Int J Thyroidol.
2018 Nov;11(2):152-159. 10.11106/ijt.2018.11.2.152.
Expression of Sodium-Iodide Symporter Depending on Mutational Status and Lymphocytic Thyroiditis in Papillary Thyroid Carcinoma
- Affiliations
-
- 1Department of Internal Medicine, Seoul National University Hospital, Seoul National University College of Medicine, Seoul, Korea. yjparkmd@snu.ac.kr
- KMID: 2448986
- DOI: http://doi.org/10.11106/ijt.2018.11.2.152
Abstract
- BACKGROUND AND OBJECTIVES
Sodium-iodine symporter (NIS) is a marker for the degree of differentiation in thyroid cancer. The genetic factors or microenvironment surrounding tumors can affect transcription of NIS. In this study, we investigated the NIS mRNA expression according to mutational status and coexistent lymphocytic thyroiditis in papillary thyroid cancer (PTC).
MATERIALS AND METHODS
The RNA expression levels of NIS in the samples from database of The Caner Genome Atlas (TCGA; n=494) and our institute (n=125) were analyzed.
RESULTS
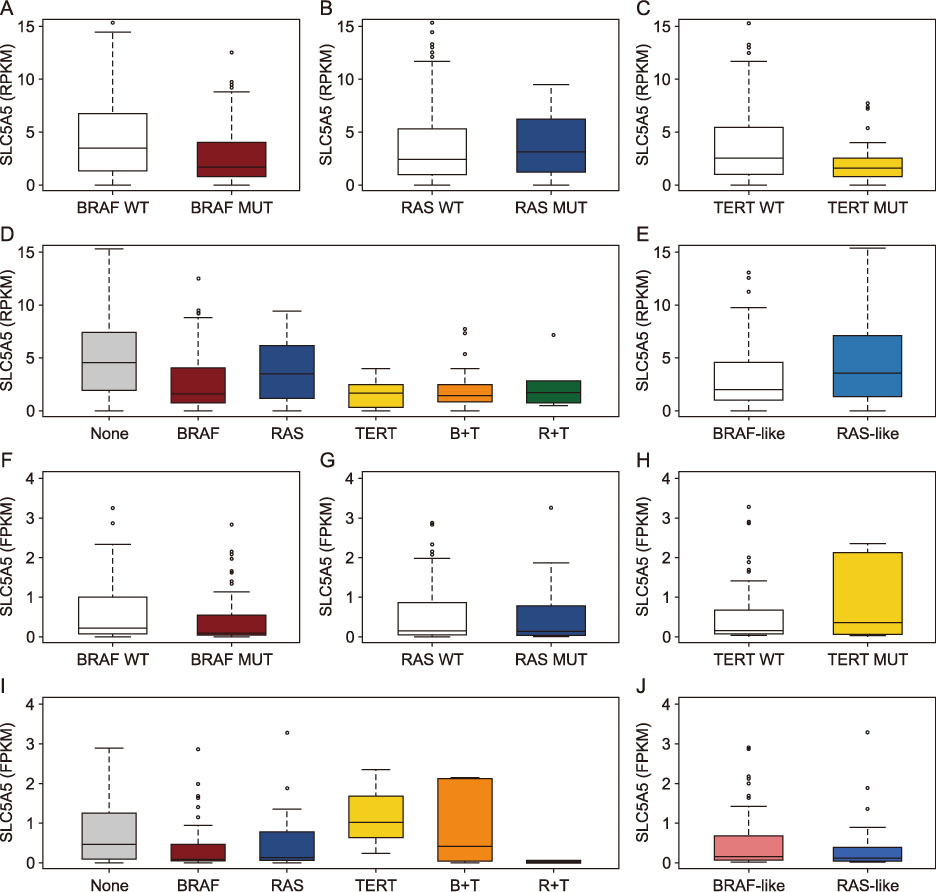
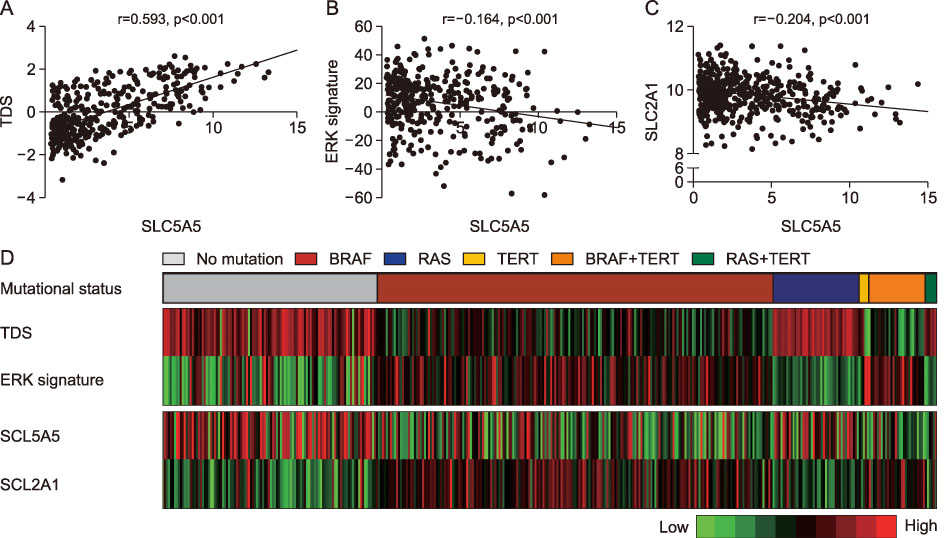
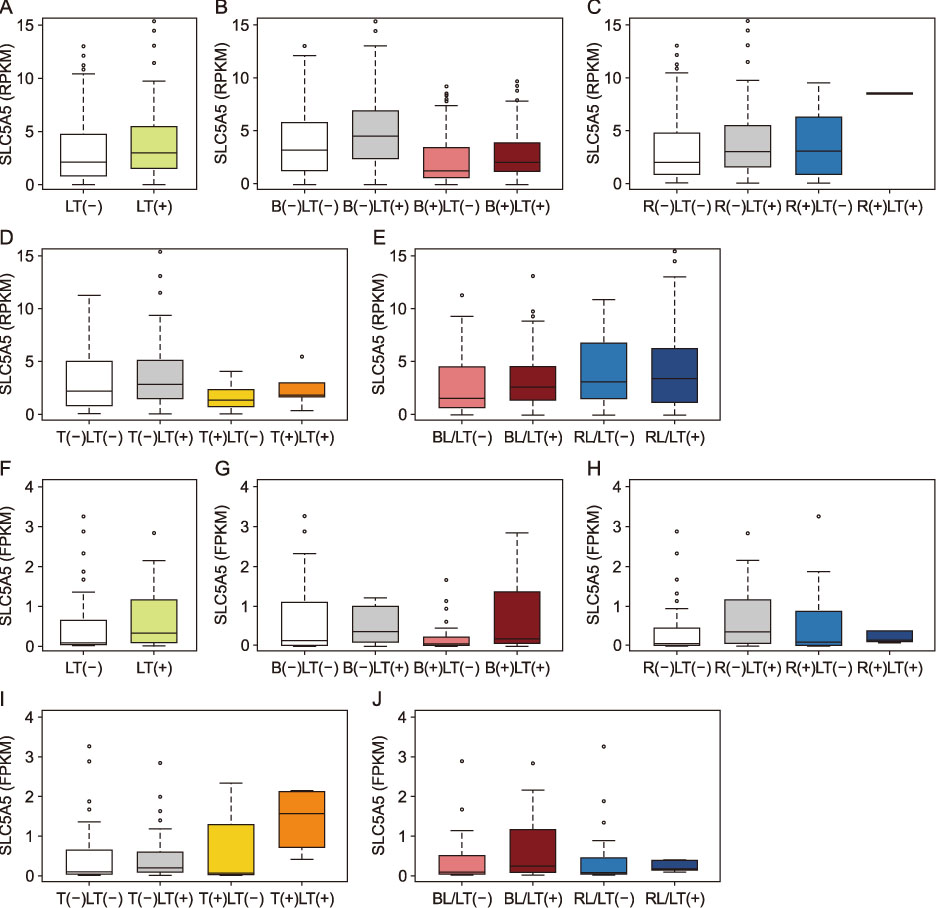
The PTCs with the BRAFV600E mutation and the coexistence of BRAFV600E and TERT promoter mutations showed significantly lower expression of NIS (p < 0.001, respectively), and those with BRAF-like molecular subtype also had reduced expression of NIS (p < 0.001). NIS expression showed a positive correlation with thyroid differentiation score (r=0.593, p < 0.001) and negative correlations with expressions of genes involved in ERK signaling (r=−0.164, p < 0.001) and GLUT-1 gene (r=−0.204, p < 0.001). The PTCs with lymphocytic thyroiditis showed significantly higher NIS expression (p=0.013), regardless of mutational status.
