Clin Exp Vaccine Res.
2019 Jul;8(2):110-115. 10.7774/cevr.2019.8.2.110.
A meningococcal B vaccine induces cross-protection against gonorrhea
- Affiliations
-
- 1Department of Immunology, Institute of Basic and Preclinical Sciences, University of Medical Sciences, Havana, Cuba. rochoa@infomed.sld.cu
- KMID: 2455082
- DOI: http://doi.org/10.7774/cevr.2019.8.2.110
Abstract
- PURPOSE
Neisseria meningitidis and Neisseria gonorrhoeae share between 80% and 90% of their genetic sequence. Meningococcal serogroup B vaccines based on outer membrane vesicles"”such as VA-MENGOC-BC"”could cross-protect against gonorrhea. The aim of this study was to analyze the incidence rates of gonorrhea and other sexually transmitted diseases with respect to the use of the VA-MENGOC-BC vaccine.
MATERIALS AND METHODS
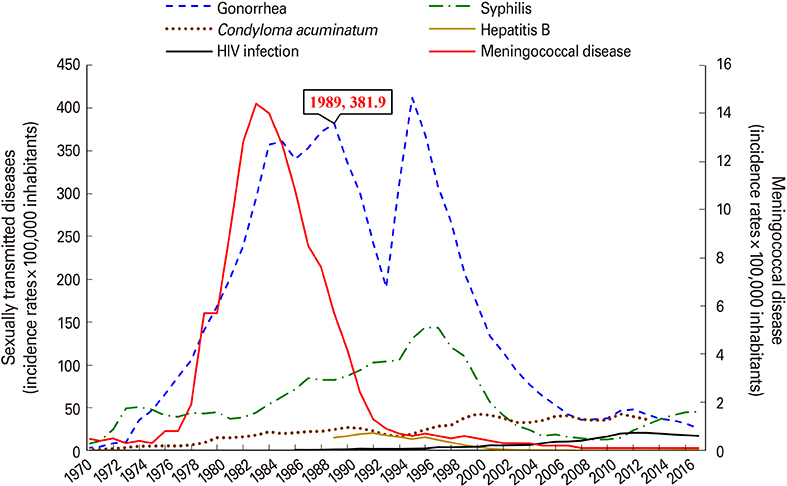
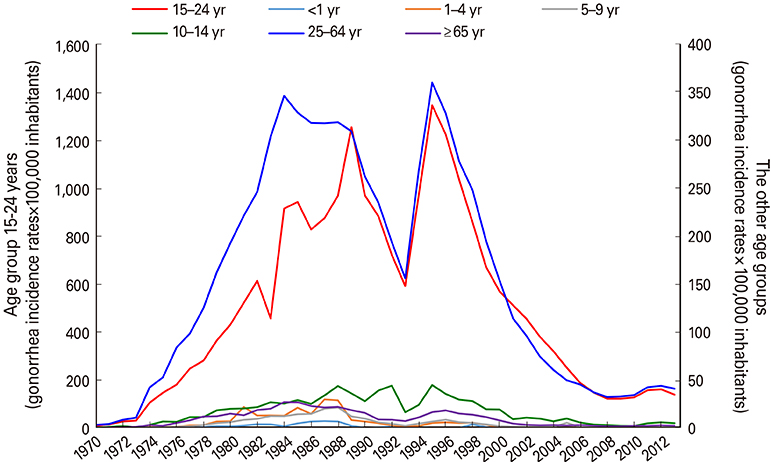
Health statistics between 1970 and 2017 were reviewed and the incidence of meningococcal disease and sexually transmitted diseases (gonorrhea, syphilis, condyloma acuminatum, hepatitis B and human immunodeficiency virus infection) were analyzed during the pre- and post-vaccination periods. Gonorrhea incidence was also analyzed by age groups.
RESULTS
VA-MENGOC-BC was successfully used to control a meningococcal epidemic in Cuba. The strategy to combat the epidemic was carried out in two stages. The first one was a nationwide mass-vaccination campaign from 1989 to 1990, targeting the population at highest-risk aged 3 months to 24 years. During the second stage, begun in 1991, it was included in the Expanded Immunization Program. Gonorrhea incidence increased from 1970 to 1989. However, after the VA-MENGOC-BC massive vaccination campaign a sharp decrease of gonorrhea incidence was observed. It lasted between 1989 and 1993. A second incidence peak was detected in 1995, but it dropped again. Data clearly show a decline in the incidence of gonorrhea following massive vaccination, in contrast with other sexually transmitted diseases. Incidence rates in unvaccinated age groups also decreased, probably due to herd immunity.
