, Ahmadi G, et al. Optic nerve diffusion tensor imaging after acute optic neuritis predicts axonal and visual outcomes. PLoS One. 2013; 8:e83825.
Article14. Han JS, Seo HS, Lee YH, Lee H, Suh SI, Jeong EK, et al. Fractional anisotropy and diffusivity changes in thyroid-associated orbitopathy. Neuroradiology. 2016; 58:1189–1196.
Article15. Lee H, Lee YH, Suh SI, Jeong EK, Baek S, Seo HS. Characterizing intraorbital optic nerve changes on diffusion tensor imaging in thyroid eye disease before dysthyroid optic neuropathy. J Comput Assist Tomogr. 2018; 42:293–298.
Article16. Kim YJ, Kim SH, Kang BJ, Park CS, Kim HS, Son YH, et al. Readout-segmented echo-planar imaging in diffusion-weighted MR imaging in breast cancer: comparison with single-shot echo-planar imaging in image quality. Korean J Radiol. 2014; 15:403–410.
Article17. Porter D, Mueller E. Multi-shot diffusion-weighted EPI with readout mosaic segmentation and 2D navigator correction. In : International Society for Magnetic Resonance in Medicine twelfth scientific meeting and exhibition; 2004 May 15–21; Kyoto, Japan.18. Xu XQ, Liu J, Hu H, Su GY, Zhang YD, Shi HB, et al. Improve the image quality of orbital 3 T diffusion-weighted magnetic resonance imaging with readout-segmented echo-planar imaging. Clin Imaging. 2016; 40:793–796.
Article19. Bartley GB, Gorman CA. Diagnostic criteria for Graves' ophthalmopathy. Am J Ophthalmol. 1995; 119:792–795.
Article20. Barrio-Barrio J, Sabater AL, Bonet-Farriol E, Velázquez-Villoria Á, Galofré JC. Graves' ophthalmopathy: VISA versus EUGOGO classification, assessment, and management. J Ophthalmol. 2015; 2015:249125.
Article21. Politi LS, Godi C, Cammarata G, Ambrosi A, Iadanza A, Lanzi R, et al. Magnetic resonance imaging with diffusion-weighted imaging in the evaluation of thyroid-associated orbitopathy: getting below the tip of the iceberg. Eur Radiol. 2014; 24:1118–1126.
Article22. Kilicarslan R, Alkan A, Ilhan MM, Yetis H, Aralasmak A, Tasan E. Graves' ophthalmopathy: the role of diffusion-weighted imaging in detecting involvement of extraocular muscles in early period of disease. Br J Radiol. 2015; 88:20140677.
Article23. Sotak CH. Nuclear magnetic resonance (NMR) measurement of the apparent diffusion coefficient (ADC) of tissue water and its relationship to cell volume changes in pathological states. Neurochem Int. 2004; 45:569–582.
Article24. Bryant ND, Li K, Does MD, Barnes S, Gochberg DF, Yankeelov TE, et al. Multi-parametric MRI characterization of inflammation in murine skeletal muscle. NMR Biomed. 2014; 27:716–725.
Article25. Cheung JS, Fan SJ, Gao DS, Chow AM, Man K, Wu EX. Diffusion tensor imaging of liver fibrosis in an experimental model. J Magn Reson Imaging. 2010; 32:1141–1148.
Article26. Heemskerk AM, Strijkers GJ, Vilanova A, Drost MR, Nicolay K. Determination of mouse skeletal muscle architecture using three-dimensional diffusion tensor imaging. Magn Reson Med. 2005; 53:1333–1340.
Article27. Seo HS, Kim SE, Rose J, Hadley JR, Parker DL, Jeong EK. Diffusion tensor imaging of extraocular muscle using two-dimensional single-shot interleaved multiple inner volume imaging diffusion-weighted EPI at 3 Tesla. J Magn Reson Imaging. 2013; 38:1162–1168.
Article28. Abdullah OM, Drakos SG, Diakos NA, Wever-Pinzon O, Kfoury AG, Stehlik J, et al. Characterization of diffuse fibrosis in the failing human heart via diffusion tensor imaging and quantitative histological validation. NMR Biomed. 2014; 27:1378–1386.
Article29. Yamada H, Yamamoto A, Okada T, Kanagaki M, Fushimi Y, Porter DA, et al. Diffusion tensor imaging of the optic chiasm in patients with intra- or parasellar tumor using readout-segmented echo-planar. Magn Reson Imaging. 2016; 34:654–661.
Article30. Santarelli X, Garbin G, Ukmar M, Longo R. Dependence of the fractional anisotropy in cervical spine from the number of diffusion gradients, repeated acquisition and voxel size. Magn Reson Imaging. 2010; 28:70–76.
Article31. Oouchi H, Yamada K, Sakai K, Kizu O, Kubota T, Ito H, et al. Diffusion anisotropy measurement of brain white matter is affected by voxel size: underestimation occurs in areas with crossing fibers. AJNR Am J Neuroradiol. 2007; 28:1102–1106.
Article32. Wu CJ, Wang Q, Li H, Wang XN, Liu XS, Shi HB, et al. DWI-associated entire-tumor histogram analysis for the differentiation of low-grade prostate cancer from intermediate-high-grade prostate cancer. Abdom Imaging. 2015; 40:3214–3221.
Article33. Özkan B, Anik Y, Katre B, Altintas¸ Ö, Gençtürk M, Yüksel N. Quantitative assessment of optic nerve with diffusion tensor imaging in patients with thyroid orbitopathy. Ophthalmic Plast Reconstr Surg. 2015; 31:391–395.
Article34. Lingam RK, Mundada P, Lee V. Novel use of non-echo-planar diffusion weighted MRI in monitoring disease activity and treatment response in active Grave's orbitopathy: an initial observational cohort study. Orbit. 2018; 37:325–330.
Article35. Paul K, Graessl A, Rieger J, Lysiak D, Huelnhagen T, Winter L, et al. Diffusion-sensitized ophthalmic magnetic resonance imaging free of geometric distortion at 3.0 and 7.0 T: a feasibility study in healthy subjects and patients with intraocular masses. Invest Radiol. 2015; 50:309–321.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Multi-slice Multi-echo Pulsed-gradient Spin-echo (MePGSE) Sequence for Diffusion Tensor Imaging MRI: A Preliminary Result
- Utility of Readout-Segmented Echo-Planar Imaging-Based Diffusion Kurtosis Imaging for Differentiating Malignant from Benign Masses in Head and Neck Region
- Bilateral Extraocular Muscle Metastasis of Nasal Rhabdomyosarcoma Mimicking a Thyroid Associated Orbitopathy: A Case Report
- Diffusion Tensor Tractography of the Brainstem Pyramidal Tract: A Study on the Optimal Reduction Factor in Parallel Imaging
- Analysis of Apparent Diffusion Coefficients of the Brain in Healthy Controls: A Comparison Study between Single-Shot Echo-Planar Imaging and Read-out-Segmented Echo-Planar Imaging