Brain Tumor Res Treat.
2019 Oct;7(2):92-97. 10.14791/btrt.2019.7.e36.
Survival and Prognosis of Patients with Pilocytic Astrocytoma: A Single-Center Study
- Affiliations
-
- 1Department of Pediatrics, Keimyung University School of Medicine, Keimyung University Dongsan Medical Center, Daegu, Korea. yejeeshim@dsmc.or.kr
- 2Department of Neurosurgery, Keimyung University School of Medicine, Keimyung University Dongsan Medical Center, Daegu, Korea.
- 3Department of Radiology, Keimyung University School of Medicine, Keimyung University Dongsan Medical Center, Daegu, Korea.
- 4Department of Pathology, Keimyung University School of Medicine, Keimyung University Dongsan Medical Center, Daegu, Korea.
- KMID: 2461181
- DOI: http://doi.org/10.14791/btrt.2019.7.e36
Abstract
- BACKGROUND
Pilocytic astrocytoma (PA) is a brain tumor that is relatively more common in children and young adults.
METHODS
We retrospectively reviewed the medical records of patients with PA treated at a single center between 1988 and 2018.
RESULTS
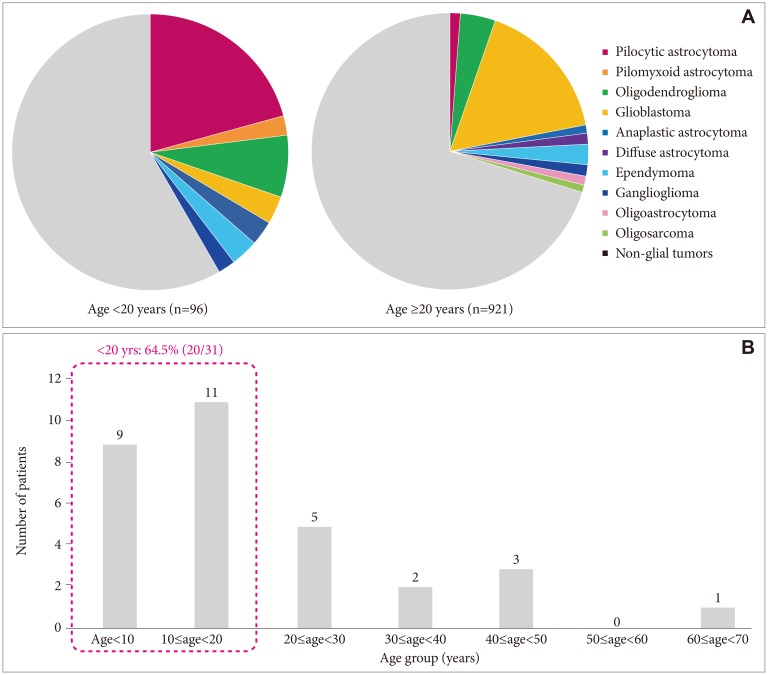
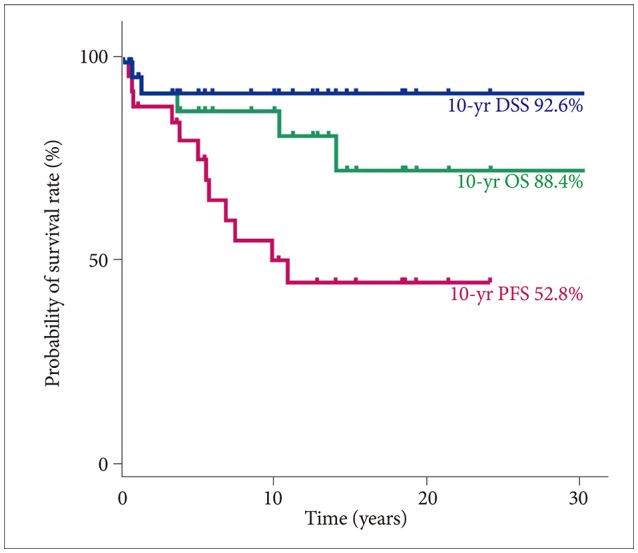
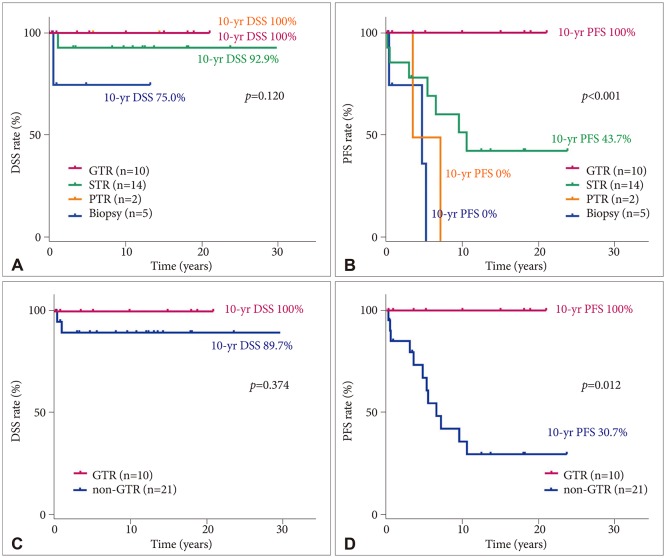
We included 31 subjects with PA. The median age at diagnosis was 13.4 years, and the median follow-up duration was 9.9 years. The total PA group had a 10-year disease-specific survival (DSS) rate of 92.6% [95% confidence interval (CI), 82.6-100] and 10-year progression-free survival (PFS) rate of 52.8% (95% CI, 32.0-73.6). In patients aged <20 years, tumors were more likely to be located in sites in which gross total tumor resection (GTR) was impossible. No statistically significant difference in 10-year DSS was found between the GTR (100%) and non-GTR (89.7%; 95% CI, 76.2-100; p=0.374) groups. However, a statistically significant difference in 10-year PFS was found between the GTR (100%) and non-GTR groups (30.7%; 95% CI, 8.6-52.8; p=0.012). In the non-GTR group, no statistically significant difference in 10-year DSS was found between the patients who received immediate additional chemotherapy and/or radiotherapy (Add-Tx group, 92.9%; 95% CI, 79.4-100) and the non-Add-Tx group (83.3%; 95% CI, 53.5-100; p=0.577). No statistically significant difference in 10-year PFS was found between the Add-Tx group (28.9%; 95% CI, 1.7-56.1) and non-Add-Tx group (33.3%; 95% CI, 0-70.9; p=0.706).
