Korean J Radiol.
2019 May;20(5):830-843. 10.3348/kjr.2018.0757.
Evaluation of Renal Pathophysiological Processes Induced by an Iodinated Contrast Agent in a Diabetic Rabbit Model Using Intravoxel Incoherent Motion and Blood Oxygenation Level-Dependent Magnetic Resonance Imaging
- Affiliations
-
- 1Department of Radiology, First Hospital of China Medical University, Shenyang, China. renke815@sina.com
- 2Department of Radiology, First Hospital of Shanxi Medical University, Taiyuan, China.
- 3Department of Radiology, The Qianfoshan Hospital of Shandong Province, Jinan, China.
- 4Cardiac Surgery, First Hospital of China Medical University, Shenyang, China.
- KMID: 2442717
- DOI: http://doi.org/10.3348/kjr.2018.0757
Abstract
OBJECTIVE
To examine the potential of intravoxel incoherent motion (IVIM) and blood oxygen level-dependent (BOLD) magnetic resonance imaging for detecting renal changes after iodinated contrast-induced acute kidney injury (CI-AKI) development in a diabetic rabbit model.
MATERIALS AND METHODS
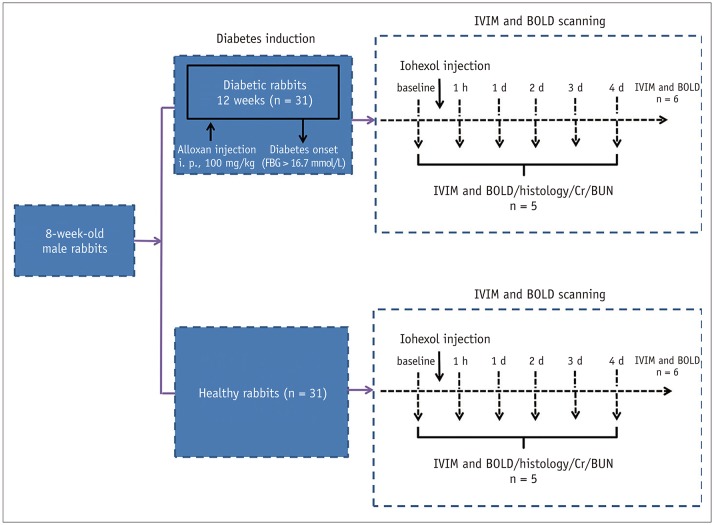
Sixty-two rabbits were randomized into 2 groups: diabetic rabbits with the contrast agent (DCA) and healthy rabbits with the contrast agent (NCA). In each group, 6 rabbits underwent IVIM and BOLD imaging at 1 hour, 1 day, 2 days, 3 days, and 4 days after an iohexol injection while 5 rabbits were selected to undergo blood and histological examinations at these specific time points. Iohexol was administrated at a dose of 2.5 g I/kg of body weight. Further, the apparent transverse relaxation rate (R2*), average pure molecular diffusion coefficient (D), pseudo-diffusion coefficient (D*), and perfusion fraction (f) were calculated.
