Dement Neurocogn Disord.
2018 Jun;17(2):57-65. 10.12779/dnd.2018.17.2.57.
Gait Patterns in Parkinson's Disease with or without Cognitive Impairment
- Affiliations
-
- 1Department of Neurology, Veterans Health Service Medical Center, Seoul, Korea. kh21762@hanmail.net
- 2Department of Physical Medicine and Rehabilitation, Veterans Health Service Medical Center, Seoul, Korea.
- KMID: 2442809
- DOI: http://doi.org/10.12779/dnd.2018.17.2.57
Abstract
- BACKGROUND AND PURPOSE
Cognitive and gait disturbance are common symptoms in Parkinson's disease (PD). Although the relationship between cognitive impairment and gait dysfunction in PD has been suggested, specific gait patterns according to cognition are not fully demonstrated yet. Therefore, the aim of this study was to investigate gait patterns in PD patients with or without cognitive impairment.
METHODS
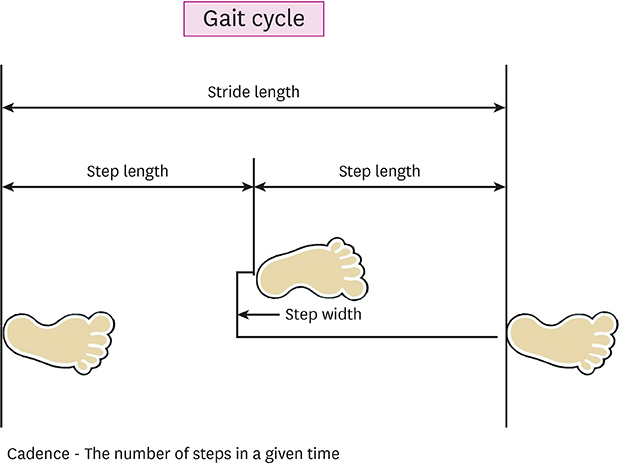
We studied 86 patients at an average of 4.8 years after diagnosis of PD. Cognitive impairment was defined as scoring 1.5 standard deviation below age- and education-specific means on the Korean version of the Mini-Mental State Examination (K-MMSE). Three-dimensional gait analysis was conducted for all patients and quantified gait parameters of temporal-spatial data were used. Relationships among cognition, demographic characteristics, clinical features, and gait pattern were evaluated.
RESULTS
Cognitive impairment was observed in 41 (47.7%) patients. Compared to patients without cognitive impairment, patients with cognitive impairment displayed reduced gait speed, step length, and stride length. Among K-MMSE subcategories, "registration,""attention/calculation," and "visuospatial function" were significantly associated with speed, step length, and stride length. However, age, disease duration, Hoehn-Yahr (HY) stage, or Unified Parkinson's Disease Rating Scale (UPDRS) motor score was not significantly related to any gait analysis parameter.
CONCLUSIONS
Our present study shows that cognitive impairment is associated with slow and short-stepped gait regardless of HY stage or UPDRS motor score, suggesting that cognitive impairment may serve as a surrogate marker of gait disturbance or fall in PD patients.
Keyword
Figure
Reference
-
1. Morris ME, Iansek R, Matyas TA, Summers JJ. The pathogenesis of gait hypokinesia in Parkinson's disease. Brain. 1994; 117:1169–1181.
Article2. Blin O, Ferrandez AM, Serratrice G. Quantitative analysis of gait in Parkinson patients: increased variability of stride length. J Neurol Sci. 1990; 98:91–97.
Article3. Allen NE, Schwarzel AK, Canning CG. Recurrent falls in Parkinson's disease: a systematic review. Parkinsons Dis. 2013; 2013:906274.
Article4. Amboni M, Barone P, Hausdorff JM. Cognitive contributions to gait and falls: evidence and implications. Mov Disord. 2013; 28:1520–1533.
Article5. van Iersel MB, Hoefsloot W, Munneke M, Bloem BR, Olde Rikkert MG. Systematic review of quantitative clinical gait analysis in patients with dementia. Z Gerontol Geriatr. 2004; 37:27–32.