CONCLUSION
The PFS of the patients with PA in our study depended only on the degree of surgical excision associated with tumor location. This study is limited by its small number of patients and retrospective nature. A multicenter and prospective study is necessary to confirm these findings.
Keyword
MeSH Terms
Figure
Reference
-
1. Ostrom QT, Gittleman H, Liao P, et al. CBTRUS statistical report: primary brain and other central nervous system tumors diagnosed in the United States in 2010-2014. Neuro Oncol. 2017; 19(suppl_5):v1–v88. PMID: 29117289.
Article2. Ostrom QT, Gittleman H, Xu J, et al. CBTRUS statistical report: primary brain and other central nervous system tumors diagnosed in the United States in 2009-2013. Neuro Oncol. 2016; 18(suppl_5):v1–v75. PMID: 28475809.
Article3. Jung SY, Jin SL, Hahn SM, et al. Radiation or chemotherapy rather than observation may be a better modality after subtotal resection for pilocytic astrocytoma in children. Clin Pediatr Hematol Oncol. 2014; 21:95–103.
Article4. Ryu HH, Jung TY, Lee GJ, et al. Differences in the clinical courses of pediatric and adult pilocytic astrocytomas with progression: a single-institution study. Childs Nerv Syst. 2015; 31:2063–2069. PMID: 26293677.
Article5. Cho BK, Jung HL, Ghim TT, et al. KSPNO protocol for glioma. Korean J Pediatr Hematol Oncol. 2005; 12:244–285.6. Banerjee A, Nicolaides T. Low-gade gliomas. In : Gupta N, Banerjee A, Haas-Kogan DA, editors. Pediatric CNS tumors. 3rd ed. Heidelberg: Springer;2017. p. 1–35.7. Dirven CM, Mooij JJ, Molenaar WM. Cerebellar pilocytic astrocytoma: a treatment protocol based upon analysis of 73 cases and a review of the literature. Childs Nerv Syst. 1997; 13:17–23. PMID: 9083697.
Article8. Fernandez C, Figarella-Branger D, Girard N, et al. Pilocytic astrocytomas in children: prognostic factors--a retrospective study of 80 cases. Neurosurgery. 2003; 53:544–553. PMID: 12943571.
Article9. Shaw EG, Wisoff JH. Prospective clinical trials of intracranial low-grade glioma in adults and children. Neuro Oncol. 2003; 5:153–160. PMID: 12816721.10. Pollack IF, Claassen D, al-Shboul Q, Janosky JE, Deutsch M. Low-grade gliomas of the cerebral hemispheres in children: an analysis of 71 cases. J Neurosurg. 1995; 82:536–547. PMID: 7897512.
Article11. Karim AB, Afra D, Cornu P, et al. Randomized trial on the efficacy of radiotherapy for cerebral low-grade glioma in the adult: European Organization for Research and Treatment of Cancer Study 22845 with the Medical Research Council study BRO4: an interim analysis. Int J Radiat Oncol Biol Phys. 2002; 52:316–324. PMID: 11872276.
Article12. Gnekow AK, Falkenstein F, von Hornstein S, et al. Long-term follow-up of the multicenter, multidisciplinary treatment study HIT-LGG-1996 for low-grade glioma in children and adolescents of the German Speaking Society of Pediatric Oncology and Hematology. Neuro Oncol. 2012; 14:1265–1284. PMID: 22942186.
Article13. Ater JL, Zhou T, Holmes E, et al. Randomized study of two chemotherapy regimens for treatment of low-grade glioma in young children: a report from the Children’s Oncology Group. J Clin Oncol. 2012; 30:2641–2647. PMID: 22665535.
Article14. Bandopadhayay P, Bergthold G, London WB, et al. Long-term outcome of 4,040 children diagnosed with pediatric low-grade gliomas: an analysis of the Surveillance Epidemiology and End Results (SEER) database. Pediatr Blood Cancer. 2014; 61:1173–1179. PMID: 24482038.
Article15. Kortmann RD, Timmermann B, Taylor RE, et al. Current and future strategies in radiotherapy of childhood low-grade glioma of the brain. Part II: treatment-related late toxicity. Strahlenther Onkol. 2003; 179:585–597. PMID: 14628124.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Multiple Solid Pilocytic Astrocytomas in Cerebellum with Neurofibromatosis Type I: A Case Report
- Supratentorial Pilocytic Astrocytoma Mimicking Convexity Meningioma with Early Anaplastic Transformation: A Case Report
- A Case of Huge Pilocytic Astrocytoma Causing Eyeball Subluxation
- Pilocytic Astrocytoma Occuring in a Patient Treated with Gamma Knife Surgery: Case Report
- Juvenile Pilocytic Astrocytoma of the Hypohtalamus: Case Report