Int J Stem Cells.
2019 Jul;12(2):331-339. 10.15283/ijsc18133.
Mesenchymal Stem Cell Transplantation Promotes Functional Recovery through MMP2/STAT3 Related Astrogliosis after Spinal Cord Injury
- Affiliations
-
- 1Department of Neurosurgery, Kangwon National University School of Medicine, Chuncheon, Korea.
- 2Divisions of Applied Animal Science and Animal Resource Science, Department of Animal Life Science, Kangwon National University, Chuncheon, Korea.
- 3Department of Thoracic and Cardiovascular Surgery, Kangwon National University School of Medicine, Chuncheon, Korea. seran.yang@gmail.com
- 4Department of Neurosurgery, Seoul National University College of Medicine, Seoul, Korea. chungc@snu.ac.kr
- KMID: 2465902
- DOI: http://doi.org/10.15283/ijsc18133
Abstract
- BACKGROUND AND OBJECTIVES
Treatment with mesenchymal stem cells (MSC) in spinal cord injury (SCI) has been highlighted as therapeutic candidate for SCI. Although astrogliosis is a major phenomenon after SCI, the role of astrogliosis is still controversial. In this study, we determined whether acute transplantation of MSC improves the outcome of SCI through modulating astrogliosis.
METHODS
Bone marrow derived rat MSCs were induced neural differentiation and transplanted after acute SCI rats. Matrix metalloproteinase (MMP) and neuro-inflammatory pathway were analyzed for acute astrogliosis at 1, 3 and 7 d after SCI in RT-PCR- and western blot analysis. Functional outcome was