Intest Res.
2019 Apr;17(2):192-201. 10.5217/ir.2018.00106.
Evaluation of commercial probiotic lactic cultures against biofilm formation by Cronobacter sakazakii
- Affiliations
-
- 1Department of Biotechnology and Bioinformatics, Jaypee University of Information Technology, Waknaghat, India. gunjanmicro@gmail.com
- 2Department of Microbiology, School of Interdisciplinary and Applied Life Sciences, Central University of Haryana, Mahendergarh, India.
- KMID: 2449960
- DOI: http://doi.org/10.5217/ir.2018.00106
Abstract
- BACKGROUND/AIMS
Cronobacter sakazakii, an emergent pathogen is considered as a major concern to infants and neonates fed on reconstituted powdered infant milk formula. In conjunction with many other factors, biofilm forming capacity adds to its pathogenic potential. In view of the facts that infants are at highest risk to C. sakazakii infections, and emerging antibiotic resistance among pathogens, it is imperative to evaluate probiotic cultures for their efficacy against C. sakazakii. Therefore, pure probiotic strains were isolated from commercial probiotic products and tested for their antimicrobial and anti-biofilm activities against C. sakazakii.
METHODS
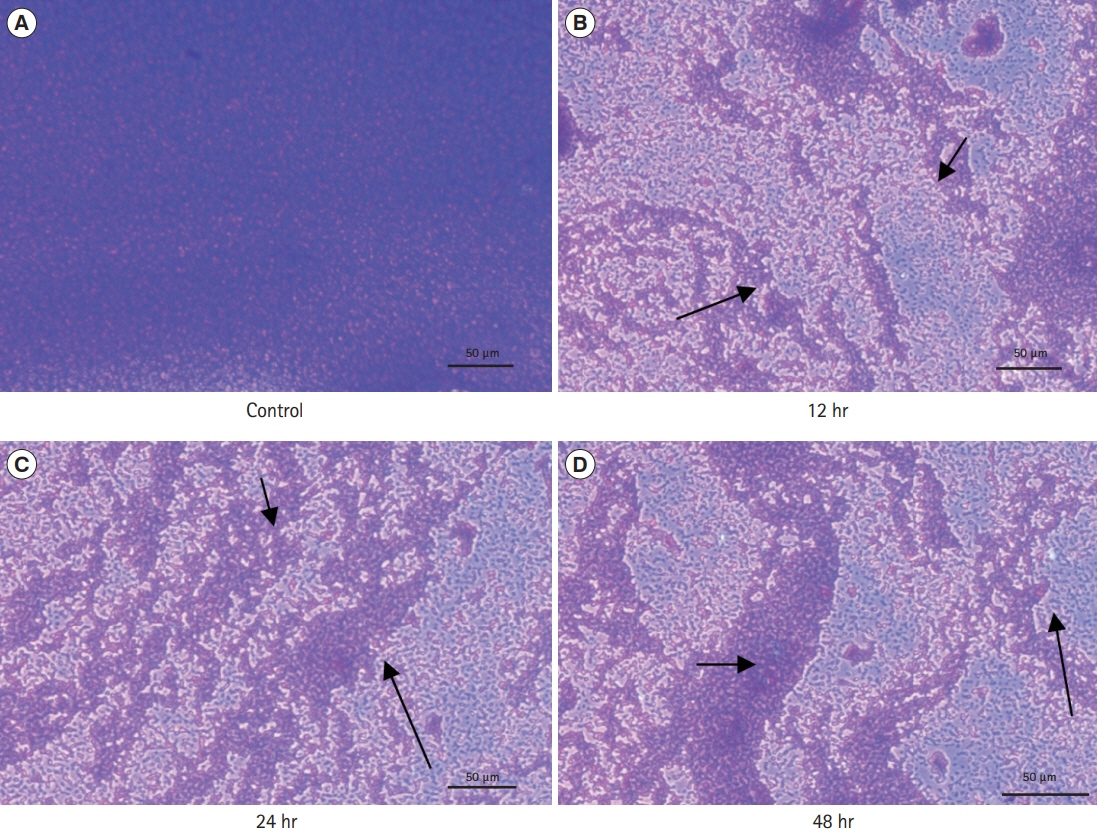
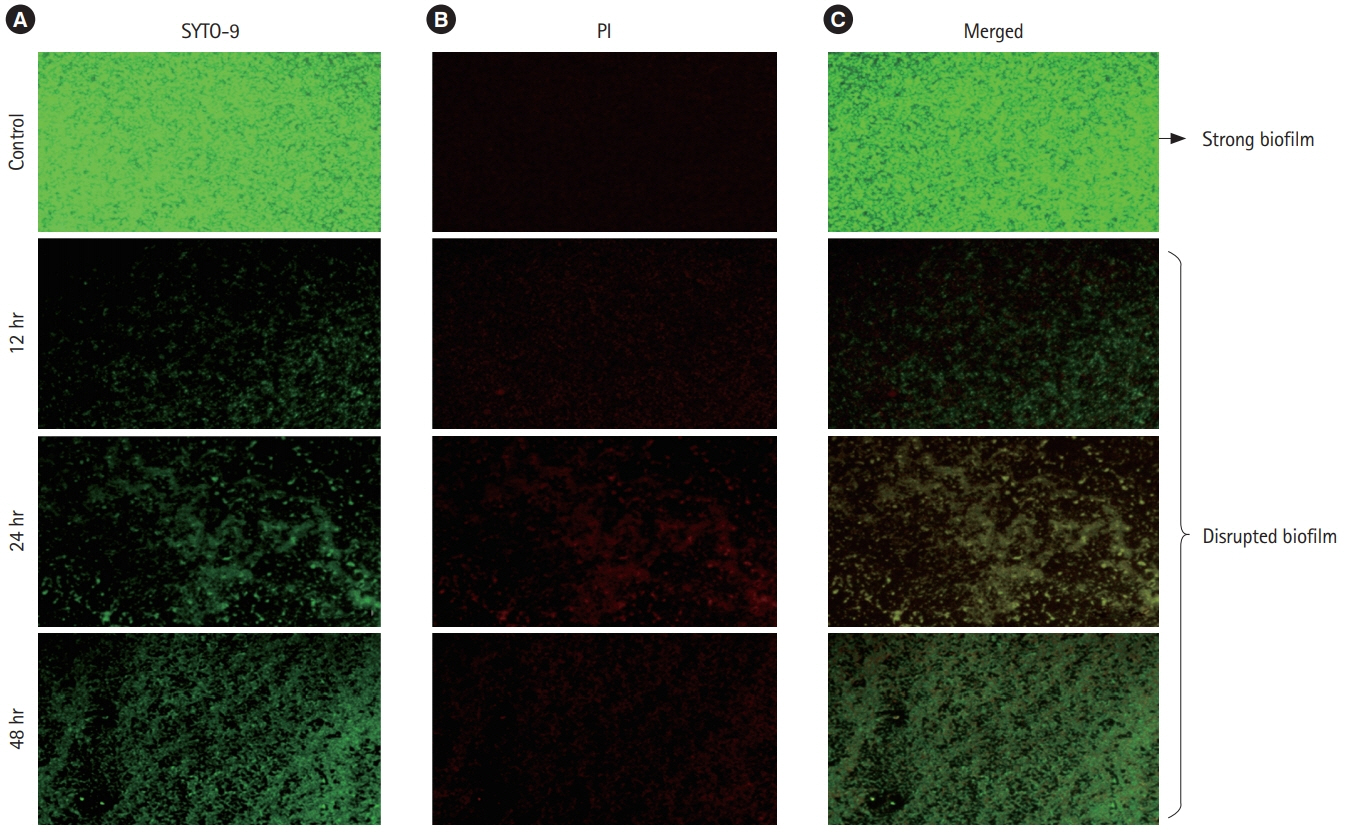
