World J Mens Health.
2020 Jan;38(1):123-131. 10.5534/wjmh.180091.
A Simple and Nonenzymatic Method to Isolate Human Corpus Cavernosum Endothelial Cells and Pericytes for the Study of Erectile Dysfunction
- Affiliations
-
- 1National Research Center for Sexual Medicine and Department of Urology, Inha University School of Medicine, Incheon, Korea. jksuh@inha.ac.kr rjk0929@inha.ac.kr
- 2Department of Urology, Yantai Yuhuangding Hospital Affiliated to Medical College of Qingdao University, Yantai, China.
- KMID: 2465417
- DOI: http://doi.org/10.5534/wjmh.180091
Abstract
- PURPOSE
To establish a simple and nonenzymatic technique to isolate endothelial cells (ECs) and pericytes from human corpus cavernosum tissue and to evaluate the angiogenic ability of the human cavernous EC or pericytes for the study of high glucose-induced angiopathy.
MATERIALS AND METHODS
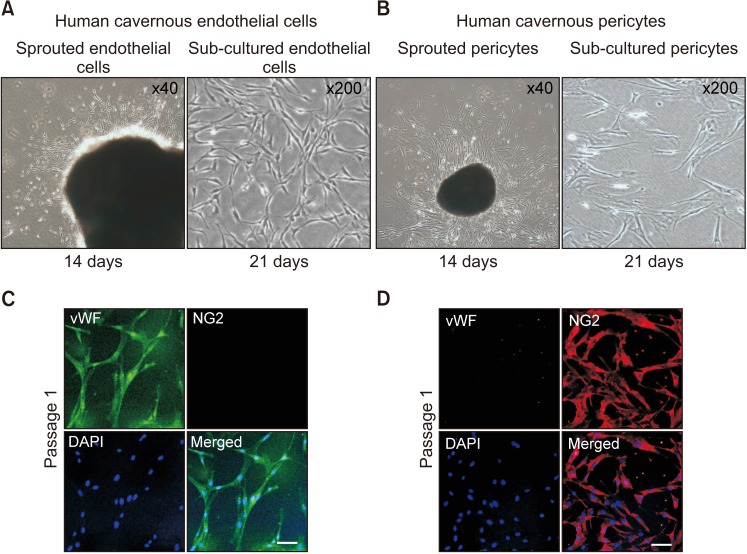
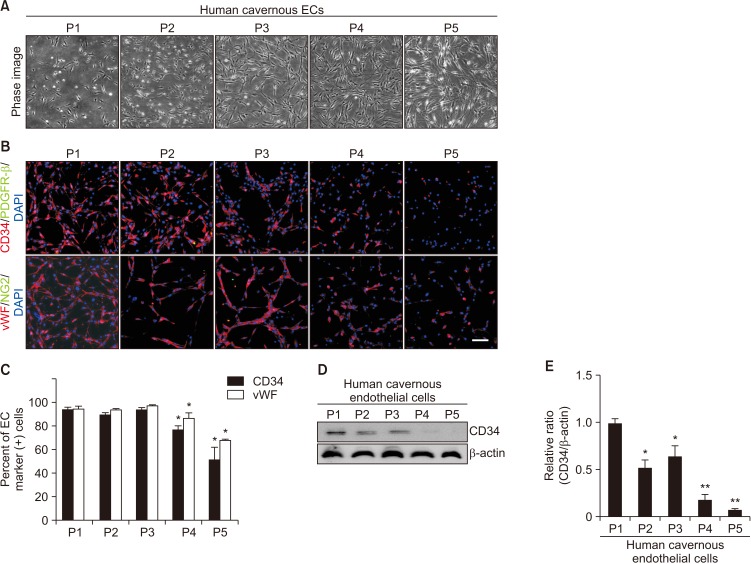
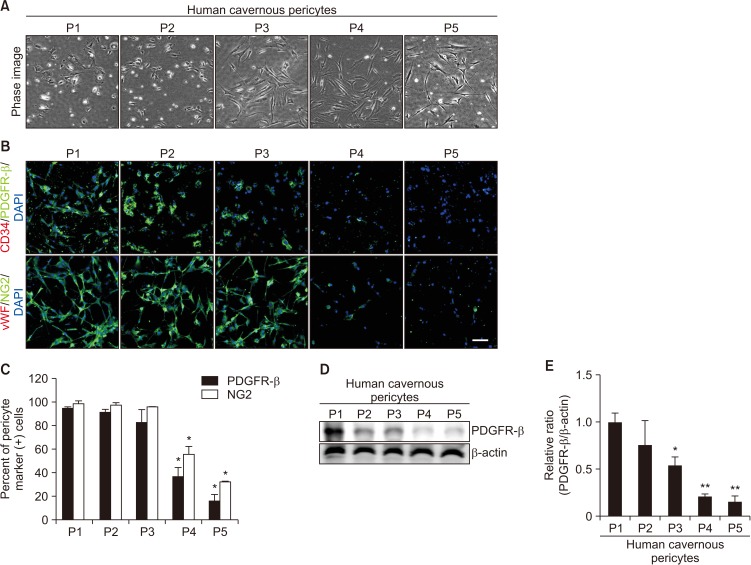
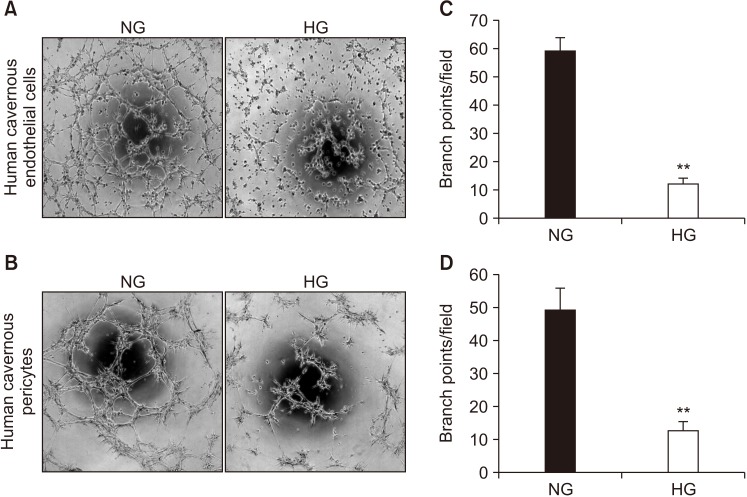