RESULTS
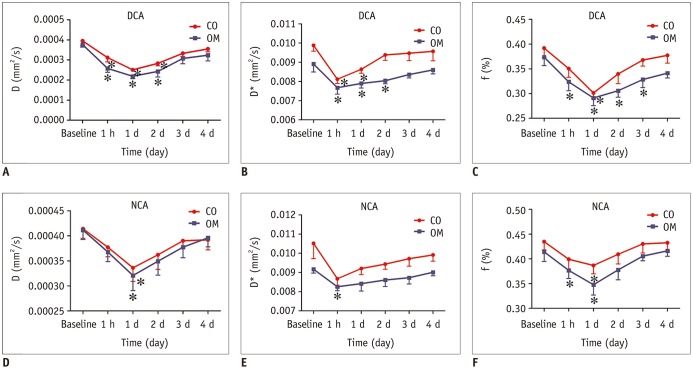
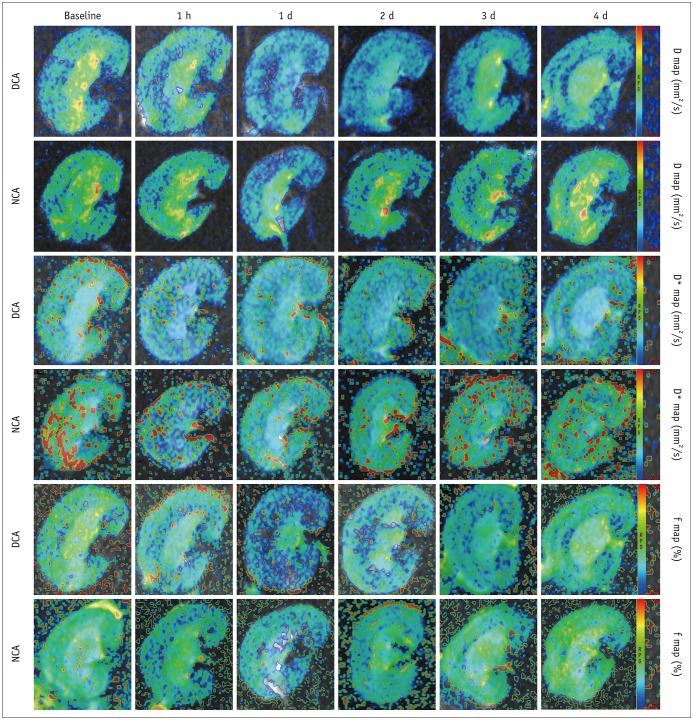
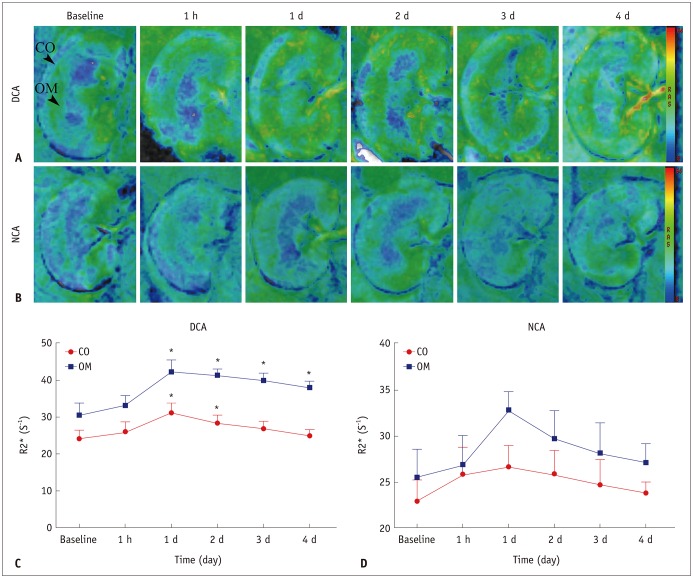
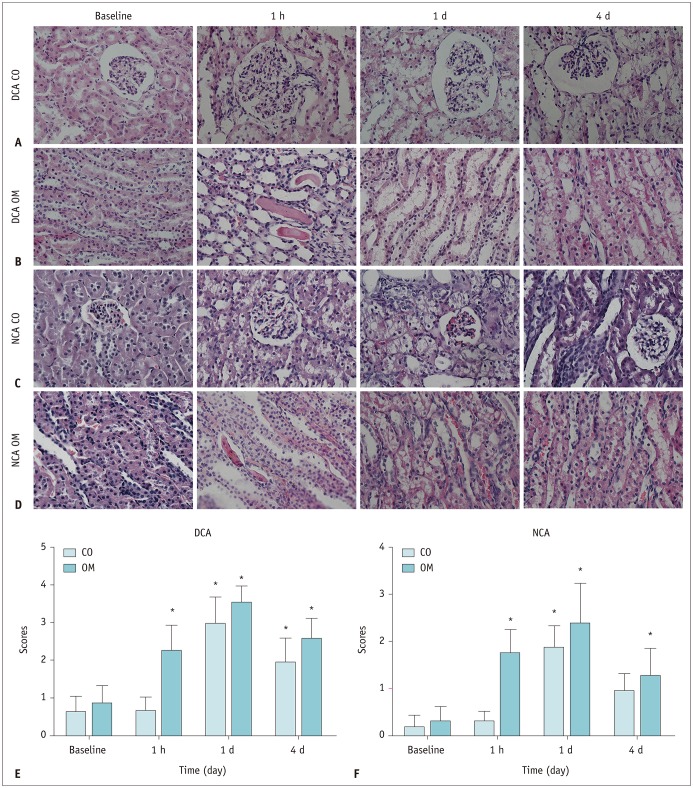
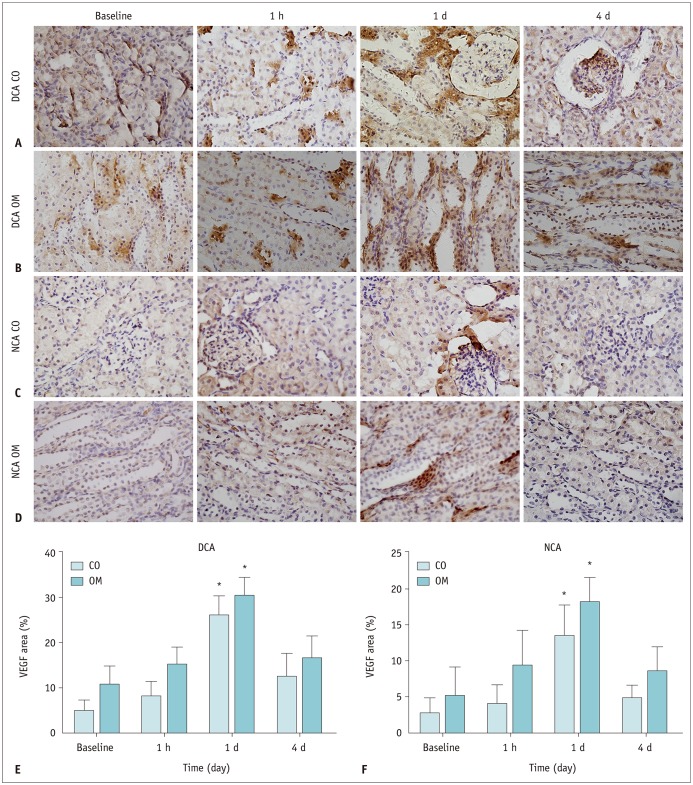
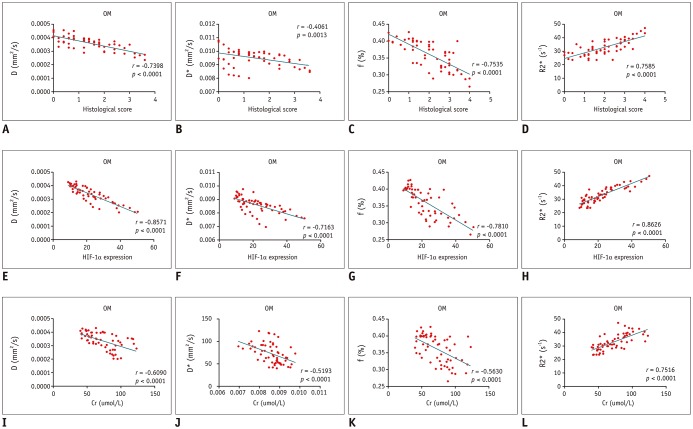
The D and f values of the renal cortex (CO) and outer medulla (OM) were significantly decreased compared to baseline values in the 2 groups 1 day after the iohexol injection (p < 0.05). A marked reduction in the D* values for both the CO and OM was also observed after 1 hour in each group (p < 0.05). In the OM, a persistent elevation of the R2* was detected for 4 days in the DCA group (p < 0.05). Histopathological changes were prominent, and the pathological features of CI-AKI aggravated in the DCA group until day 4. The D, f, and R2* values significantly correlated with the histological damage scores, hypoxia-inducible transcription factor-1α expression scores, and serum creatinine levels.
CONCLUSION
A combination of IVIM and BOLD imaging may serve as a noninvasive method for detecting and monitoring CI-AKI in the early stages in the diabetic kidney.
Keyword
MeSH Terms
Figure
Reference
-
1. McCullough PA. Contrast-induced acute kidney injury. J Am Coll Cardiol. 2008; 51:1419–1428. PMID: 18402894.
Article2. Li LP, Lu J, Zhou Y, Papadopoulou MV, Franklin T, Bokhary U, et al. Evaluation of intrarenal oxygenation in iodinated contrast-induced acute kidney injury-susceptible rats by blood oxygen level-dependent magnetic resonance imaging. Invest Radiol. 2014; 49:403–410. PMID: 24566288.
