Ann Rehabil Med.
2018 Feb;42(1):8-17. 10.5535/arm.2018.42.1.8.
Crossed Cerebellar Diaschisis: Risk Factors and Correlation to Functional Recovery in Intracerebral Hemorrhage
- Affiliations
-
- 1Department of Rehabilitation Medicine, Wonkwang University School of Medicine & Hospital, Iksan, Korea. helmaine@naver.com
- 2Department of Nuclear Medicine, Wonkwang University School of Medicine & Hospital, Iksan, Korea.
- KMID: 2440964
- DOI: http://doi.org/10.5535/arm.2018.42.1.8
Abstract
OBJECTIVE
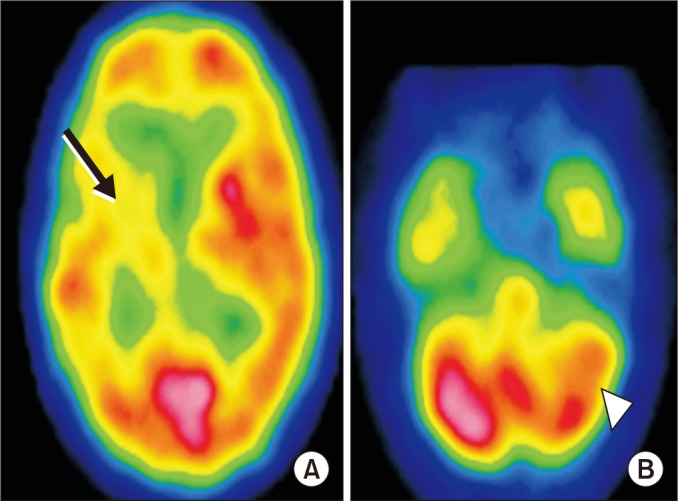
The purpose of this study is to investigate predictors of crossed cerebellar diaschisis (CCD), and the effects of CCD on functional outcomes including motor function, activities of daily living, cognitive function, and ambulation 6 months after onset in patients with intracerebral hemorrhage (ICH).
METHODS
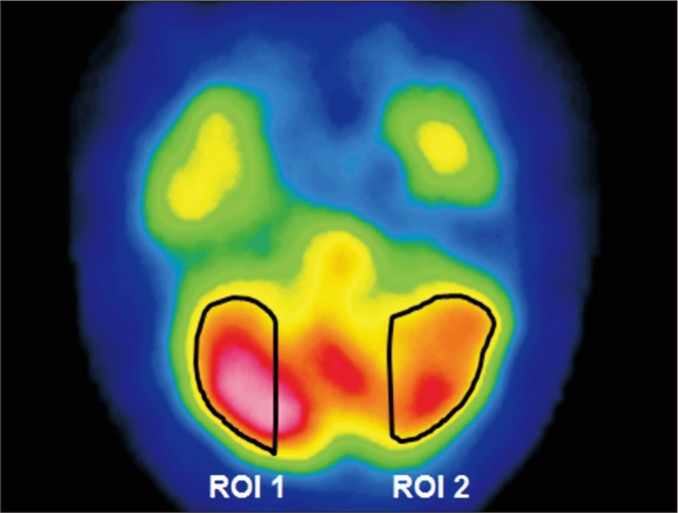
A total of 74 patients experiencing their first ICH were recruited. If the asymmetric index was more than 10% using single photon emission computed tomography (SPECT), a diagnosis of CCD was confirmed. Clinical factors were retrospectively assessed by reviewing medical records. Radiologic factors encompassed the concomitance of intraventricular hemorrhage, side and location of the lesion, and hemorrhage volume. Functional outcomes were evaluated using the Fugl-Meyer Assessment, the Korean version of the Mini-Mental State Examination, the Korean version of the Modified Barthel Index, and measurement of the Functional Ambulatory Category at the time of SPECT measurement and 6 months post-ICH.
RESULTS
Lesion location, especially in the basal ganglia (odds ratio [OR]=6.138, p=0.011), and hemorrhagic volume (OR=1.055, p=0.046) were independent predictors for CCD according to multivariate logistic regression analysis. In addition, the presence of CCD was significantly related to the improvement in Fugl-Meyer Assessment score after 6 months (adjusted R2=0.152, p=0.036).
CONCLUSION
Lesion location and hemorrhagic volume were the predisposing factors for CCD, and the CCD was associated with poor motor recovery over 6 months in patients with hemorrhagic stroke.
