Ann Dermatol.
2019 Oct;31(5):511-517. 10.5021/ad.2019.31.5.511.
Expression Pattern and Role of Klotho in Human Hair Follicles
- Affiliations
-
- 1Department of Dermatology and Institute of Hair and Cosmetic Medicine, Yonsei University Wonju College of Medicine, Wonju, Korea. leewonsoo@yonsei.ac.kr
- 2Department of Dermatology, Affiliated Hospital of Yanbian University, Yanji, China.
- KMID: 2456227
- DOI: http://doi.org/10.5021/ad.2019.31.5.511
Abstract
- BACKGROUND
Klotho protein plays a pivotal role in aging regulation. However, it is unclear whether klotho is expressed in human hair follicles and is correlated with hair growth.
OBJECTIVE
The purpose of this study was to determine the expression pattern and role of klotho in human hair follicles.
METHODS
We examined the klotho expression patterns in human hair follicles from young and aged donors. Furthermore, we examined the functional roles of klotho on human hair growth using klotho siRNA and klotho recombinant protein.
RESULTS
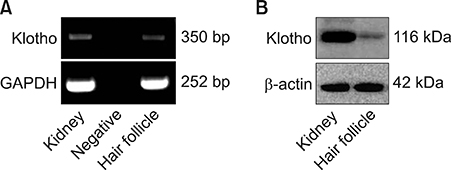
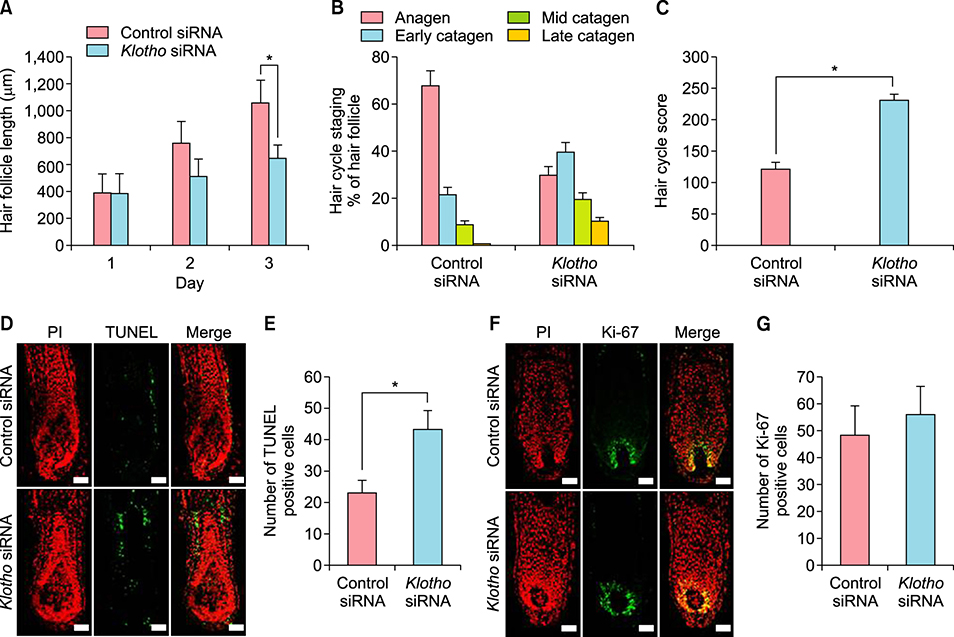
Interestingly, klotho was expressed in human hair follicles at both gene and protein levels. In hair follicles, prominent klotho expression was mainly observed in the outermost regions of the outer root sheath and hair bulb matrix cells. Quantification of klotho protein expression in young and aged donors showed that klotho expression decreased with aging. In human hair follicle organ culture, klotho silencing promoted premature catagen induction and inhibited human hair growth. Otherwise, klotho protein prolonged human hair growth.
CONCLUSION
These results indicate that klotho might be an important regulatory factor for human hair growth and hair cycle change.
Keyword
MeSH Terms
Figure
Reference
-
1. Stenn KS, Paus R. Controls of hair follicle cycling. Physiol Rev. 2001; 81:449–494.
Article2. Paus R, Cotsarelis G. The biology of hair follicles. N Engl J Med. 1999; 341:491–497.
Article3. Alonso L, Fuchs E. The hair cycle. J Cell Sci. 2006; 119(Pt 3):391–393.
Article4. Ahn SY, Pi LQ, Hwang ST, Lee WS. Effect of IGF-I on hair growth is related to the anti-apoptotic effect of IGF-I and up-regulation of PDGF-A and PDGF-B. Ann Dermatol. 2012; 24:26–31.
Article5. Rishikaysh P, Dev K, Diaz D, Qureshi WM, Filip S, Mokry J. Signaling involved in hair follicle morphogenesis and development. Int J Mol Sci. 2014; 15:1647–1670.