A total of 6 probiotic strains were tested for their antibiotic susceptibility followed by antimicrobial activity using cell-free supernatant (CFS) against C. sakazakii. The inhibitory activity of CFS against biofilm formation by C. sakazakii was determined using standard crystal violet assay and microscopic observations.
RESULTS
All the probiotic strains were sensitive to ampicillin, tetracycline, vancomycin and carbenicillin whereas most of the strains were resistant to erythromycin and novobiocin. Four of the 6 probiotic derived CFS possessed antimicrobial activity against C. sakazakii at a level of 40 μL. A higher biofilm inhibitory activity (>80%) was observed at initial stages of biofilm formation with weaker activity during longer incubation upto 48 hours (50%-60%).
CONCLUSIONS
The study indicated the efficacy of isolated commercial probiotics strains as potential inhibitor of biofilm formation by C. sakazakii and could be further explored for novel bioactive molecules to limit the emerging infections of C. sakazakii.
Keyword
MeSH Terms
Figure
Reference
-
1. Kumar N, Raghavendra M, Tokas J, Singal HR. Flavor addition in dairy products: health benefits and risks. Watson RR, Cllier RJ, Preedy VR. Nutrients in dairy and their implications on health and disease. Cambridge: Academic Press;2018. p. 123–135.2. Rondanelli M, Faliva MA, Perna S, Giacosa A, Peroni G, Castellazzi AM. Using probiotics in clinical practice: where are we now? A review of existing meta-analyses. Gut Microbes. 2017; 8:521–543.