For primary human cavernous EC culture, cavernous tissues were implanted into Matrigel in dishes. For primary human cavernous pericyte culture, cavernous tissues were settled by gravity into dishes. We performed immunocytochemistry and Western blot to determine phenotype and morphologic changes from passage 1 to 5. The primary cultured cells were exposed to a normal-glucose (5 mmol/L) or a high-glucose (30 mmol/L) condition, and then tube formation assay was done.
RESULTS
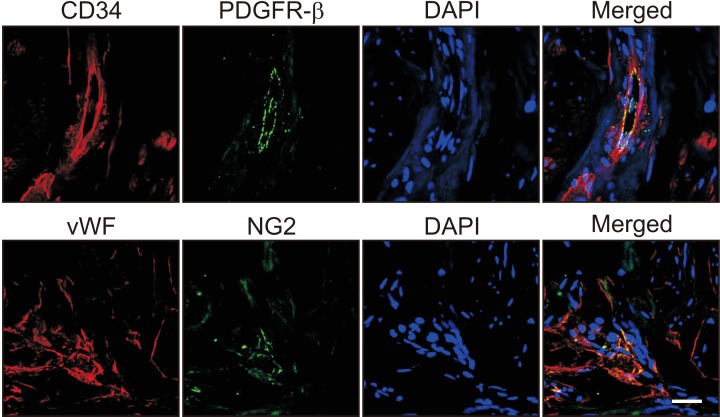
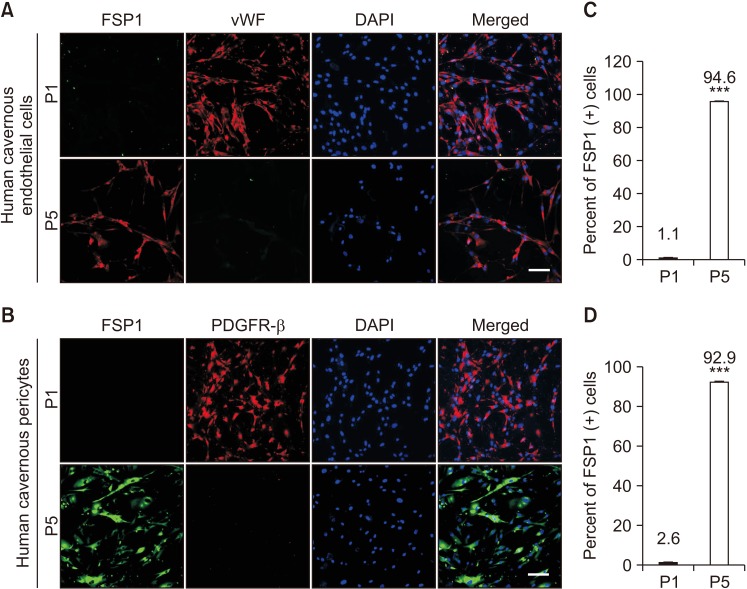
We successfully isolated high-purity EC and pericytes from human corpus cavernosum tissue. Primary cultured EC showed highly positive staining for von Willebrand factor, and pericyte revealed positive staining for NG2 and platelet-derived growth factor receptor-β. Primary cultured EC and pericytes maintained their cellular characteristics up to passage 2 or 3. However, we observed significant changes in their typical phenotype from the passage 4 and morphological characteristics from the passage 3. Human cavernous EC or pericytes formed well-organized capillary-like structures in normal-glucose condition, whereas severely impaired tube formation was detected in high-glucose condition.