CONCLUSION
There is evidence that VA-MENGOC-BC could induce a moderate protection against gonorrhea.
MeSH Terms
Figure
Reference
-
1. Gonococcal Vaccine Development. Gonococcal vaccine development: lessons from group B meningococcal vaccines. EBioMedicine. 2017; 22:1.2. Abbasi J. New hope for a gonorrhea vaccine. JAMA. 2017; 318:894–895.
Article3. Petousis-Harris H. Impact of meningococcal group B OMV vaccines, beyond their brief. Hum Vaccin Immunother. 2018; 14:1058–1063.
Article4. Petousis-Harris H, Paynter J, Morgan J, et al. Effectiveness of a group B outer membrane vesicle meningococcal vaccine against gonorrhoea in New Zealand: a retrospective case-control study. Lancet. 2017; 390:1603–1610.
Article5. Seib KL. Gonorrhoea vaccines: a step in the right direction. Lancet. 2017; 390:1567–1569.
Article6. Ochoa-Azze RF. Cross-protection induced by VA-MENGOC-BC vaccine. Hum Vaccin Immunother. 2018; 14:1064–1068.
Article7. Perez O, del Campo J, Cuello M, et al. Mucosal approaches in Neisseria Vaccinology. Vaccimonitor. 2009; 18:55–57.8. Sierra GV, Campa HC, Varcacel NM, et al. Vaccine against group B Neisseria meningitidis: protection trial and mass vaccination results in Cuba. NIPH Ann. 1991; 14:195–207.9. Sotolongo F, Campa C, Casanueva V, Fajardo EM, Cuevas IE, Gonzalez N. Cuban meningococcal BC vaccine: experiences & contributions from 20 years of application. MEDICC Rev. 2007; 9:16–22.10. Ministerio de Salud Publica (MINSAP). Direccion Nacional de Registros Medicos y Estadistica de Salud. Indicadores del estado de salud de la poblacion. Series de tiempo. Cuba 1970–2013 [Internet]. La Habana: MINSAP;c2014. cited 2019 Feb 19. Available from: http://files.sld.cu/dne/files/2014/06/series-de-tiempo-cuba-1970_2013.pdf.11. Ministerio de Salud Publica (MINSAP). Direccion Nacional de Registros Medicos y Estadistica de Salud. Anuario Estadistico de Salud 2017 [Internet]. La Habana: MINSAP;c2018. cited 2019 Feb 19. Available from: http://bvscuba.sld.cu/2017/11/20/anuario-estadistico-de-salud-de-cuba/.12. Joint United Nations Programme on HIV and AIDS (UNAIDS). Country factsheets [Internet]. Geneva: UNAIDS;c2017. cited 2019 Feb 19. Available from: http://www.unaids.org/en/regionscountries/countries/cuba.13. Gorry C. Cuba's national HIV/AIDS program. MEDICC Rev. 2011; 13:5–8.
Article14. Ochoa-Azze RF, Garcia-Imia L, Verez-Bencomo V. Effectiveness of a serogroup B and C meningococcal vaccine developed in Cuba. MEDICC Rev. 2018; 20:22–29.
Article15. Acevedo R, Bai X, Borrow R, et al. The Global Meningococcal Initiative meeting on prevention of meningococcal disease worldwide: epidemiology, surveillance, hypervirulent strains, antibiotic resistance and high-risk populations. Expert Rev Vaccines. 2019; 18:15–30.
Article16. Cuello M, Cabrera O, Acevedo R, et al. Nasal immunization with AFCo1 induces immune response to N. gonorrhoeae in mice. Vaccimonitor. 2009; 18:78–80.17. Smith KA. Smallpox: can we still learn from the journey to eradication? Indian J Med Res. 2013; 137:895–899.18. World Health Organization (WHO). WHO/RHR/16.09. Global health sector strategy on sexually transmitted infections. 2016–2021 [Internet]. Geneva: WHO;c2016. cited 2019 Feb 19. Available from: http://www.who.int/reproductivehealth/publications/rtis/ghss-stis/en/.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Meningococcal Vaccines
- A Case Study of Meningococcal Vaccination At a University Hospital
- Neonatal Sepsis and Meningitis Caused by Neisseria meningitidis Serogroup B: a Case Report
- General Recommendations and Recent Update in Adult Immunization
- A Korean familial case of hereditary complement 7 deficiency