assessed serially at postoperative 1 d and weekly for 4 weeks. Histopathologic analysis was undertaken at 7 and 28 d following injury in immunohistochemistry.
RESULTS
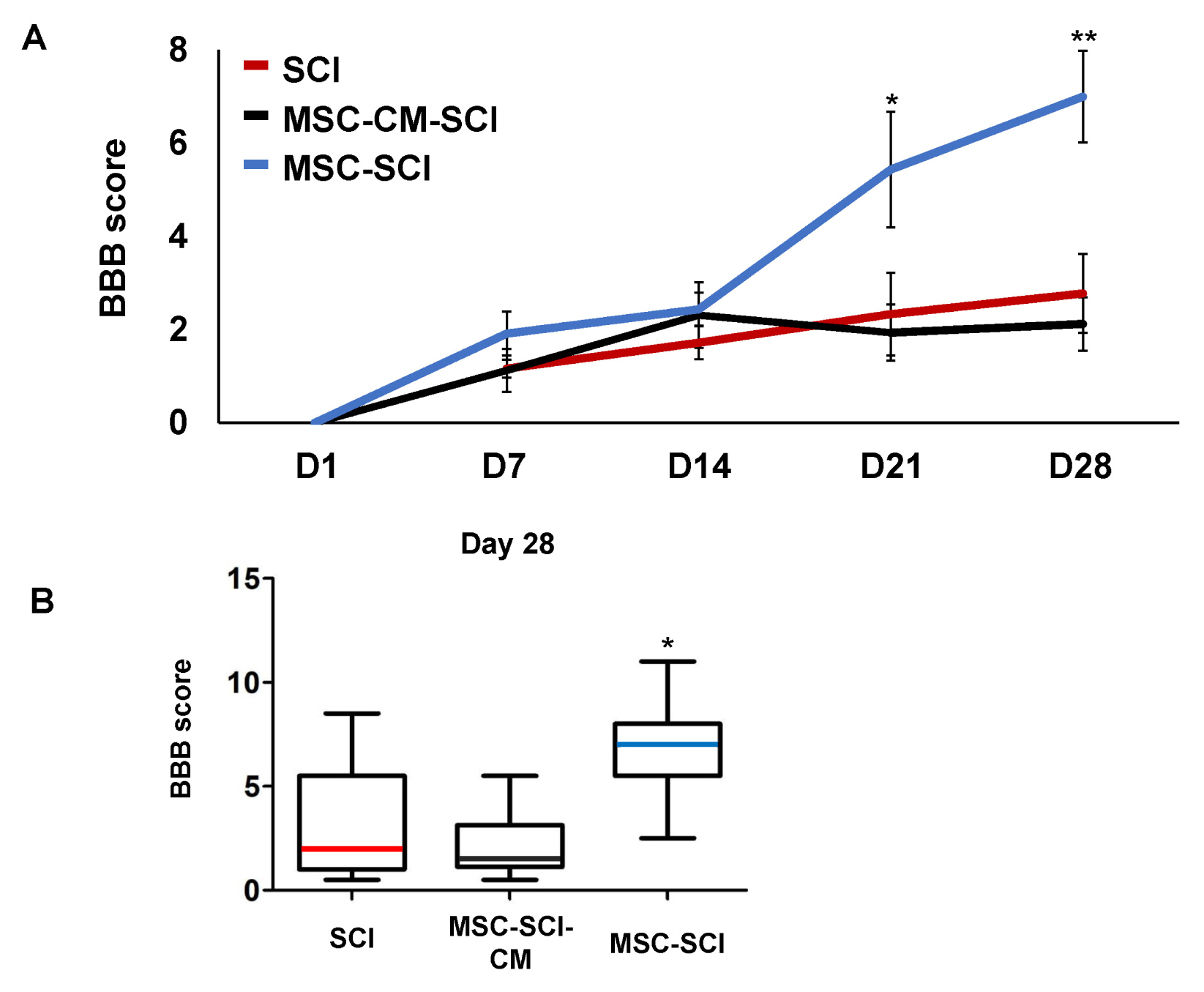
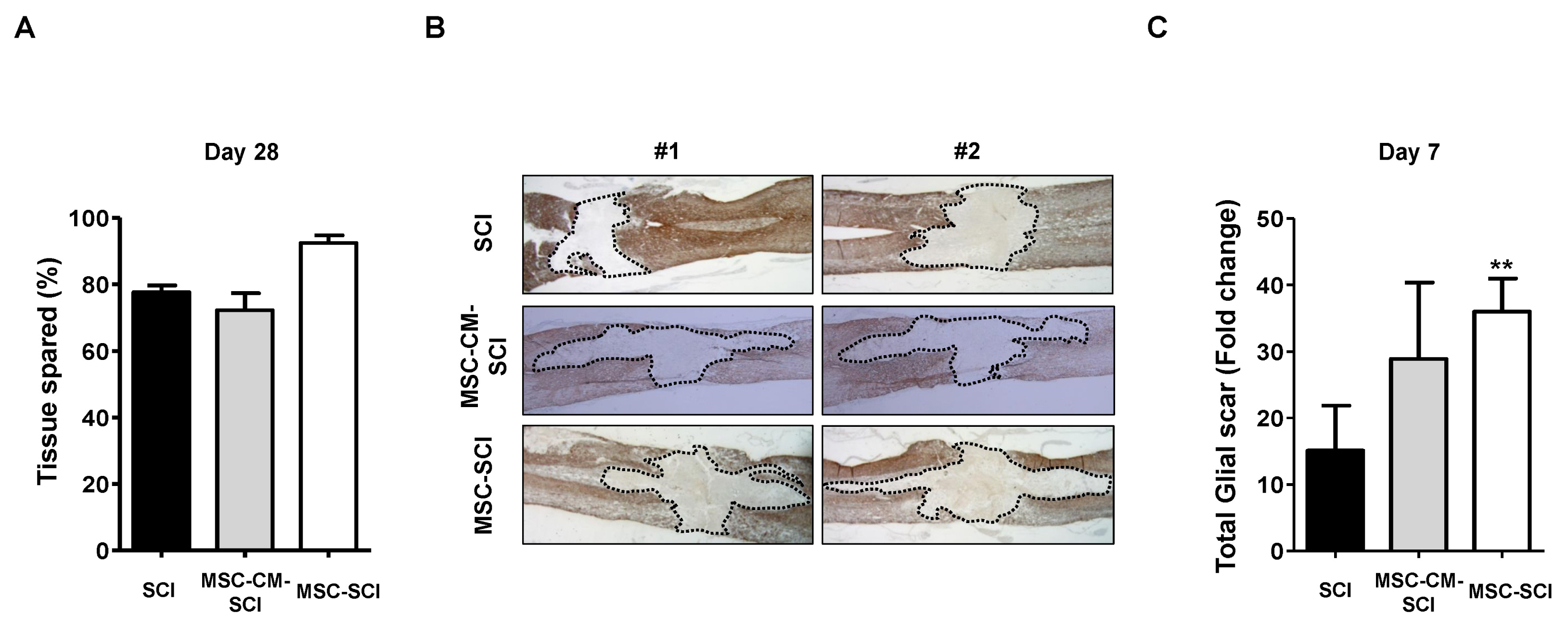
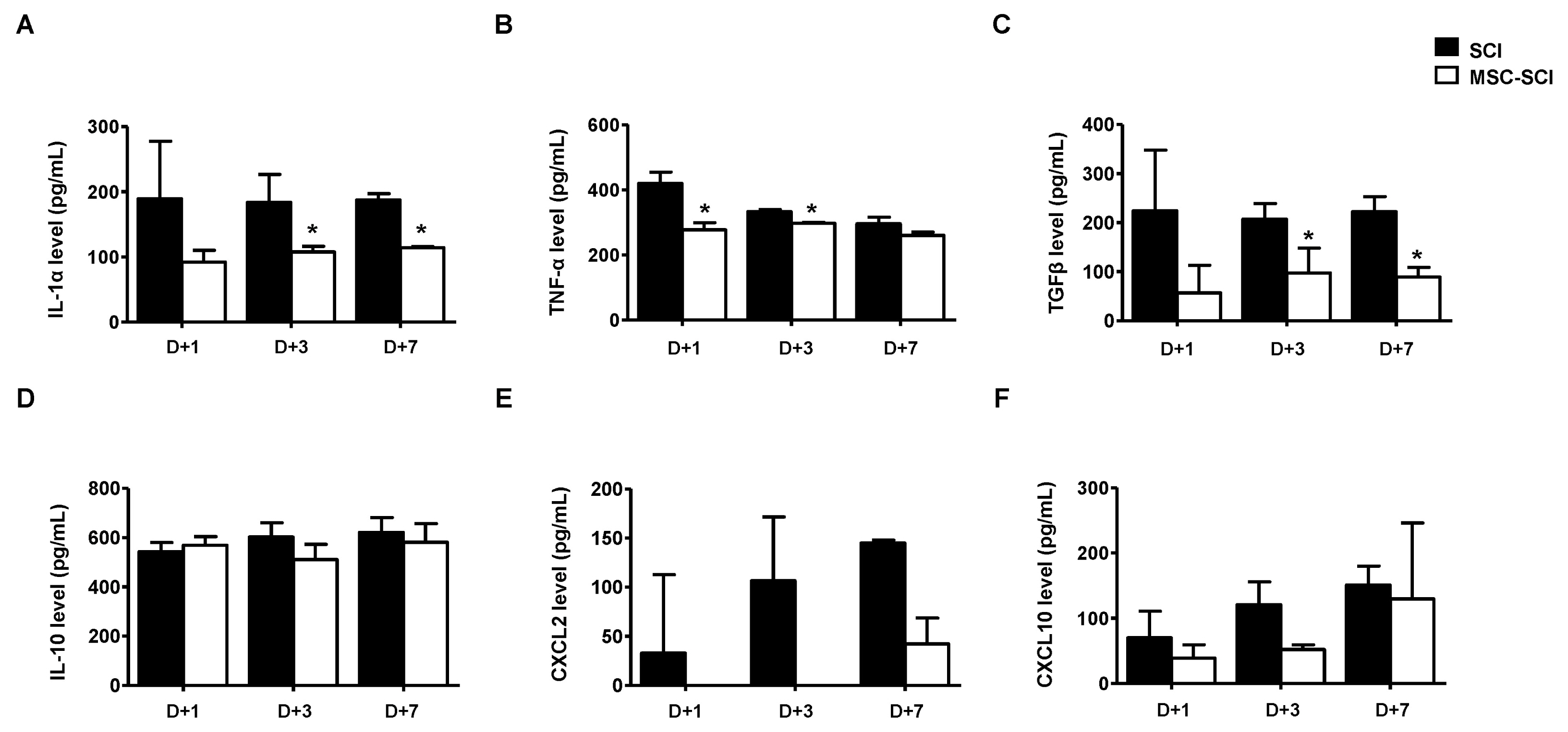
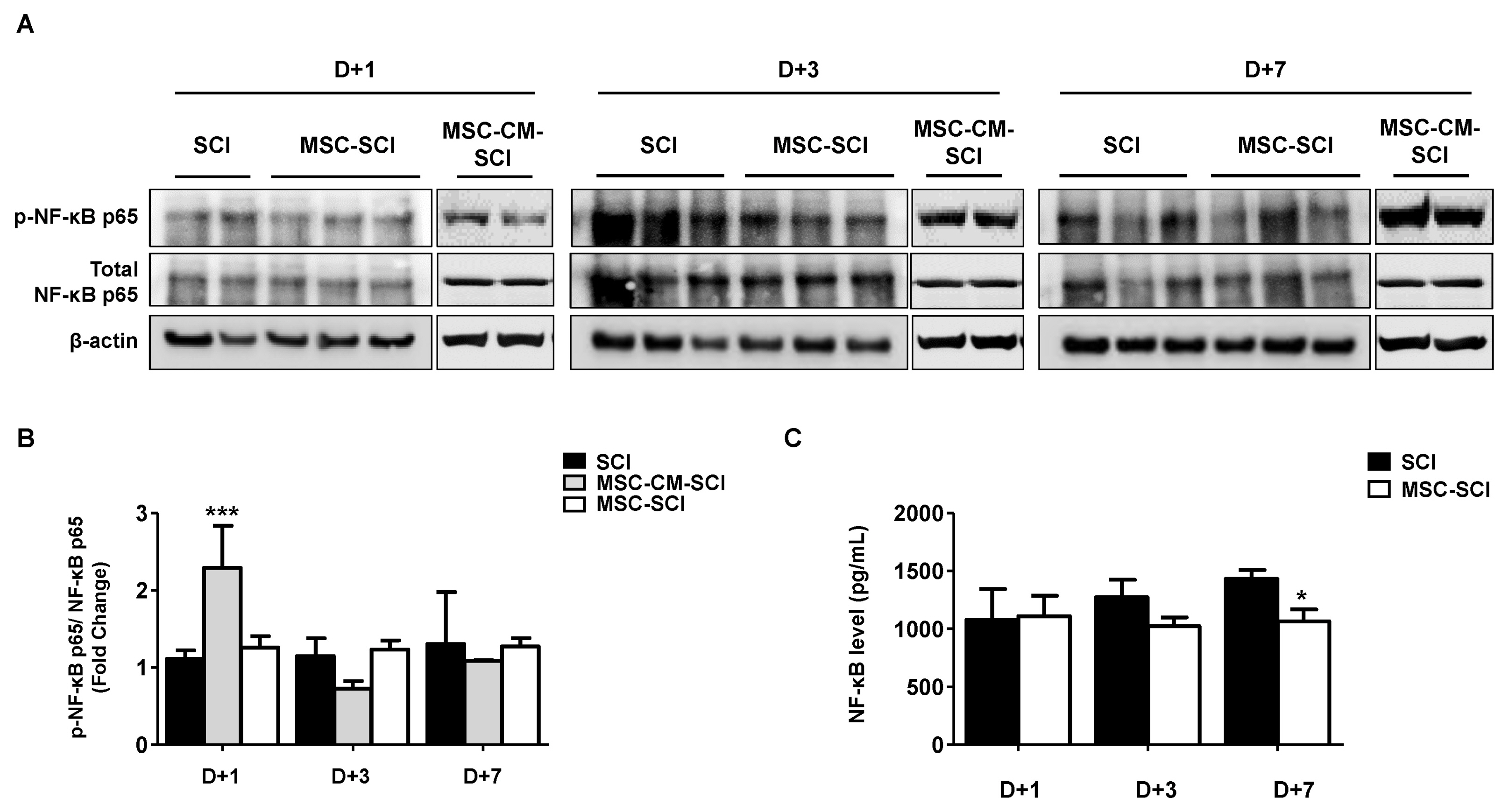
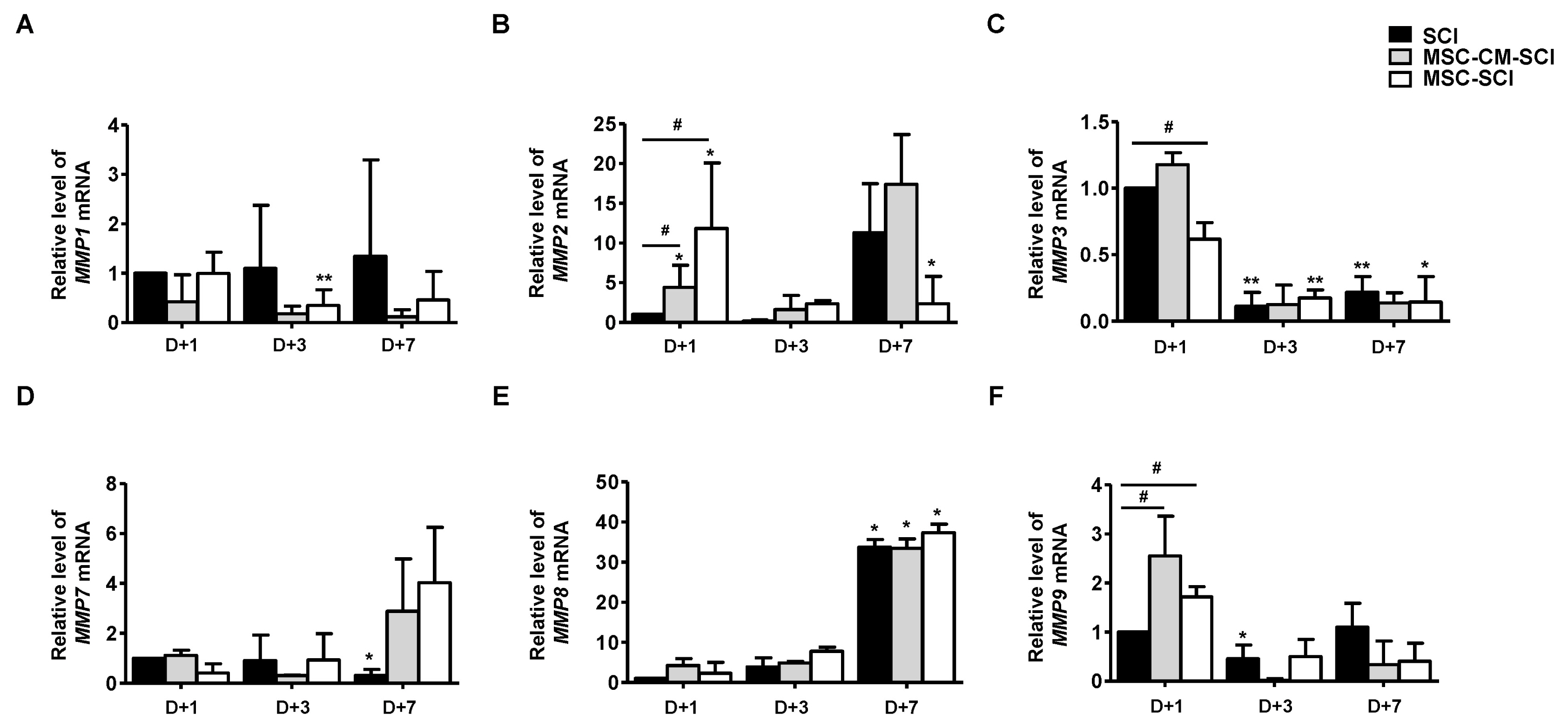
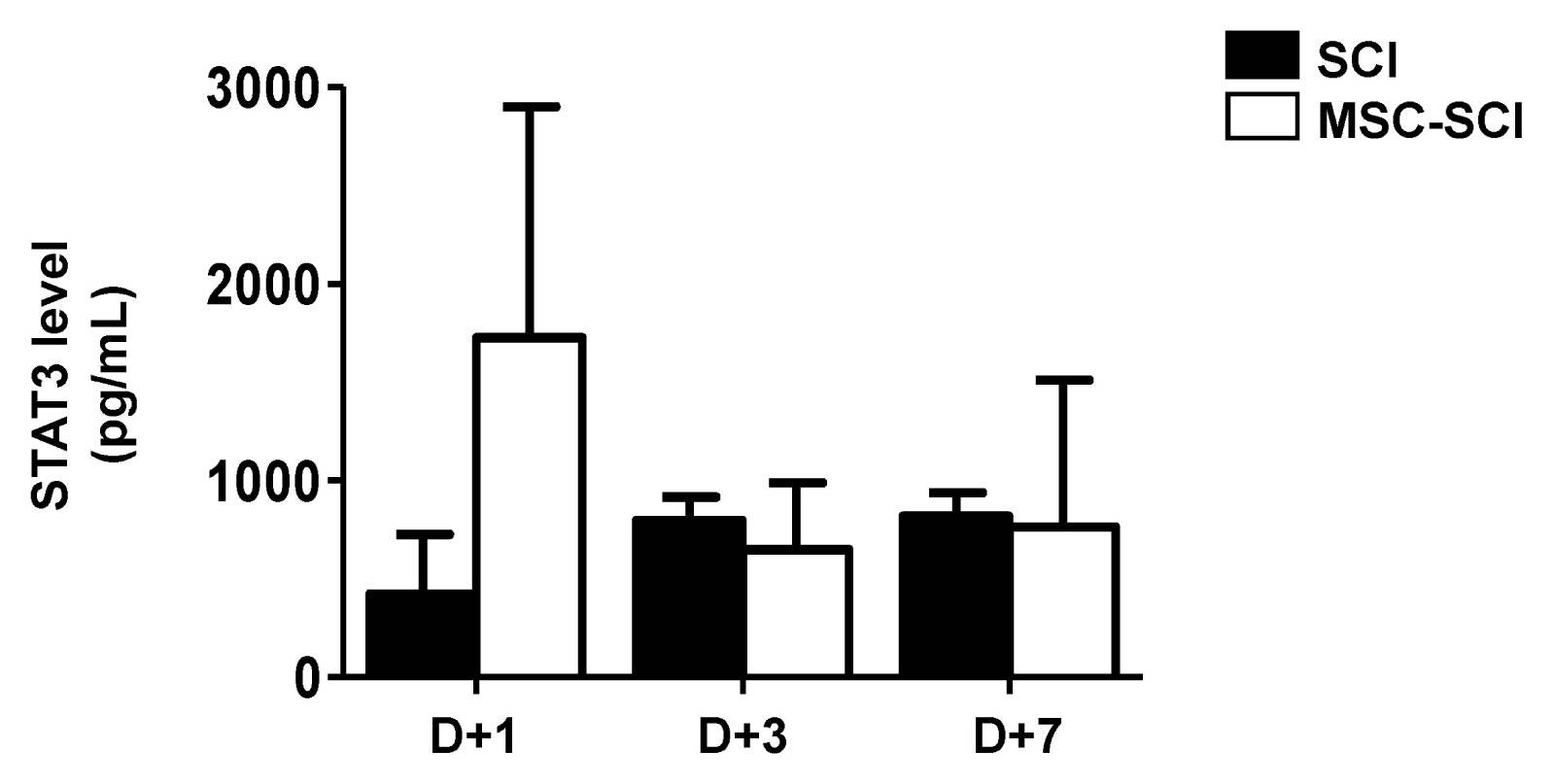
Transplantation of MSCs decreased IL-1α, CXCL-2, CXCL-10, TNF-α and TGF-β in a rat model of contusive SCI. Protein level of NF-κB p65 was slightly decreased while level of STAT-3 was increased. In immunohistochemistry, MSC transplantation increased acute astrogliosis whereas attenuated scar formation with increased sparing white matter of spinal cord lesions. In RT-PCR analysis, mRNA levels of MMP2 was significantly increased in MSC transplanted rats. In BBB locomotor scale, the rats of MSC treated group exhibited improvement of functional recovery.