CONCLUSIONS
This study provides a simple and nonenzymatic method for primary culture of human cavernous EC and pericytes. Our study will aid us to understand the pathophysiology of diabetic erectile dysfunction, and also be a valuable tool for determining the efficacy of candidate therapeutic targets.
MeSH Terms
Figure
Reference
-
1. Andersson KE. Mechanisms of penile erection and basis for pharmacological treatment of erectile dysfunction. Pharmacol Rev. 2011; 63:811–859. PMID: 21880989.
Article2. Yin GN, Das ND, Choi MJ, Song KM, Kwon MH, Ock J, et al. The pericyte as a cellular regulator of penile erection and a novel therapeutic target for erectile dysfunction. Sci Rep. 2015; 5:10891. PMID: 26044953.
Article3. Yin GN, Park SH, Choi MJ, Limanjaya A, Ghatak K, Minh NN, et al. Penile neurovascular structure revisited: immunohistochemical studies with three-dimensional reconstruction. Andrology. 2017; 5:964–970. PMID: 28805947.
Article4. Verma S, Buchanan MR, Anderson TJ. Endothelial function testing as a biomarker of vascular disease. Circulation. 2003; 108:2054–2059. PMID: 14581384.
Article5. Díaz-Flores L, Gutiérrez R, Varela H, Rancel N, Valladares F. Microvascular pericytes: a review of their morphological and functional characteristics. Histol Histopathol. 1991; 6:269–286. PMID: 1802127.6. Bauer AL, Jackson TL, Jiang Y. A cell-based model exhibiting branching and anastomosis during tumor-induced angiogenesis. Biophys J. 2007; 92:3105–3121. PMID: 17277180.
Article7. Bookholt FD, Monsuur HN, Gibbs S, Vermolen FJ. Mathematical modelling of angiogenesis using continuous cell-based models. Biomech Model Mechanobiol. 2016; 15:1577–1600. PMID: 27037954.
Article8. Finkenzeller G, Torio-Padron N, Momeni A, Mehlhorn AT, Stark GB. In vitro angiogenesis properties of endothelial progenitor cells: a promising tool for vascularization of ex vivo engineered tissues. Tissue Eng. 2007; 13:1413–1420. PMID: 17550338.
Article9. Baudin B, Bruneel A, Bosselut N, Vaubourdolle M. A protocol for isolation and culture of human umbilical vein endothelial cells. Nat Protoc. 2007; 2:481–485. PMID: 17406610.
Article10. Bryan BA, D'Amore PA. Pericyte isolation and use in endothelial/pericyte coculture models. Methods Enzymol. 2008; 443:315–331. PMID: 18772023.11. Maier CL, Shepherd BR, Yi T, Pober JS. Explant outgrowth, propagation and characterization of human pericytes. Microcirculation. 2010; 17:367–380. PMID: 20618694.
Article12. Mogensen C, Bergner B, Wallner S, Ritter A, d'Avis S, Ninichuk V, et al. Isolation and functional characterization of pericytes derived from hamster skeletal muscle. Acta Physiol (Oxf). 2011; 201:413–426. PMID: 20969729.
Article13. Rops AL, van der Vlag J, Jacobs CW, Dijkman HB, Lensen JF, Wijnhoven TJ, et al. Isolation and characterization of conditionally immortalized mouse glomerular endothelial cell lines. Kidney Int. 2004; 66:2193–2201. PMID: 15569308.
Article14. Tigges U, Welser-Alves JV, Boroujerdi A, Milner R. A novel and simple method for culturing pericytes from mouse brain. Microvasc Res. 2012; 84:74–80. PMID: 22484453.