Keyword
MeSH Terms
Figure
Reference
-
1. Baron JC, Bousser MG, Comar D, Castaigne P. “Crossed cerebellar diaschisis” in human supratentorial brain infarction. Trans Am Neurol Assoc. 1981; 105:459–461. PMID: 19645126.2. Sommer WH, Bollwein C, Thierfelder KM, Baumann A, Janssen H, Ertl-Wagner B, et al. Crossed cerebellar diaschisis in patients with acute middle cerebral artery infarction: occurrence and perfusion characteristics. J Cereb Blood Flow Metab. 2016; 36:743–754. PMID: 26661242.
Article4. Tien RD, Ashdown BC. Crossed cerebellar diaschisis and crossed cerebellar atrophy: correlation of MR findings, clinical symptoms, and supratentorial diseases in 26 patients. AJR Am J Roentgenol. 1992; 158:1155–1159. PMID: 1566683.
Article5. Kim SE, Choi CW, Yoon BW, Chung JK, Roh JH, Lee MC, et al. Crossed-cerebellar diaschisis in cerebral infarction: technetium-99m-HMPAO SPECT and MRI. J Nucl Med. 1997; 38:14–19. PMID: 8998142.6. Di Piero V, Chollet F, Dolan RJ, Thomas DJ, Frackowiak R. The functional nature of cerebellar diaschisis. Stroke. 1990; 21:1365–1369. PMID: 2396276.
Article7. Engelborghs S, Pickut BA, Marien P, Opsomer F, De Deyn PP. Crossed cerebellar diaschisis and hemiataxia after thalamic hemorrhage. J Neurol. 2000; 247:476–477. PMID: 10929283.
Article8. Liu Y, Karonen JO, Nuutinen J, Vanninen E, Kuikka JT, Vanninen RL. Crossed cerebellar diaschisis in acute ischemic stroke: a study with serial SPECT and MRI. J Cereb Blood Flow Metab. 2007; 27:1724–1732. PMID: 17311077.
Article9. Sobesky J, Thiel A, Ghaemi M, Hilker RH, Rudolf J, Jacobs AH, et al. Crossed cerebellar diaschisis in acute human stroke: a PET study of serial changes and response to supratentorial reperfusion. J Cereb Blood Flow Metab. 2005; 25:1685–1691. PMID: 15931159.
Article10. Forster A, Kerl HU, Goerlitz J, Wenz H, Groden C. Crossed cerebellar diaschisis in acute isolated thalamic infarction detected by dynamic susceptibility contrast perfusion MRI. PLoS One. 2014; 9:e88044. PMID: 24505372.
Article11. Komaba Y, Mishina M, Utsumi K, Katayama Y, Kobayashi S, Mori O. Crossed cerebellar diaschisis in patients with cortical infarction: logistic regression analysis to control for confounding effects. Stroke. 2004; 35:472–476. PMID: 14739422.12. Chon JS, Chun SI, Yoo WK, Lee JD, Doh WS. Diaschisis and motor recovery in stroke patients. J Korean Acad Rehabil Med. 1998; 22:822–827.13. Szilagyi G, Vas A, Kerenyi L, Nagy Z, Csiba L, Gulyas B. Correlation between crossed cerebellar diaschisis and clinical neurological scales. Acta Neurol Scand. 2012; 125:373–381. PMID: 21781057.14. Infeld B, Davis SM, Lichtenstein M, Mitchell PJ, Hopper JL. Crossed cerebellar diaschisis and brain recovery after stroke. Stroke. 1995; 26:90–95. PMID: 7839405.
Article15. Laloux P, Richelle F, Jamart J, De Coster P, Laterre C. Comparative correlations of HMPAO SPECT indices, neurological score, and stroke subtypes with clinical outcome in acute carotid infarcts. Stroke. 1995; 26:816–821. PMID: 7740573.
Article16. Choi IS, Kim JH, Lee SY, Kang KJ, Kim JH, Lee SG. The correlation between crossed cerebellar diaschisis and motor impairment of hemiplegic upper extremity in stroke patients. J Korean Acad Rehabil Med. 2005; 29:15–22.17. Small SL, Hlustik P, Noll DC, Genovese C, Solodkin A. Cerebellar hemispheric activation ipsilateral to the paretic hand correlates with functional recovery after stroke. Brain. 2002; 125(Pt 7):1544–1557. PMID: 12077004.