Article6. Abe N, Mori E. Cognitive impairment in patients with Parkinson disease. Brain Nerve. 2012; 64:321–331.7. Williams-Gray CH, Evans JR, Goris A, Foltynie T, Ban M, Robbins TW, et al. The distinct cognitive syndromes of Parkinson's disease: 5 year follow-up of the CamPaIGN cohort. Brain. 2009; 132:2958–2969.
Article8. Hausdorff JM, Doniger GM, Springer S, Yogev G, Simon ES, Giladi N. A common cognitive profile in elderly fallers and in patients with Parkinson's disease: the prominence of impaired executive function and attention. Exp Aging Res. 2006; 32:411–429.
Article9. Mak MK, Wong A, Pang MY. Impaired executive function can predict recurrent falls in Parkinson's disease. Arch Phys Med Rehabil. 2014; 95:2390–2395.
Article10. Maetzler W, Liepelt I, Berg D. Progression of Parkinson's disease in the clinical phase: potential markers. Lancet Neurol. 2009; 8:1158–1171.
Article11. Schache AG, Baker R, Vaughan CL. Differences in lower limb transverse plane joint moments during gait when expressed in two alternative reference frames. J Biomech. 2007; 40:9–19.
Article12. Kang YW. A normative study of the Korean-Mini Mental State Examination (K-MMSE) in the elderly. Korean J Psychol Gen. 2006; 25:1–12.13. Yogev-Seligmann G, Hausdorff JM, Giladi N. The role of executive function and attention in gait. Mov Disord. 2008; 23:329–342.
Article14. Amboni M, Barone P, Iuppariello L, Lista I, Tranfaglia R, Fasano A, et al. Gait patterns in Parkinsonian patients with or without mild cognitive impairment. Mov Disord. 2012; 27:1536–1543.
Article15. Kelly VE, Johnson CO, McGough EL, Shumway-Cook A, Horak FB, Chung KA, et al. Association of cognitive domains with postural instability/gait disturbance in Parkinson's disease. Parkinsonism Relat Disord. 2015; 21:692–697.
Article16. Verbaan D, Marinus J, Visser M, van Rooden SM, Stiggelbout AM, Middelkoop HA, et al. Cognitive impairment in Parkinson's disease. J Neurol Neurosurg Psychiatry. 2007; 78:1182–1187.
Article17. Poletti M, Frosini D, Pagni C, Baldacci F, Nicoletti V, Tognoni G, et al. Mild cognitive impairment and cognitive-motor relationships in newly diagnosed drug-naive patients with Parkinson's disease. J Neurol Neurosurg Psychiatry. 2012; 83:601–606.
Article18. Williams-Gray CH, Foltynie T, Brayne CE, Robbins TW, Barker RA. Evolution of cognitive dysfunction in an incident Parkinson's disease cohort. Brain. 2007; 130:1787–1798.
Article19. Domellöf ME, Elgh E, Forsgren L. The relation between cognition and motor dysfunction in drug-naive newly diagnosed patients with Parkinson's disease. Mov Disord. 2011; 26:2183–2189.
Article20. Braak H, Del Tredici K. Invited article: nervous system pathology in sporadic Parkinson disease. Neurology. 2008; 70:1916–1925.
Article21. Kehagia AA, Barker RA, Robbins TW. Neuropsychological and clinical heterogeneity of cognitive impairment and dementia in patients with Parkinson's disease. Lancet Neurol. 2010; 9:1200–1213.
Article22. Blin O, Ferrandez AM, Pailhous J, Serratrice G. Dopa-sensitive and dopa-resistant gait parameters in Parkinson's disease. J Neurol Sci. 1991; 103:51–54.
Article23. Rochester L, Baker K, Nieuwboer A, Burn D. Targeting dopa-sensitive and dopa-resistant gait dysfunction in Parkinson's disease: selective responses to internal and external cues. Mov Disord. 2011; 26:430–435.