CONCLUSIONS
Transplantation of MSC reduces the inflammatory reaction and modulates astrogliosis via MMP2/STAT3 pathway leading to improve functional recovery after SCI in rats.
Keyword
MeSH Terms
Figure
Reference
-
References
1. Dewan MC, Rattani A, Gupta S, Baticulon RE, Hung YC, Punchak M, Agrawal A, Adeleye AO, Shrime MG, Rubiano AM, Rosenfeld JV, Park KB. Estimating the global incidence of traumatic brain injury. J Neurosurg. 2018; [Epub ahead of print]. DOI: 10.3171/2017.10.JNS17352. PMID: 29701556.
Article2. Wyndaele M, Wyndaele JJ. Incidence, prevalence and epidemiology of spinal cord injury: what learns a worldwide literature survey? Spinal Cord. 2006; 44:523–529. DOI: 10.1038/sj.sc.3101893. PMID: 16389270.
Article3. Rossignol S, Schwab M, Schwartz M, Fehlings MG. Spinal cord injury: time to move? J Neurosci. 2007; 27:11782–11792. DOI: 10.1523/JNEUROSCI.3444-07.2007. PMID: 17978014.
Article4. Sofroniew MV. Molecular dissection of reactive astrogliosis and glial scar formation. Trends Neurosci. 2009; 32:638–647. DOI: 10.1016/j.tins.2009.08.002. PMID: 19782411. PMCID: PMC2787735.
Article5. Zhang H, Adwanikar H, Werb Z, Noble-Haeusslein LJ. Matrix metalloproteinases and neurotrauma: evolving roles in injury and reparative processes. Neuroscientist. 2010; 16:156–170. DOI: 10.1177/1073858409355830. PMID: 20400713. PMCID: PMC2858362.
Article6. Zhang H, Chang M, Hansen CN, Basso DM, Noble-Haeusslein LJ. Role of matrix metalloproteinases and therapeutic benefits of their inhibition in spinal cord injury. Neurotherapeutics. 2011; 8:206–220. DOI: 10.1007/s13311-011-0038-0. PMID: 21455784. PMCID: PMC3077748.
Article7. Hofstetter CP, Schwarz EJ, Hess D, Widenfalk J, El Manira A, Prockop DJ, Olson L. Marrow stromal cells form guiding strands in the injured spinal cord and promote recovery. Proc Natl Acad Sci U S A. 2002; 99:2199–2204. DOI: 10.1073/pnas.042678299. PMID: 11854516. PMCID: PMC122342.
Article8. Parr AM, Kulbatski I, Wang XH, Keating A, Tator CH. Fate of transplanted adult neural stem/progenitor cells and bone marrow-derived mesenchymal stromal cells in the injured adult rat spinal cord and impact on functional recovery. Surg Neurol. 2008; 70:600–607. discussion 607. DOI: 10.1016/j.surneu.2007.09.043. PMID: 18291482.
Article9. Anderson MA, Burda JE, Ren Y, Ao Y, O’Shea TM, Kawaguchi R, Coppola G, Khakh BS, Deming TJ, Sofroniew MV. Astrocyte scar formation aids central nervous system axon regeneration. Nature. 2016; 532:195–200. DOI: 10.1038/nature17623. PMID: 27027288. PMCID: PMC5243141.
Article10. Faulkner JR, Herrmann JE, Woo MJ, Tansey KE, Doan NB, Sofroniew MV. Reactive astrocytes protect tissue and preserve function after spinal cord injury. J Neurosci. 2004; 24:2143–2155. DOI: 10.1523/JNEUROSCI.3547-03.2004. PMID: 14999065.
Article11. Okada S, Hara M, Kobayakawa K, Matsumoto Y, Nakashima Y. Astrocyte reactivity and astrogliosis after spinal cord injury. Neurosci Res. 2018; 126:39–43. DOI: 10.1016/j.neures.2017.10.004. PMID: 29054466.