Article18. Kandel ER, Schwartz JH, Jessell TM. Principles of neural science. 4th ed. New York: McGraw-Hill;2000.19. Chambers WW, Sprague JM. Functional localization in the cerebellum. II. Somatotopic organization in cortexand nuclei. AMA Arch Neurol Psychiatry. 1955; 74:653–680. PMID: 13268132.20. Chambers WW, Sprague JM. Functional localizationin the cerebellum. I. Organization in longitudinal cortico-nuclear zones and their contribution to the control of posture, both extrapyramidal and pyramidal. J Comp Neurol. 1955; 103:105–129. PMID: 13263445.21. Lim JS, Ryu YH, Kim BM, Lee JD. Crossed cerebellar diaschisis due to intracranial hematoma in basal ganglia or thalamus. J Nucl Med. 1998; 39:2044–2047. PMID: 9867139.22. Fazekas F, Payer F, Valetitsch H, Schmidt R, Flooh E. Brain stem infarction and diaschisis: a SPECT cerebral perfusion study. Stroke. 1993; 24:1162–1166. PMID: 8342190.
Article23. Lee M, Saver JL, Chang KH, Liao HW, Chang SC, Ovbiagele B. Low glomerular filtration rate and risk of stroke: meta-analysis. BMJ. 2010; 341:c4249. PMID: 20884696.
Article24. Kothari RU, Brott T, Broderick JP, Barsan WG, Sauerbeck LR, Zuccarello M, et al. The ABCs of measuring intracerebral hemorrhage volumes. Stroke. 1996; 27:1304–1305. PMID: 8711791.
Article25. Gladstone DJ, Danells CJ, Black SE. The Fugl-Meyer assessment of motor recovery after stroke: a critical review of its measurement properties. Neurorehabil Neural Repair. 2002; 16:232–240. PMID: 12234086.
Article26. Holden MK, Gill KM, Magliozzi MR, Nathan J, Piehl-Baker L. Clinical gait assessment in the neurologically impaired: reliability and meaningfulness. Phys Ther. 1984; 64:35–40. PMID: 6691052.27. Hendricks HT, van Limbeek J, Geurts AC, Zwarts MJ. Motor recovery after stroke: a systematic review of the literature. Arch Phys Med Rehabil. 2002; 83:1629–1637. PMID: 12422337.
Article28. Coupar F, Pollock A, Rowe P, Weir C, Langhorne P. Predictors of upper limb recovery after stroke: a systematic review and meta-analysis. Clin Rehabil. 2012; 26:291–313. PMID: 22023891.
Article29. Gottesman RF, Hillis AE. Predictors and assessment of cognitive dysfunction resulting from ischaemic stroke. Lancet Neurol. 2010; 9:895–905. PMID: 20723846.
Article30. Henon H, Durieu I, Guerouaou D, Lebert F, Pasquier F, Leys D. Poststroke dementia: incidence and relationship to prestroke cognitive decline. Neurology. 2001; 57:1216–1222. PMID: 11591838.
Article31. Veerbeek JM, Kwakkel G, van Wegen EE, Ket JC, Heymans MW. Early prediction of outcome of activities of daily living after stroke: a systematic review. Stroke. 2011; 42:1482–1488. PMID: 21474812.32. Jorgensen HS, Nakayama H, Raaschou HO, Olsen TS. Recovery of walking function in stroke patients: the Copenhagen Stroke Study. Arch Phys Med Rehabil. 1995; 76:27–32. PMID: 7811170.33. Lin DD, Kleinman JT, Wityk RJ, Gottesman RF, Hillis AE, Lee AW, et al. Crossed cerebellar diaschisis in acute stroke detected by dynamic susceptibility contrast MR perfusion imaging. AJNR Am J Neuroradiol. 2009; 30:710–715. PMID: 19193758.
Article34. Watanabe H, Niwa N, Kanai M, Sasaki S. The relation between crossed cerebellar blood flow and severities of hemiplegia: a technetium-(99m) hexamethylpropylene amine oxine SPECT study using Patlak plot method. Tohoku J Exp Med. 1996; 179:177–181. PMID: 8888506.
Article35. De Reuck J, Decoo D, Lemahieu I, Strijckmans K, Goethals P, Van Maele G. Crossed cerebellar diaschisis after middle cerebral artery infarction. Clin Neurol Neurosurg. 1997; 99:11–16. PMID: 9107461.
Article36. Kwakkel G, Wagenaar RC, Koelman TW, Lankhorst GJ, Koetsier JC. Effects of intensity of rehabilitation after stroke: a research synthesis. Stroke. 1997; 28:1550–1556. PMID: 9259747.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Crossed cerebello-cerebral diaschisis in cerebellar infraction
- Diaschisis and Motor Recovery in Stroke Patients
- Vascular Hyperemia and Crossed Cerebellar Diaschisis in MELAS Patient Presented as Stroke-Like Episode and Seizure
- Crossed Cerebellar and Cerebral Cortical Diaschisis in Basal Ganglia Hemorrhage
- Crossed Cerebellar Diaschisis in Complex Partial Status Epilepticus