Blood Res.
2018 Jun;53(2):130-137. 10.5045/br.2018.53.2.130.
Clinical characteristics and outcomes of thrombotic microangiopathy in Malaysia
- Affiliations
-
- 1Department of Hematology, Hospital Ampang, Ampang, Malaysia. mandyyapyy@yahoo.com
- 2Clinical Trial Unit, Clinical Research Centre, Ministry of Health, Putrajaya, Malaysia.
- 3Perth Blood Institute, Murdoch University, Perth, Australia.
- 4Western Australian Centre for Thrombosis and Hemostasis, Murdoch University, Perth, Australia.
- KMID: 2451031
- DOI: http://doi.org/10.5045/br.2018.53.2.130
Abstract
- BACKGROUND
Thrombotic microangiopathy (TMA) with non-deficient ADAMTS-13 (a disintegrin-like and metalloprotease with thrombospondin type 1 motif 13) outcome is unknown hence the survival analysis correlating with ADAMTS-13 activity is conducted in Malaysia.
METHODS
This was a retrospective epidemiological study involving all cases of TMA from 2012-2016.
RESULTS
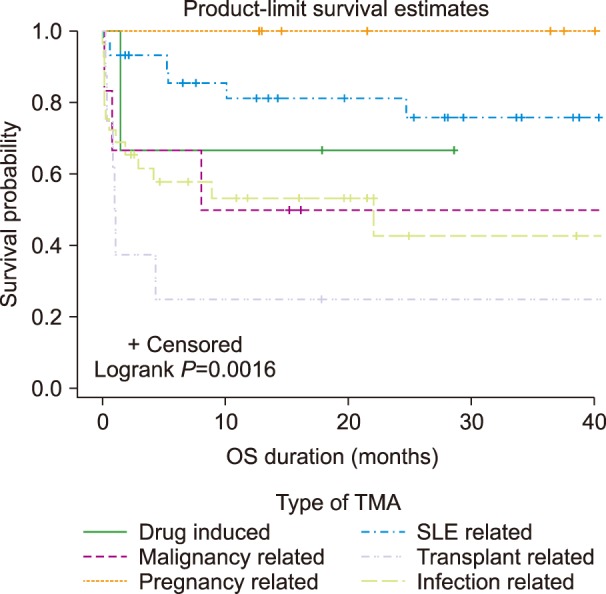
We evaluated 243 patients with a median age of 34.2 years; 57.6% were female. Majority of the patients were Malay (62.5%), followed by Chinese (23.5%) and Indian (8.6%). The proportion of patients with thrombotic thrombocytopenic purpura (TTP) was 20.9%, 72.2% of which were acquired while 27.8% were congenital. Patients with ADAMTS-13 activity ≥5% had a four-fold higher odds of mortality compared to those with ADAMTS-13 activity <5% (odds ratio: 4.133, P=0.0425). The mortality rate was 22.6% (N=55). Most cases had secondary etiologies (42.5%), followed by acquired TTP (16.6%), atypical hemolytic uremic syndrome (HUS) or HUS (12.8%) and congenital TTP (6.4%). Patients with secondary TMA had inferior overall survival (P=0.0387). The secondary causes comprised systemic lupus erythematosus (30%), infection (29%), pregnancy (10%), transplant (8%), malignancy (6%), and drugs (3%). Transplant-associated TMA had the worst OS (P=0.0016) among the secondary causes. Plasma exchange, methylprednisolone and intravenous immunoglobulin were recorded as first-line treatments in 162 patients, while rituximab, bortezomib, vincristine, azathioprine, cyclophosphamide, cyclosporine, and tacrolimus were described in 78 patients as second-line treatment.
CONCLUSION
This study showed that TMA without ADAMTS-13 deficiency yielded inferior outcomes compared to TMA with severeADAMTS-13 deficiency, although this difference was not statistically significant.
MeSH Terms
-
Asian Continental Ancestry Group
Atypical Hemolytic Uremic Syndrome
Azathioprine
Bortezomib
Cyclophosphamide
Cyclosporine
Epidemiologic Studies
Female
Humans
Immunoglobulins
Lupus Erythematosus, Systemic
Malaysia*
Methylprednisolone
Mortality
Plasma Exchange
Pregnancy
Purpura, Thrombotic Thrombocytopenic
Retrospective Studies
Rituximab
Tacrolimus
Thrombospondins
Thrombotic Microangiopathies*
Vincristine
Azathioprine
Bortezomib
Cyclophosphamide
Cyclosporine
Immunoglobulins
Methylprednisolone
Rituximab
Tacrolimus
Thrombospondins
Vincristine
Figure
Cited by 1 articles
-
Validating lactate dehydrogenase (LDH) as a component of the PLASMIC predictive tool (PLASMIC-LDH)
Christopher Chin Keong Liam, Jim Yu-Hsiang Tiao, Yee Yee Yap, Yi Lin Lee, Jameela Sathar, Simon McRae, Amanda Davis, Jennifer Curnow, Robert Bird, Philip Choi, Pantep Angchaisuksiri, Sim Leng Tien, Joyce Ching Mei Lam, Doyeun Oh, Jin Seok Kim, Sung-Soo Yoon, Raymond Siu-Ming Wong, Carolyn Lauren, Eileen Grace Merriman, Anoop Enjeti, Mark Smith, Ross Ian Baker
Blood Res. 2023;58(1):36-41. doi: 10.5045/br.2023.2022133.