Article12. Wanner IB, Anderson MA, Song B, Levine J, Fernandez A, Gray-Thompson Z, Ao Y, Sofroniew MV. Glial scar borders are formed by newly proliferated, elongated astrocytes that interact to corral inflammatory and fibrotic cells via STAT3-dependent mechanisms after spinal cord injury. J Neurosci. 2013; 33:12870–12886. DOI: 10.1523/JNEUROSCI.2121-13.2013. PMID: 23904622. PMCID: PMC3728693.
Article13. Poon PC, Gupta D, Shoichet MS, Tator CH. Clip compression model is useful for thoracic spinal cord injuries: histologic and functional correlates. Spine (Phila Pa 1976). 2007; 32:2853–2859. DOI: 10.1097/BRS.0b013e31815b7e6b. PMID: 18246008.14. Rivlin AS, Tator CH. Effect of duration of acute spinal cord compression in a new acute cord injury model in the rat. Surg Neurol. 1978; 10:38–43. PMID: 684604.15. Han NR, Yun JI, Park YH, Ahn JY, Kim C, Choi JH, Lee E, Lim JM, Lee ST. Generation of priming mesenchymal stem cells with enhanced potential to differentiate into specific cell lineages using extracellular matrix proteins. Biochem Biophys Res Commun. 2013; 436:413–417. DOI: 10.1016/j.bbrc.2013.05.116. PMID: 23747728.
Article16. Jiang J, Lv Z, Gu Y, Li J, Xu L, Xu W, Lu J, Xu J. Adult rat mesenchymal stem cells differentiate into neuronal-like phenotype and express a variety of neuro-regulatory molecules in vitro. Neurosci Res. 2010; 66:46–52. DOI: 10.1016/j.neures.2009.09.1711. PMID: 19808065.
Article17. Rabchevsky AG, Fugaccia I, Sullivan PG, Scheff SW. Cyclosporin A treatment following spinal cord injury to the rat: behavioral effects and stereological assessment of tissue sparing. J Neurotrauma. 2001; 18:513–522. DOI: 10.1089/089771501300227314. PMID: 11393254.
Article18. Fawcett JW, Asher RA. The glial scar and central nervous system repair. Brain Res Bull. 1999; 49:377–391. DOI: 10.1016/S0361-9230(99)00072-6. PMID: 10483914.
Article19. Clausen F, Lorant T, Lewén A, Hillered L. T lymphocyte trafficking: a novel target for neuroprotection in traumatic brain injury. J Neurotrauma. 2007; 24:1295–1307. DOI: 10.1089/neu.2006.0258. PMID: 17711391.
Article20. McTigue DM, Sahinkaya FR. The fate of proliferating cells in the injured adult spinal cord. Stem Cell Res Ther. 2011; 2:7. DOI: 10.1186/scrt48. PMID: 21345248. PMCID: PMC3092147.
Article21. Brambilla R, Bracchi-Ricard V, Hu WH, Frydel B, Bramwell A, Karmally S, Green EJ, Bethea JR. Inhibition of astroglial nuclear factor kappaB reduces inflammation and improves functional recovery after spinal cord injury. J Exp Med. 2005; 202:145–156. DOI: 10.1084/jem.20041918. PMID: 15998793. PMCID: PMC2212896.
Article22. All AH, Bazley FA, Gupta S, Pashai N, Hu C, Pourmorteza A, Kerr C. Human embryonic stem cell-derived oligodendrocyte progenitors aid in functional recovery of sensory pathways following contusive spinal cord injury. PLoS One. 2012; 7:e47645. DOI: 10.1371/journal.pone.0047645. PMID: 23091637. PMCID: PMC3473046.
Article23. Cusimano M, Biziato D, Brambilla E, Donegà M, Alfaro-Cervello C, Snider S, Salani G, Pucci F, Comi G, Garcia-Verdugo JM, De Palma M, Martino G, Pluchino S. Transplanted neural stem/precursor cells instruct phagocytes and reduce secondary tissue damage in the injured spinal cord. Brain. 2012; 135:447–460. DOI: 10.1093/brain/awr339. PMID: 22271661. PMCID: PMC3558737.
Article24. Sassoli C, Nosi D, Tani A, Chellini F, Mazzanti B, Quercioli F, Zecchi-Orlandini S, Formigli L. Defining the role of mesenchymal stromal cells on the regulation of matrix metalloproteinases in skeletal muscle cells. Exp Cell Res. 2014; 323:297–313. DOI: 10.1016/j.yexcr.2014.03.003. PMID: 24631289.
Article25. Lozito TP, Jackson WM, Nesti LJ, Tuan RS. Human mesenchymal stem cells generate a distinct pericellular zone of MMP activities via binding of MMPs and secretion of high levels of TIMPs. Matrix Biol. 2014; 34:132–143. DOI: 10.1016/j.matbio.2013.10.003. PMID: 24140982.