Article24. Stolze H, Klebe S, Zechlin C, Baecker C, Friege L, Deuschl G. Falls in frequent neurological diseases--prevalence, risk factors and aetiology. J Neurol. 2004; 251:79–84.25. Mak MK. Reduced step length, not step length variability is central to gait hypokinesia in people with Parkinson's disease. Clin Neurol Neurosurg. 2013; 115:587–590.
Article26. Verghese J, Wang C, Lipton RB, Holtzer R, Xue X. Quantitative gait dysfunction and risk of cognitive decline and dementia. J Neurol Neurosurg Psychiatry. 2007; 78:929–935.
Article27. Verghese J, Robbins M, Holtzer R, Zimmerman M, Wang C, Xue X, et al. Gait dysfunction in mild cognitive impairment syndromes. J Am Geriatr Soc. 2008; 56:1244–1251.
Article28. Lord S, Rochester L, Hetherington V, Allcock LM, Burn D. Executive dysfunction and attention contribute to gait interference in ‘off’ state Parkinson's disease. Gait Posture. 2010; 31:169–174.
Article29. Hausdorff JM, Balash J, Giladi N. Effects of cognitive challenge on gait variability in patients with Parkinson's disease. J Geriatr Psychiatry Neurol. 2003; 16:53–58.
Article30. Brauer SG, Woollacott MH, Lamont R, Clewett S, O'Sullivan J, Silburn P, et al. Single and dual task gait training in people with Parkinson's disease: a protocol for a randomised controlled trial. BMC Neurol. 2011; 11:90.
Article31. Almeida QJ, Lebold CA. Freezing of gait in Parkinson's disease: a perceptual cause for a motor impairment? J Neurol Neurosurg Psychiatry. 2010; 81:513–518.
Article32. Cowie D, Limousin P, Peters A, Day BL. Insights into the neural control of locomotion from walking through doorways in Parkinson's disease. Neuropsychologia. 2010; 48:2750–2757.
Article33. Rochester L, Hetherington V, Jones D, Nieuwboer A, Willems AM, Kwakkel G, et al. Attending to the task: interference effects of functional tasks on walking in Parkinson's disease and the roles of cognition, depression, fatigue, and balance. Arch Phys Med Rehabil. 2004; 85:1578–1585.
Article34. van der Marck MA, Klok MP, Okun MS, Giladi N, Munneke M, Bloem BR, et al. Consensus-based clinical practice recommendations for the examination and management of falls in patients with Parkinson's disease. Parkinsonism Relat Disord. 2014; 20:360–369.
Article35. Chung KA, Lobb BM, Nutt JG, Horak FB. Effects of a central cholinesterase inhibitor on reducing falls in Parkinson disease. Neurology. 2010; 75:1263–1269.
Article36. Henderson EJ, Lord SR, Brodie MA, Gaunt DM, Lawrence AD, Close JC, et al. Rivastigmine for gait stability in patients with Parkinson's disease (ReSPonD): a randomised, double-blind, placebo-controlled, phase 2 trial. Lancet Neurol. 2016; 15:249–258.
Article37. Kucinski A, Sarter M. Modeling Parkinson's disease falls associated with brainstem cholinergic systems decline. Behav Neurosci. 2015; 129:96–104.
Article38. Chou KL, Amick MM, Brandt J, Camicioli R, Frei K, Gitelman D, et al. A recommended scale for cognitive screening in clinical trials of Parkinson's disease. Mov Disord. 2010; 25:2501–2507.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- The Pattern of Cognitive Impairment Associated with the Motor Subtype in Parkinson's Disease
- The Association of Gait Disturbance and Cognitive Impairment
- Constipation is Associated With Mild Cognitive Impairment in Patients With de novo Parkinson’s Disease
- Cognitive Impairment in Parkinson's Disease without Dementia: Subtypes and Influences of Age
- Characteristice of cognitive impairment in parkinson's disease