CONCLUSION
The NIS expression was reduced by the BRAFV600E mutation and MAPK/ERK pathway activation, but restored by the presence of lymphocytic thyroiditis.
Keyword
MeSH Terms
Figure
Reference
-
1. Schlumberger M, Lacroix L, Russo D, Filetti S, Bidart JM. Defects in iodide metabolism in thyroid cancer and implications for the follow-up and treatment of patients. Nat Clin Pract Endocrinol Metab. 2007; 3(3):260–269.
Article2. Spitzweg C, Bible KC, Hofbauer LC, Morris JC. Advanced radioiodine-refractory differentiated thyroid cancer: the sodium iodide symporter and other emerging therapeutic targets. Lancet Diabetes Endocrinol. 2014; 2(10):830–842.
Article3. Cancer Genome Atlas Research Network. Integrated genomic characterization of papillary thyroid carcinoma. Cell. 2014; 159(3):676–690.4. Yoo SK, Lee S, Kim SJ, Jee HG, Kim BA, Cho H, et al. Comprehensive analysis of the transcriptional and mutational landscape of follicular and papillary thyroid cancers. PLoS Genet. 2016; 12(8):e1006239.
Article5. Song YS, Lim JA, Choi H, Won JK, Moon JH, Cho SW, et al. Prognostic effects of TERT promoter mutations are enhanced by coexistence with BRAF or RAS mutations and strengthen the risk prediction by the ATA or TNM staging system in differentiated thyroid cancer patients. Cancer. 2016; 122(9):1370–1379.
Article6. Lazar V, Bidart JM, Caillou B, Mahe C, Lacroix L, Filetti S, et al. Expression of the Na+/I− symporter gene in human thyroid tumors: a comparison study with other thyroid-specific genes. J Clin Endocrinol Metab. 1999; 84(9):3228–3234.
Article7. Durante C, Puxeddu E, Ferretti E, Morisi R, Moretti S, Bruno R, et al. BRAF mutations in papillary thyroid carcinomas inhibit genes involved in iodine metabolism. J Clin Endocrinol Metab. 2007; 92(7):2840–2843.
Article8. Kim S, Chung JK, Min HS, Kang JH, Park DJ, Jeong JM, et al. Expression patterns of glucose transporter-1 gene and thyroid specific genes in human papillary thyroid carcinoma. Nucl Med Mol Imaging. 2014; 48(2):91–97.
Article9. Dong H, Shen WZ, Yan YJ, Yi JL, Zhang L. Effects of BRAF(V600E) mutation on Na(+)/I(−) symporter expression in papillary thyroid carcinoma. J Huazhong Univ Sci Technolog Med Sci. 2016; 36(1):77–81.
Article10. Chakravarty D, Santos E, Ryder M, Knauf JA, Liao XH, West BL, et al. Small-molecule MAPK inhibitors restore radioiodine incorporation in mouse thyroid cancers with conditional BRAF activation. J Clin Invest. 2011; 121(12):4700–4711.
Article11. Ricarte-Filho JC, Ryder M, Chitale DA, Rivera M, Heguy A, Ladanyi M, et al. Mutational profile of advanced primary and metastatic radioactive iodine-refractory thyroid cancers reveals distinct pathogenetic roles for BRAF, PIK3CA, and AKT1. Cancer Res. 2009; 69(11):4885–4893.