Article3. Calvin AD, Misra S, Pflueger A. Contrast-induced acute kidney injury and diabetic nephropathy. Nat Rev Nephrol. 2010; 6:679–688. PMID: 20877303.
Article4. Stacul F, van der Molen AJ, Reimer P, Webb JA, Thomsen HS, Morcos SK, Contrast Media Safety Committee of European Society of Urogenital Radiology (ESUR), et al. Contrast induced nephropathy: updated ESUR Contrast Media Safety Committee guidelines. Eur Radiol. 2011; 21:2527–2541. PMID: 21866433.
Article5. Chalikias G, Drosos I, Tziakas DN. Contrast-induced acute kidney injury: an update. Cardiovasc Drugs Ther. 2016; 30:215–228. PMID: 26780748.
Article6. Kurnik BR, Allgren RL, Genter FC, Solomon RJ, Bates ER, Weisberg LS. Prospective study of atrial natriuretic peptide for the prevention of radiocontrast-induced nephropathy. Am J Kidney Dis. 1998; 31:674–680. PMID: 9531185.
Article7. Su J, Zou W, Cai W, Chen X, Wang F, Li S, et al. Atorvastatin ameliorates contrast medium-induced renal tubular cell apoptosis in diabetic rats via suppression of rho-kinase pathway. Eur J Pharmacol. 2014; 723:15–22. PMID: 24445019.
Article8. Li LP, Lu J, Franklin T, Zhou Y, Solomon R, Prasad PV. Effect of iodinated contrast medium in diabetic rat kidneys as evaluated by blood-oxygenation-level-dependent magnetic resonance imaging and urinary neutrophil gelatinase-associated lipocalin. Invest Radiol. 2015; 50:392–396. PMID: 25668748.
Article9. Inoue T, Kozawa E, Okada H, Inukai K, Watanabe S, Kikuta T, et al. Noninvasive evaluation of kidney hypoxia and fibrosis using magnetic resonance imaging. J Am Soc Nephrol. 2011; 22:1429–1434. PMID: 21757771.
Article10. dos Santos EA, Li LP, Ji L, Prasad PV. Early changes with diabetes in renal medullary hemodynamics as evaluated by fiberoptic probes and BOLD magnetic resonance imaging. Invest Radiol. 2007; 42:157–162. PMID: 17287645.
Article11. Hueper K, Hartung D, Gutberlet M, Gueler F, Sann H, Husen B, et al. Assessment of impaired vascular reactivity in a rat model of diabetic nephropathy: effect of nitric oxide synthesis inhibition on intrarenal diffusion and oxygenation measured by magnetic resonance imaging. Am J Physiol Renal Physiol. 2013; 305:F1428–F1435. PMID: 24005471.
Article12. Liang L, Chen WB, Chan KW, Li YG, Zhang B, Liang CH, et al. Using intravoxel incoherent motion MR imaging to study the renal pathophysiological process of contrast-induced acute kidney injury in rats: comparison with conventional DWI and arterial spin labelling. Eur Radiol. 2016; 26:1597–1605. PMID: 26373761.
Article13. Cai XR, Yu J, Zhou QC, Du B, Feng YZ, Liu XL. Use of intravoxel incoherent motion MRI to assess renal fibrosis in a rat model of unilateral ureteral obstruction. J Magn Reson Imaging. 2016; 44:698–706. PMID: 26841951.
Article14. Ichikawa S, Motosugi U, Ichikawa T, Sano K, Morisaka H, Araki T. Intravoxel incoherent motion imaging of the kidney: alterations in diffusion and perfusion in patients with renal dysfunction. Magn Reson Imaging. 2013; 31:414–417. PMID: 23102943.
Article15. Ding Y, Zeng M, Rao S, Chen C, Fu C, Zhou J. Comparison of biexponential and monoexponential model of diffusion-weighted imaging for distinguishing between common renal cell carcinoma and fat poor angiomyolipoma. Korean J Radiol. 2016; 17:853–863. PMID: 27833401.
Article16. Wang Y, Ren K, Liu Y, Sun WG, Wang JH, Zhang X, et al. Application of BOLD MRI and DTI for the evaluation of renal effect related to viscosity of iodinated contrast agent in a rat model. J Magn Reson Imaging. 2017; 46:1320–1331. PMID: 28248433.