Article26. Veeravalli KK, Dasari VR, Tsung AJ, Dinh DH, Gujrati M, Fassett D, Rao JS. Human umbilical cord blood stem cells upregulate matrix metalloproteinase-2 in rats after spinal cord injury. Neurobiol Dis. 2009; 36:200–212. DOI: 10.1016/j.nbd.2009.07.012. PMID: 19631747.
Article27. Renault-Mihara F, Mukaino M, Shinozaki M, Kumamaru H, Kawase S, Baudoux M, Ishibashi T, Kawabata S, Nishiyama Y, Sugai K, Yasutake K, Okada S, Nakamura M, Okano H. Regulation of RhoA by STAT3 coordinates glial scar formation. J Cell Biol. 2017; 216:2533–2550. DOI: 10.1083/jcb.201610102. PMID: 28642362. PMCID: PMC5551705.
Article28. Colombo E, Farina C. Astrocytes: key regulators of neuroinflammation. Trends Immunol. 2016; 37:608–620. DOI: 10.1016/j.it.2016.06.006. PMID: 27443914.
Article29. Song K, Li W, Li M. Acute promyelocytic leukemia following autologous bone marrow-derived mesenchymal stem cell transplantation for traumatic brain injury: a case report. Oncol Lett. 2015; 10:2905–2908. DOI: 10.3892/ol.2015.3636. PMID: 26722261. PMCID: PMC4665314.
Article30. Cheriyan T, Ryan DJ, Weinreb JH, Cheriyan J, Paul JC, Lafage V, Kirsch T, Errico TJ. Spinal cord injury models: a review. Spinal Cord. 2014; 52:588–595. DOI: 10.1038/sc.2014.91. PMID: 24912546.
Article31. Fehlings MG, Tator CH. The relationships among the severity of spinal cord injury, residual neurological function, axon counts, and counts of retrogradely labeled neurons after experimental spinal cord injury. Exp Neurol. 1995; 132:220–228. DOI: 10.1016/0014-4886(95)90027-6. PMID: 7789460.
Article32. Bracken MB, Shepard MJ, Collins WF, Holford TR, Young W, Baskin DS, Eisenberg HM, Flamm E, Leo-Summers L, Maroon J, Marshall LF, Perot PL, Piepmeier J, Sonntag VKH, Wagner FC, Wilberger JE, Winn HR. A randomized, controlled trial of methylprednisolone or naloxone in the treatment of acute spinal-cord injury. Results of the Second National Acute Spinal Cord Injury Study. N Engl J Med. 1990; 322:1405–1411. DOI: 10.1056/NEJM199005173222001. PMID: 2278545.
Article33. Fehlings MG, Vaccaro A, Wilson JR, Singh A, W Cadotte D, Harrop JS, Aarabi B, Shaffrey C, Dvorak M, Fisher C, Arnold P, Massicotte EM, Lewis S, Rampersaud R. Early versus delayed decompression for traumatic cervical spinal cord injury: results of the Surgical Timing in Acute Spinal Cord Injury Study (STASCIS). PLoS One. 2012; 7:e32037. DOI: 10.1371/journal.pone.0032037. PMID: 22384132. PMCID: PMC3285644.
Article34. Lee H, Han NR, Hwang JY, Yun JI, Kim C, Park KH, Lee ST. Gelatin Directly enhances neurogenic differentiation potential in bone marrow-derived mesenchymal stem cells without stimulation of neural progenitor cell proliferation. DNA Cell Biol. 2016; 35:530–536. DOI: 10.1089/dna.2016.3237. PMID: 27171118.
Article35. Park YH, Yun JI, Han NR, Park HJ, Ahn JY, Kim C, Choi JH, Lee E, Lim JM, Lee ST. Mass production of early-stage bone-marrow-derived mesenchymal stem cells of rat using gelatin-coated matrix. Biomed Res Int. 2013; 2013:347618. DOI: 10.1155/2013/347618. PMID: 24288676. PMCID: PMC3833006.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Olig2-expressing Mesenchymal Stem Cells Enhance Functional Recovery after Contusive Spinal Cord Injury
- Current Concept and Future of the Management of Spinal Cord Injury: A Systematic Review
- Clinical Response of 277 Patients with Spinal Cord Injury to Stem Cell Therapy in Iraq
- Effects of Human Mesenchymal Stem Cell Transplantation Combined with Polymer on Functional Recovery Following Spinal Cord Hemisection in Rats
- The Effect of Human Adipose Tissue Derived Mesenchymal Stem Cells and Growth Hormone on the Recovery of Neurological Deficits due to Experimental Spinal Cord Injury in Rat