Article3. Sánchez B, Delgado S, Blanco-Míguez A, Lourenço A, Gueimonde M, Margolles A. Probiotics, gut microbiota, and their influence on host health and disease. Mol Nutr Food Res. 2017; 61:201600240.
Article4. Beuchat LR, Kim H, Gurtler JB, Lin LC, Ryu JH, Richards GM. Cronobacter sakazakii in foods and factors affecting its survival, growth, and inactivation. Int J Food Microbiol. 2009; 136:204–213.
Article5. Bowen AB, Braden CR. Invasive Enterobacter sakazakii disease in infants. Emerg Infect Dis. 2006; 12:1185–1189.6. Forsythe SJ. Enterobacter sakazakii and other bacteria in powdered infant milk formula. Matern Child Nutr. 2005; 1:44–50.
Article7. Umeda NS, de Filippis I, Forsythe SJ, Brandão ML. Phenotypic characterization of Cronobacter spp. strains isolated from foods and clinical specimens in Brazil. Food Res Int. 2017; 102:61–67.
Article8. Osaili T, Forsythe S. Desiccation resistance and persistence of Cronobacter species in infant formula. Int J Food Microbiol. 2009; 136:214–220.
Article9. Singh N, Goel G, Raghav M. Insights into virulence factors determining the pathogenicity of Cronobacter sakazakii. Virulence. 2015; 6:433–440.
Article10. Singh N, Patil A, Prabhune AA, Raghav M, Goel G. Diverse profiles of N-acyl-homoserine lactones in biofilm forming strains of Cronobacter sakazakii. Virulence. 2017; 8:275–281.
Article11. Singh N, Raghav M, Narula S, Tandon S, Goel G. Profiling of virulence determinants in Cronobacter sakazakii isolates from different plant and environmental commodities. Curr Microbiol. 2017; 74:560–565.
Article12. Collado MC, Isolauri E, Salminen S. Specific probiotic strains and their combinations counteract adhesion of Enterobacter sakazakii to intestinal mucus. FEMS Microbiol Lett. 2008; 285:58–64.
Article13. Sharma G, Prakash A. Susceptibility of Cronobacter sakazakii to plant products, antibiotics, and to lactic acid bacteria. Int J Nutr Pharmacol Neurol Dis. 2013; 3:263–268.
Article14. Clinical and Laboratory Standards Institute. Performance standards for antimicrobial susceptibility testing. 27th ed. CLSI supplement M100. Wayne: CLSI;2017.15. European Food Safety Authority (EFSA). Technical guidance: update of the criteria used in the assessment of bacterial resistance to antibiotics of human or veterinary importance. EFSA J. 2008; 6:732.16. Singh N, Patil A, Prabhune A, Goel G. Inhibition of quorumsensing-mediated biofilm formation in Cronobacter sakazakii strains. Microbiology. 2016; 162:1708–1714.
Article17. Iversen C, Forsythe S. Isolation of Enterobacter sakazakii and other Enterobacteriaceae from powdered infant formula milk and related products. Food Microbiol. 2004; 21:771–777.
Article18. Lehner A, Riedel K, Eberl L, Breeuwer P, Diep B, Stephan R. Biofilm formation, extracellular polysaccharide production, and cell-to-cell signaling in various Enterobacter sakazakii strains: aspects promoting environmental persistence. J Food Prot. 2005; 68:2287–2294.
Article19. Kim H, Ryu JH, Beuchat LR. Attachment of and biofilm formation by Enterobacter sakazakii on stainless steel and enteral feeding tubes. Appl Environ Microbiol. 2006; 72:5846–5856.
Article20. Georgieva R, Yocheva L, Tserovska L, et al. Antimicrobial activity and antibiotic susceptibility of Lactobacillus and Bifidobacterium spp. intended for use as starter and probiotic cultures. Biotechnol Biotechnol Equip. 2015; 29:84–91.
Article21. Neut C, Mahieux S, Dubreuil LJ. Antibiotic susceptibility of probiotic strains: is it reasonable to combine probiotics with antibiotics? Med Mal Infect. 2017; 47:477–483.
Article22. Shokri D, Khorasgani MR, Mohkam M, Fatemi SM, Ghasemi Y, Taheri-Kafrani A. The inhibition effect of Lactobacilli against growth and biofilm formation of Pseudomonas aeruginosa. Probiotics Antimicrob Proteins. 2018; 10:34–42.