Article15. Bernas MJ, Cardoso FL, Daley SK, Weinand ME, Campos AR, Ferreira AJG, et al. Establishment of primary cultures of human brain microvascular endothelial cells to provide an in vitro cellular model of the blood-brain barrier. Nat Protoc. 2010; 5:1265–1272. PMID: 20595955.
Article16. Yin GN, Ryu JK, Kwon MH, Shin SH, Jin HR, Song KM, et al. Matrigel-based sprouting endothelial cell culture system from mouse corpus cavernosum is potentially useful for the study of endothelial and erectile dysfunction related to high-glucose exposure. J Sex Med. 2012; 9:1760–1772. PMID: 22548733.
Article17. Bala K, Ambwani K, Gohil NK. Effect of different mitogens and serum concentration on HUVEC morphology and characteristics: implication on use of higher passage cells. Tissue Cell. 2011; 43:216–222. PMID: 21511321.
Article18. Neng L, Zhang W, Hassan A, Zemla M, Kachelmeier A, Fridberger A, et al. Isolation and culture of endothelial cells, pericytes and perivascular resident macrophage-like melanocytes from the young mouse ear. Nat Protoc. 2013; 8:709–720. PMID: 23493068.
Article19. Allen DA, Harwood S, Varagunam M, Raftery MJ, Yaqoob MM. High glucose-induced oxidative stress causes apoptosis in proximal tubular epithelial cells and is mediated by multiple caspases. Faseb J. 2003; 17:908–910. PMID: 12670885.
Article20. Rajesh M, Mukhopadhyay P, Bátkai S, Haskó G, Liaudet L, Drel VR, et al. Cannabidiol attenuates high glucose-induced endothelial cell inflammatory response and barrier disruption. Am J Physiol Heart Circ Physiol. 2007; 293:H610–H619. PMID: 17384130.
Article21. Yu T, Sheu SS, Robotham JL, Yoon Y. Mitochondrial fission mediates high glucose-induced cell death through elevated production of reactive oxygen species. Cardiovasc Res. 2008; 79:341–351. PMID: 18440987.
Article22. Langley RR, Ramirez KM, Tsan RZ, Van Arsdall M, Nilsson MB, Fidler IJ. Tissue-specific microvascular endothelial cell lines from H-2K(b)-tsA58 mice for studies of angiogenesis and metastasis. Cancer Res. 2003; 63:2971–2976. PMID: 12782605.23. Moon KH, Park SY, Kim YW. Obesity and erectile dysfunction: from bench to clinical implication. World J Mens Health. 2019; 37:138–147. PMID: 30079640.
Article24. Armulik A, Genové G, Betsholtz C. Pericytes: developmental, physiological, and pathological perspectives, problems, and promises. Dev Cell. 2011; 21:193–215. PMID: 21839917.
Article25. Armulik A, Abramsson A, Betsholtz C. Endothelial/pericyte interactions. Circ Res. 2005; 97:512–523. PMID: 16166562.
Article26. Dellavalle A, Sampaolesi M, Tonlorenzi R, Tagliafico E, Sacchetti B, Perani L, et al. Pericytes of human skeletal muscle are myogenic precursors distinct from satellite cells. Nat Cell Biol. 2007; 9:255–267. PMID: 17293855.
Article27. Capetandes A, Gerritsen ME. Simplified methods for consistent and selective culture of bovine retinal endothelial cells and pericytes. Invest Ophthalmol Vis Sci. 1990; 31:1738–1744. PMID: 2120144.28. Helmbold P, Nayak RC, Marsch WC, Herman IM. Isolation and in vitro characterization of human dermal microvascular pericytes. Microvasc Res. 2001; 61:160–165. PMID: 11254395.
Article29. Yin GN, Park SH, Song KM, Limanjaya A, Ghatak K, Minh NN, et al. Establishment of in vitro model of erectile dysfunction for the study of high-glucose-induced angiopathy and neuropathy. Andrology. 2017; 5:327–335. PMID: 27992968.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Pericyte-derived heme-binding protein 1 promotes angiogenesis and improves erectile function in diabetic mice
- The Expression of eNOS and ET-1 in Corpus Cavernosum in Male Rat with Partial Bladder Outlet Obstruction
- Changes in Corpus Cavernosum after Partial Bladder Outlet Obstruction in Rat
- Stem Cell Therapy for Erectile Dysfunction
- The Effect and Mechanism of the Specific Phosphodiesterase (PDE) III-Inhibitor Milrinone on Human and Rabbit Corpus Cavernosum