Reference
-
1. Scully M, Goodship T. How I treat thrombotic thrombocytopenic purpura and atypical haemolytic uraemic syndrome. Br J Haematol. 2014; 164:759–766. PMID: 24387053.
Article3. Sayani FA, Abrams CS. How I treat refractory thrombotic thrombocytopenic purpura. Blood. 2015; 125:3860–3867. PMID: 25784681.
Article4. Joly BS, Coppo P, Veyradier A. Thrombotic thrombocytopenic purpura. Blood. 2017; 129:2836–2846. PMID: 28416507.
Article5. Masias C, Vasu S, Cataland SR. None of the above: thrombotic microangiopathy beyond TTP and HUS. Blood. 2017; 129:2857–2863. PMID: 28416509.
Article6. Bendapudi PK, Makar RS. An algorithmic approach to the diagnosis and management of the thrombotic microangiopathies. Am J Clin Pathol. 2016; 145:152–154. PMID: 26864515.
Article7. Caprioli J, Noris M, Brioschi S, et al. Genetics of HUS: the impact of MCP, CFH, and IF mutations on clinical presentation, response to treatment, and outcome. Blood. 2006; 108:1267–1279. PMID: 16621965.
Article8. Sadler JE. What's new in the diagnosis and pathophysiology of thrombotic thrombocytopenic purpura. Hematology Am Soc Hematol Educ Program. 2015; 2015:631–636. PMID: 26637781.
Article9. Alwan F, Vendramin C, Vanhoorelbeke K, et al. Presenting ADAMTS13 antibody and antigen levels predict prognosis in immune-mediated thrombotic thrombocytopenic purpura. Blood. 2017; 130:466–471. PMID: 28576877.
Article10. Green D. Predicting mortality in thrombotic thrombocytopenic purpura. Waltham, MA: NEJM Journal Watch;2017. Accessed July 5, 2017. at http://www.jwatch.org/na44328/2017/06/09/predicting-mortality-thrombotic-thrombocytopenic-purpura.11. Bendapudi PK, Li A, Hamdan A, et al. Impact of severe ADAMTS13 deficiency on clinical presentation and outcomes in patients with thrombotic microangiopathies: the experience of the Harvard TMA Research Collaborative. Br J Haematol. 2015; 171:836–844. PMID: 26314936.
Article12. Bendapudi PK, Li A, Hamdan A, et al. Derivation and prospective validation of a predictive score for the rapid diagnosis of thrombotic thrombocytopenic purpura: the plasmic score. Blood (ASH Annual Meeting Abstracts). 2014; 124(Suppl):abst 231.
Article13. Jodele S, Laskin BL, Dandoy CE, et al. A new paradigm: Diagnosis and management of HSCT-associated thrombotic microangiopathy as multi-system endothelial injury. Blood Rev. 2015; 29:191–204. PMID: 25483393.
Article14. Matevosyan K, Sarode R. Thrombosis, microangiopathies, and inflammation. Semin Thromb Hemost. 2015; 41:556–562. PMID: 26276936.
Article15. Fuchs TA, Kremer Hovinga JA, Schatzberg D, Wagner DD, Lämmle B. Circulating DNA and myeloperoxidase indicate disease activity in patients with thrombotic microangiopathies. Blood. 2012; 120:1157–1164. PMID: 22611154.
Article16. Loirat C, Fakhouri F, Ariceta G, et al. An international consensus approach to the management of atypical hemolytic uremic syndrome in children. Pediatr Nephrol. 2016; 31:15–39. PMID: 25859752.
Article17. Coppo P. French Reference Center for Thrombotic Microangiopathies. Treatment of autoimmune thrombotic thrombocytopenic purpura in the more severe forms. Transfus Apher Sci. 2017; 56:52–56. PMID: 28110841.
Article18. Scully M, McDonald V, Cavenagh J, et al. A phase 2 study of the safety and efficacy of rituximab with plasma exchange in acute acquired thrombotic thrombocytopenic purpura. Blood. 2011; 118:1746–1753. PMID: 21636861.
Article19. Froissart A, Buffet M, Veyradier A, et al. Efficacy and safety of first-line rituximab in severe, acquired thrombotic thrombocytopenic purpura with a suboptimal response to plasma exchange. Experience of the French Thrombotic Microangiopathies Reference Center. Crit Care Med. 2012; 40:104–111. PMID: 21926591.
Article20. Page EE, Kremer Hovinga JA, Terrell DR, Vesely SK, George JN. Rituximab reduces risk for relapse in patients with thrombotic thrombocytopenic purpura. Blood. 2016; 127:3092–3094. PMID: 27060171.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Characteristics and management of thrombotic microangiopathy in kidney transplantation
- Changes in VWF-cleaving Metalloprotease (ADAMTS 13) activity in the thrombotic microangiopathy after kidney tranplantation
- Antiphospholipid Syndrome Presenting as Recurrent Pancreatitis and Renal Thrombotic Microangiopathy
- A Case of Locally Advanced Breast Cancer Complicated by Pulmonary Tumor Thrombotic Microangiopathy
- Thrombotic microangiopathy resulting from neglected blood pressure control