Article12. Ho AL, Grewal RK, Leboeuf R, Sherman EJ, Pfister DG, Deandreis D, et al. Selumetinib-enhanced radioiodine uptake in advanced thyroid cancer. N Engl J Med. 2013; 368(7):623–632.
Article13. Rothenberg SM, Daniels GH, Wirth LJ. Redifferentiation of iodine-refractory BRAF V600E-mutant metastatic papillary thyroid cancer with dabrafenib-response. Clin Cancer Res. 2015; 21(24):5640–5641.
Article14. Wang W, Larson SM, Tuttle RM, Kalaigian H, Kolbert K, Sonenberg M, et al. Resistance of [18f]-fluorodeoxyglucose-avid metastatic thyroid cancer lesions to treatment with high-dose radioactive iodine. Thyroid. 2001; 11(12):1169–1175.
Article15. Robbins RJ, Wan Q, Grewal RK, Reibke R, Gonen M, Strauss HW, et al. Real-time prognosis for metastatic thyroid carcinoma based on 2-[18F]fluoro-2-deoxy-D-glucose-positron emission tomography scanning. J Clin Endocrinol Metab. 2006; 91(2):498–505.
Article16. Feine U, Lietzenmayer R, Hanke JP, Held J, Wohrle H, Muller-Schauenburg W. Fluorine-18-FDG and iodine-131-iodide uptake in thyroid cancer. J Nucl Med. 1996; 37(9):1468–1472.17. Loh KC, Greenspan FS, Dong F, Miller TR, Yeo PP. Influence of lymphocytic thyroiditis on the prognostic outcome of patients with papillary thyroid carcinoma. J Clin Endocrinol Metab. 1999; 84(2):458–463.
Article18. Kim EY, Kim WG, Kim WB, Kim TY, Kim JM, Ryu JS, et al. Coexistence of chronic lymphocytic thyroiditis is associated with lower recurrence rates in patients with papillary thyroid carcinoma. Clin Endocrinol (Oxf). 2009; 71(4):581–586.
Article19. Lang BH, Chai YJ, Cowling BJ, Min HS, Lee KE, Youn YK. Is BRAFV600E mutation a marker for central nodal metastasis in small papillary thyroid carcinoma? Endocr Relat Cancer. 2014; 21(2):285–295.
Article20. Jeong JS, Kim HK, Lee CR, Park S, Park JH, Kang SW, et al. Coexistence of chronic lymphocytic thyroiditis with papillary thyroid carcinoma: clinical manifestation and prognostic outcome. J Korean Med Sci. 2012; 27(8):883–889.
Article21. Lee JH, Kim Y, Choi JW, Kim YS. The association between papillary thyroid carcinoma and histologically proven Hashimoto's thyroiditis: a meta-analysis. Eur J Endocrinol. 2013; 168(3):343–349.
Article22. Marotta V, Guerra A, Zatelli MC, Uberti ED, Di Stasi V, Faggiano A, et al. BRAF mutation positive papillary thyroid carcinoma is less advanced when Hashimoto's thyroiditis lymphocytic infiltration is present. Clin Endocrinol (Oxf). 2013; 79(5):733–738.23. Kim SK, Woo JW, Lee JH, Park I, Choe JH, Kim JH, et al. Chronic lymphocytic thyroiditis and BRAF V600E in papillary thyroid carcinoma. Endocr Relat Cancer. 2016; 23(1):27–34.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Sodium Iodide Symporter (NIS) in the Management of Patients with Thyroid Carcinoma
- Relationship between The Expressions of Sodium/Iodide Symporter and The Findings of Thallium-201 Scan in Thyroid Nodules
- Expression of Human Sodium Iodide Symporter mRNA in Papillary Thyroid Carcinoma
- Expression of Sodium-Iodide Symporter (NIS) in Thyroid Nodules: Comparison of RT-PCR and Immunohistochemical Staining Methods
- Comparison of Na+/I- Symporter Expression Rate in Malignant and Benign Thyroid Diseases: Immunohistochemical Study