Article23. Kivanc M, Yilmaz M, Cakir E. Isolation and identification of lactic acid bacteria from boza, and their microbial activity against several reporter strains. Turk J Biol. 2011; 35:313–324.24. Denkova RS, Yanakieva VB, Denkova ZR, Hristozova P. Inhibitory activity of the probiotic strain Bifidobacterium bifidum Bif. 4 of human origin against pathogens. J Food Packag Sci Tech Technol. 2013; (2):21–25.25. Hayes M, Barrett E, Ross RP, Fitzgerald GF, Hill C, Stanton C. Evaluation of an antimicrobial ingredient prepared from a Lactobacillus acidophilus casein fermentate against Enterobacter sakazakii. J Food Prot. 2009; 72:340–346.
Article26. Awaisheh SS, Al-Nabulsi AA, Osaili TM, Ibrahim S, Holley R. Inhibition of Cronobacter sakazakii by heat labile bacteriocins produced by probiotic LAB isolated from healthy infants. J Food Sci. 2013; 78:M1416–M1420.27. Kim DH, Jeong D, Song KY, Kang IB, Kim H, Seo KH. Culture supernatant produced by Lactobacillus kefiri from kefir inhibits the growth of Cronobacter sakazakii. J Dairy Res. 2018; 85:98–103.
Article28. Yi L, Dang Y, Wu J, et al. Purification and characterization of a novel bacteriocin produced by Lactobacillus crustorum MN047 isolated from koumiss from Xinjiang, China. J Dairy Sci. 2016; 99:7002–7015.
Article29. Drenkard E, Ausubel FM. Pseudomonas biofilm formation and antibiotic resistance are linked to phenotypic variation. Nature. 2002; 416:740–743.
Article30. François ZN, Marie KP, Noëlle TA, Emeric GW. Antimicrobial activity of a bacteriocin produced by Lactobacillus plantarum 29V and strain’s viability in palm kernel oil. Int J Nutr Food Sci. 2013; 2:102–108.
Article31. Rao KP, Chennappa G, Suraj U, Nagaraja H, Raj AP, Sreenivasa MY. Probiotic potential of lactobacillus strains isolated from sorghum-based traditional fermented food. Probiotics Antimicrob Proteins. 2015; 7:146–156.
Article32. Schaffer AC, Lee JC. Vaccination and passive immunisation against Staphylococcus aureus. Int J Antimicrob Agents. 2008; 32 Suppl 1:S71–S78.
Article33. Saravanakumari P, Mani K. Structural characterization of a novel xylolipid biosurfactant from Lactococcus lactis and analysis of antibacterial activity against multi-drug resistant pathogens. Bioresour Technol. 2010; 101:8851–8854.
Article34. Shokouhfard M, Kermanshahi RK, Shahandashti RV, Feizabadi MM, Teimourian S. The inhibitory effect of a Lactobacillus acidophilus derived biosurfactant on biofilm producer Serratia marcescens. Iran J Basic Med Sci. 2015; 18:1001–1007.35. Sambanthamoorthy K, Feng X, Patel R, Patel S, Paranavitana C. Antimicrobial and antibiofilm potential of biosurfactants isolated from lactobacilli against multi-drug-resistant pathogens. BMC Microbiol. 2014; 14:197.
Article36. Candela M, Perna F, Carnevali P, et al. Interaction of probiotic Lactobacillus and Bifidobacterium strains with human intestinal epithelial cells: adhesion properties, competition against enteropathogens and modulation of IL-8 production. Int J Food Microbiol. 2008; 125:286–292.
Article37. Kim Y, Oh S, Kim SH. Released exopolysaccharide (r-EPS) produced from probiotic bacteria reduce biofilm formation of enterohemorrhagic Escherichia coli O157:H7. Biochem Biophys Res Commun. 2009; 379:324–329.
Article38. Bernal P, Llamas MA. Promising biotechnological applications of antibiofilm exopolysaccharides. Microb Biotechnol. 2012; 5:670–673.
Article39. Patel S, Majumder A, Goyal A. Potentials of exopolysaccharides from lactic acid bacteria. Indian J Microbiol. 2012; 52:3–12.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Cronobacter sakazakii Infection Induced Fatal Clinical Sequels Including Meningitis in Neonatal ICR Mice
- Effects of Lactobacillus casei and Aggregatibactor actinomycetemcomitans against Streptococcus mutans according to the Concentration of Sucrose
- A Case of Enterobacter sakazakii Epidural Abscess in Neonate
- Virulence genes of Streptococcus mutans and dental caries
- Inhibition of biofilm formation of periodontal pathogens by D-Arabinose