Article17. Heyman SN, Rosen S, Rosenberger C. Renal parenchymal hypoxia, hypoxia adaptation, and the pathogenesis of radiocontrast nephropathy. Clin J Am Soc Nephrol. 2008; 3:288–296. PMID: 18057308.
Article18. Rosenberger C, Heyman SN, Rosen S, Shina A, Goldfarb M, Griethe W, et al. Up-regulation of HIF in experimental acute renal failure: evidence for a protective transcriptional response to hypoxia. Kidney Int. 2005; 67:531–542. PMID: 15673301.
Article19. Shukla R, Pandey N, Banerjee S, Tripathi YB. Effect of extract of Pueraria tuberosa on expression of hypoxia inducible factor-1α and vascular endothelial growth factor in kidney of diabetic rats. Biomed Pharmacother. 2017; 93:276–285. PMID: 28648975.
Article20. Manotham K, Tanaka T, Ohse T, Kojima I, Miyata T, Inagi R, et al. A biologic role of HIF-1 in the renal medulla. Kidney Int. 2005; 67:1428–1439. PMID: 15780095.
Article21. Rosenberger C, Griethe W, Gruber G, Wiesener M, Frei U, Bachmann S, et al. Cellular responses to hypoxia after renal segmental infarction. Kidney Int. 2003; 64:874–886. PMID: 12911537.
Article22. Zhao Q, Li J, Yan J, Liu S, Guo Y, Chen D, et al. Lycium barbarum polysaccharides ameliorates renal injury and inflammatory reaction in alloxan-induced diabetic nephropathy rabbits. Life Sci. 2016; 157:82–90. PMID: 27262790.
Article23. Papadimitriou A, Silva KC, Peixoto EB, Borges CM, Lopes de, Lopes de. Theobromine increases NAD+/Sirt-1 activity and protects the kidney under diabetic conditions. Am J Physiol Renal Physiol. 2015; 308:F209–F225. PMID: 25411384.24. Pettersson G, Towart R, Grant D, Thyberg K, Golman K. The rabbit renal toxicity test: a sensitive in vivo test for the nephrotoxicity of contrast agents. Acad Radiol. 2002; 9(Suppl 1):S62–S64. PMID: 12019897.25. Lauver DA, Carey EG, Bergin IL, Lucchesi BR, Gurm HS. Sildenafil citrate for prophylaxis of nephropathy in an animal model of contrast-induced acute kidney injury. PLoS One. 2014; 9:e113598. PMID: 25426714.
Article26. Wang JH, Ren K, Sun WG, Zhao L, Zhong HS, Xu K. Effects of iodinated contrast agents on renal oxygenation level determined by blood oxygenation level dependent magnetic resonance imaging in rabbit models of type 1 and type 2 diabetic nephropathy. BMC Nephrol. 2014; 15:140. PMID: 25182068.
Article27. Vivier PH, Storey P, Chandarana H, Yamamoto A, Tantillo K, Khan U, et al. Renal blood oxygenation level–dependent imaging contribution of R2 to R2* values. Invest Radiol. 2013; 48:501–508. PMID: 23385400.28. Le Bihan D, Breton E, Lallemand D, Aubin ML, Vignaud J, Laval-Jeantet M. Separation of diffusion and perfusion in intravoxel incoherent motion MR imaging. Radiology. 1988; 168:497–505. PMID: 3393671.
Article29. Tsamouri MM, Rapti M, Kouka P, Nepka C, Tsarouhas K, Soumelidis A, et al. Histopathological evaluation and redox assessment in blood and kidney tissues in a rabbit contrast-induced nephrotoxicity model. Food Chem Toxicol. 2017; 108(Pt A):186–193. PMID: 28774741.
Article30. Ari E, Kedrah AE, Alahdab Y, Bulut G, Eren Z, Baytekin O, et al. Antioxidant and renoprotective effects of paricalcitol on experimental contrast-induced nephropathy model. Br J Radiol. 2012; 85:1038–1043. PMID: 22815410.
Article31. Wang Y, Ren K, Xie L, Sun W, Liu Y, Li S. Effect of repeated injection of iodixanol on renal function in healthy Wistar rats using functional MRI. Biomed Res Int. 2018; 2018:7272485. PMID: 29850557.