Article6. Kuro-O M. Phosphate and Klotho. Kidney Int Suppl. 2011; 79:S20–S23.
Article7. Matsumura Y, Aizawa H, Shiraki-Iida T, Nagai R, Kuro-o M, Nabeshima Y. Identification of the human klotho gene and its two transcripts encoding membrane and secreted klotho protein. Biochem Biophys Res Commun. 1998; 242:626–630.
Article8. Shiraki-Iida T, Aizawa H, Matsumura Y, Sekine S, Iida A, Anazawa H, et al. Structure of the mouse klotho gene and its two transcripts encoding membrane and secreted protein. FEBS Lett. 1998; 424:6–10.
Article9. Kuro-o M. Klotho as a regulator of oxidative stress and senescence. Biol Chem. 2008; 389:233–241.
Article10. Kurosu H, Yamamoto M, Clark JD, Pastor JV, Nandi A, Gurnani P, et al. Suppression of aging in mice by the hormone Klotho. Science. 2005; 309:1829–1833.
Article11. Liu H, Fergusson MM, Castilho RM, Liu J, Cao L, Chen J, et al. Augmented Wnt signaling in a mammalian model of accelerated aging. Science. 2007; 317:803–806.
Article12. Doi S, Zou Y, Togao O, Pastor JV, John GB, Wang L, et al. Klotho inhibits transforming growth factor-beta1 (TGF-beta1) signaling and suppresses renal fibrosis and cancer metastasis in mice. J Biol Chem. 2011; 286:8655–8665.
Article13. Kamemori M, Ohyama Y, Kurabayashi M, Takahashi K, Nagai R, Furuya N. Expression of Klotho protein in the inner ear. Hear Res. 2002; 171:103–110.
Article14. Pi LQ, Jin XH, Hwang ST, Lee WS. Effects of calcitonin gene-related peptide on the immune privilege of human hair follicles. Neuropeptides. 2013; 47:51–57.
Article15. Kloepper JE, Sugawara K, Al-Nuaimi Y, Gáspár E, Van Beek N, Paus R. Methods in hair research: how to objectively distinguish between anagen and catagen in human hair follicle organ culture. Exp Dermatol. 2010; 19:305–312.
Article16. Samuelov L, Sprecher E, Tsuruta D, Bíró T, Kloepper JE, Paus R. P-cadherin regulates human hair growth and cycling via canonical Wnt signaling and transforming growth factor-β 2. J Invest Dermatol. 2012; 132:2332–2341.
Article17. Kuro-o M. Klotho. Pflugers Arch. 2010; 459:333–343.
Article18. Nakai K, Yoneda K, Haba R, Kushida Y, Katsuki N, Moriue T, et al. Deranged epidermal differentiation in kl/kl mouse and the effects of β Klotho siRNA on the differentiation of HaCaT cells. Exp Dermatol. 2013; 22:772–774.
Article19. Courtois M, Loussouarn G, Hourseau C, Grollier JF. Ageing and hair cycles. Br J Dermatol. 1995; 132:86–93.
Article20. Trüeb RM. Aging of hair. J Cosmet Dermatol. 2005; 4:60–72.
Article21. Ellis JA, Sinclair RD. Male pattern baldness: current treatments, future prospects. Drug Discov Today. 2008; 13:791–797.
Article22. Karnik P, Shah S, Dvorkin-Wininger Y, Oshtory S, Mirmirani P. Microarray analysis of androgenetic and senescent alopecia: comparison of gene expression shows two distinct profiles. J Dermatol Sci. 2013; 72:183–186.
Article23. Borbíró I, Lisztes E, Tóth BI, Czifra G, Oláh A, Szöllosi AG, et al. Activation of transient receptor potential vanilloid-3 inhibits human hair growth. J Invest Dermatol. 2011; 131:1605–1614.
Article24. Lindner G, Botchkarev VA, Botchkareva NV, Ling G, van der Veen C, Paus R. Analysis of apoptosis during hair follicle regression (catagen). Am J Pathol. 1997; 151:1601–1617.25. Soma T, Ogo M, Suzuki J, Takahashi T, Hibino T. Analysis of apoptotic cell death in human hair follicles in vivo and in vitro. J Invest Dermatol. 1998; 111:948–954.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Hair Cycle-associated Changes of the Immunological Markers in Human Hair Follicles
- Expression of Sfrp2 Is Increased in Catagen of Hair Follicles and Inhibits Keratinocyte Proliferation
- The Analysis of the Expression of TGF-beta in Human Hair Follicles in vivo
- Trichofolliculoma
- Hair Follicle Nevus Located on the Neck: Comparison with Accessory Tragus, Cervical Chondrocutaneous Branchial Remnants and Trichofolliculoma