Article32. Ries M, Basseau F, Tyndal B, Jones R, Deminière C, Catargi B, et al. Renal diffusion and BOLD MRI in experimental diabetic nephropathy. Blood oxygen level-dependent. J Magn Reson Imaging. 2003; 17:104–113. PMID: 12500279.33. Cheung JS, Fan SJ, Gao DS, Chow AM, Man K, Wu EX. Diffusion tensor imaging of liver fibrosis in an experimental model. J Magn Reson Imaging. 2010; 32:1141–1148. PMID: 21031520.
Article34. Wang J, Zhang Y, Yang X, Wang X, Zhang J, Fang J, et al. Time course study on the effects of iodinated contrast medium on intrarenal water transport function using diffusion-weighted MRI. J Magn Reson Imaging. 2012; 35:1139–1144. PMID: 22247072.
Article35. Yan YY, Hartono S, Hennedige T, Koh TS, Chan CM, Zhou L, et al. Intravoxel incoherent motion and diffusion tensor imaging of early renal fibrosis induced in a murine model of streptozotocin induced diabetes. Magn Reson Imaging. 2017; 38:71–76. PMID: 28038964.
Article36. Nakagawa T, Sato W, Kosugi T, Johnson RJ. Uncoupling of VEGF with endothelial NO as a potential mechanism for abnormal angiogenesis in the diabetic nephropathy. J Diabetes Res. 2013; 2013:184539. PMID: 24386643.
Article37. Rauch D, Drescher P, Pereira FJ, Knes JM, Will JA, Madsen PO. Comparison of iodinated contrast media-induced renal vasoconstriction in human, rabbit, dog, and pig arteries. Invest Radiol. 1997; 32:315–319. PMID: 9179705.
Article38. Heyman SN, Rosenberger C, Rosen S, Khamaisi M. Why is diabetes mellitus a risk factor for contrast-induced nephropathy? Biomed Res Int. 2013; 2013:123589. PMID: 24350240.
Article39. Melin J, Hellberg O, Akyürek LM, Källskog O, Larsson E, Fellström BC. Ischemia causes rapidly progressive nephropathy in the diabetic rat. Kidney Int. 1997; 52:985–991. PMID: 9328937.
Article40. Goldfarb M, Rosenberger C, Abassi Z, Shina A, Zilbersat F, Eckardt KU, et al. Acute-on-chronic renal failure in the rat: functional compensation and hypoxia tolerance. Am J Nephrol. 2006; 26:22–33. PMID: 16508244.
Article41. Prasad P, Li LP, Halter S, Cabray J, Ye M, Batlle D. Evaluation of renal hypoxia in diabetic mice by BOLD MRI. Invest Radiol. 2010; 45:819–822. PMID: 20829708.
Article42. Basile DP, Donohoe DL, Roethe K, Mattson DL. Chronic renal hypoxia after acute ischemic injury: effects of L-arginine on hypoxia and secondary damage. Am J Physiol Renal Physiol. 2003; 284:F338–F348. PMID: 12388385.43. Coca SG, Singanamala S, Parikh CR. Chronic kidney disease after acute kidney injury: a systematic review and meta-analysis. Kidney Int. 2012; 81:442–448. PMID: 22113526.
Article44. Fine LG, Orphanides C, Norman JT. Progressive renal disease: the chronic hypoxia hypothesis. Kidney Int Suppl. 1998; 65:S74–S78. PMID: 9551436.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Correlation between Intravoxel Incoherent Motion Magnetic Resonance Imaging Derived Metrics and Serum Soluble CD40 Ligand Level in an Embolic Canine Stroke Model
- RE: Distinguishing between Renal Cell Carcinoma and Fat Poor Angiomyolipoma in Diffusion-Weighted Imaging
- Bilateral Renal Artery Stenosis with Renal Insufficiency: Successful Angioplasty Using Gadopentetate Dimeglumine as a Contrast Agent
- Intravoxel Incoherent Motion MR Imaging in the Head and Neck: Correlation with Dynamic Contrast-Enhanced MR Imaging and Diffusion-Weighted Imaging
- B-Value Optimization in the Estimation of Intravoxel Incoherent Motion Parameters in Patients with Cervical Cancer